Themed collection Celebrating 70 Years of Shanghai Institute of Organic Chemistry

Total synthesis of strictamine: a tutorial for novel and efficient synthesis
The novelty and efficiency of the overall synthetic route are the key values being imparted to the younger generation of synthetic chemists. In this Highlights, synthesis of strictamine was tutored to students on the creativity of synthetic design.
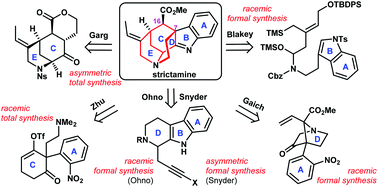
Org. Chem. Front., 2018,5, 447-452
https://doi.org/10.1039/C7QO00837F
Copper-mediated/-catalyzed fluorination reactions: new routes to aryl fluorides
Recent advances in direct fluorination of aromatic compounds by copper catalysts have been highlighted.

Org. Chem. Front., 2014,1, 430-433
https://doi.org/10.1039/C4QO00003J
Lu's [3 + 2] cycloaddition of allenes with electrophiles: discovery, development and synthetic application
The discovery of Lu's [3 + 2] cycloaddition reaction is briefly introduced, and the recent important developments and synthetic applications of Lu's [3 + 2] cycloaddition reaction are highlighted and overviewed.
![Graphical abstract: Lu's [3 + 2] cycloaddition of allenes with electrophiles: discovery, development and synthetic application](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C7QO00285H&imageInfo.ImageIdentifier.Year=2017)
Org. Chem. Front., 2017,4, 1876-1890
https://doi.org/10.1039/C7QO00285H
Recent applications of chiral N-tert-butanesulfinyl imines, chiral diene ligands and chiral sulfur–olefin ligands in asymmetric synthesis
The highly efficient asymmetric reactions of chiral sulfinyl auxiliary, diene ligands and sulfur–olefin ligands are presented.
Org. Chem. Front., 2015,2, 73-89
https://doi.org/10.1039/C4QO00245H
Conquering three-carbon axial chirality of allenes
Axially chiral allenes are an important class of molecules widely existing in nature and also valuable chiral building blocks for organic synthesis. With more and more new protocols for racemic allenes and new chiral ligands being developed, a wide variety of novel and challenging transformations have been recently unveiled for the synthesis of this type of highly versatile compounds. In this review, we present a critical account of the enantioselective synthesis of axially chiral allenes.
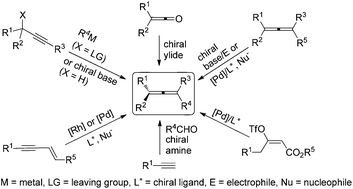
Org. Chem. Front., 2014,1, 1210-1224
https://doi.org/10.1039/C4QO00208C
Catalytic asymmetric allylation of carbonyl compounds and imines with allylic boronates
Enantioselective allylation is a highly used organic reaction to prepare chiral homoallylic alcohols and amines, which serve as important building blocks in the synthesis of a variety of natural products and pharmaceuticals.

Org. Chem. Front., 2014,1, 303-320
https://doi.org/10.1039/C3QO00081H
Dimerization–cyclization reactions of isocyanoaryl-tethered alkylidenecyclobutanes via a triplet biradical mediated process
A triplet biradical mediated dimerization–cyclization reaction of isocyanoaryl-tethered alkylidenecyclobutanes to construct macrocyclic skeletons including dihydroquinoline and quinoline units has been reported.
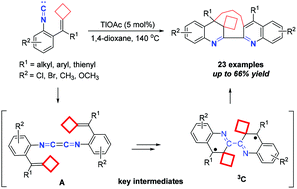
Org. Chem. Front., 2020,7, 2634-2643
https://doi.org/10.1039/D0QO00878H
DFT study on the E-stereoselective reductive A3-coupling reaction of terminal alkynes with aldehydes and 3-pyrroline
The mechanism of the Cu(I)-catalyzed reductive A3-coupling reaction of terminal alkynes with aldehydes and 3-pyrroline for the synthesis of E-allylic amines has been studied by DFT calculations.
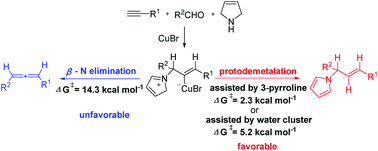
Org. Chem. Front., 2020,7, 2047-2054
https://doi.org/10.1039/D0QO00564A
Isolation and structure determination of two new nosiheptide-type compounds provide insights into the function of the cytochrome P450 oxygenase NocV in nocathiacin biosynthesis
Two new nosiheptide-type compounds isolated from an engineered strain provide insights into the function of the cytochrome P450 oxygenase NocV.
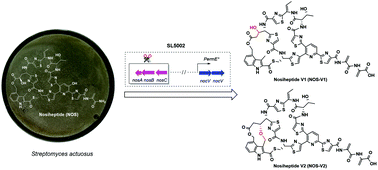
Org. Chem. Front., 2020,7, 584-589
https://doi.org/10.1039/C9QO01328H
Cascade cyclization reactions of alkylidenecyclopropanes for the construction of polycyclic lactams and lactones by visible light photoredox catalysis
A facile method for the synthesis of seven- and eight-membered ring-containing polycyclic lactams and lactones by visible light photocatalysis has been developed.
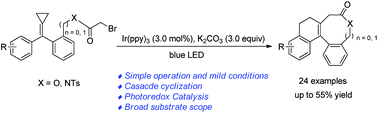
Org. Chem. Front., 2020,7, 374-379
https://doi.org/10.1039/C9QO01360A
Visible-light-induced radical hydrodifluoromethylation of alkenes
Visible-light-induced radical hydrodifluoromethylation of alkenes with the phosphonium salt [Ph3P+CF2H Br−] under transition-metal-free conditions is described.

Org. Chem. Front., 2019,6, 3580-3583
https://doi.org/10.1039/C9QO00919A
n BuLi-promoted anti-Markovnikov selective hydroboration of unactivated alkenes and internal alkynes
A novel anti-Markovnikov selective hydroboration of alkenes and alkynes with HBpin using nBuLi as an effective initiator is described.
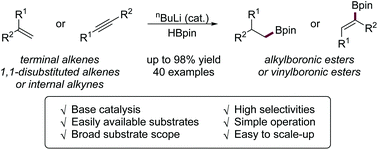
Org. Chem. Front., 2019,6, 2949-2953
https://doi.org/10.1039/C9QO00750D
Rhodium(II)-catalyzed divergent intramolecular tandem cyclization of N- or O-tethered cyclohexa-2,5-dienones with 1-sulfonyl-1,2,3-triazole: synthesis of cyclopropa[cd]indole and benzofuran derivatives
A novel rhodium(II)-catalyzed divergent intramolecular tandem cyclization of N- or O-tethered cyclohexa-2,5-dienones and 1-sulfonyl-1,2,3-triazole is disclosed, affording cyclopropa[cd]indole and benzofuran derivatives in moderate to good yields.
![Graphical abstract: Rhodium(ii)-catalyzed divergent intramolecular tandem cyclization of N- or O-tethered cyclohexa-2,5-dienones with 1-sulfonyl-1,2,3-triazole: synthesis of cyclopropa[cd]indole and benzofuran derivatives](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00714H&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 2884-2891
https://doi.org/10.1039/C9QO00714H
Ligandless nickel-catalyzed transfer hydrogenation of alkenes and alkynes using water as the hydrogen donor
The first general route for nickel-catalyzed transfer hydrogenation reaction of alkenes and alkynes using water as the hydrogen source has been developed.
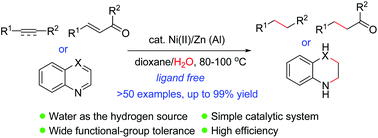
Org. Chem. Front., 2019,6, 2619-2623
https://doi.org/10.1039/C9QO00616H
Rh-Catalyzed intramolecular decarbonylative cyclization of ortho-formyl group tethered alkylidenecyclopropanes (ACPs) for the construction of 2-methylindenes
A Rh-catalyzed intramolecular cascade decarbonylative coupling reaction of ortho-formyl group-tethered alkylidenecyclopropanes has been developed, affording indene derivatives in moderate to good yields.

Org. Chem. Front., 2019,6, 2667-2671
https://doi.org/10.1039/C9QO00614A
Phosphine-catalyzed fixation of CO2 with γ-hydroxyl alkynone under ambient temperature and pressure: kinetic resolution and further conversion
Phosphine-catalyzed fixation of CO2 with γ-hydroxyl alkynone under ambient temperature and pressure was achieved and the first example of chiral phosphine catalyzed kinetic resolution of propargyl alcohols via carbon dioxide fixation was demonstrated.
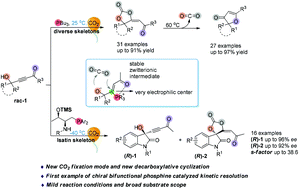
Org. Chem. Front., 2019,6, 2420-2429
https://doi.org/10.1039/C9QO00642G
Bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]−: synthesis, characterization and its application for trifluoromethylation of activated heteroaryl bromides, chlorides and iodides
The synthesis and characterization of a bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]− and its reactions with a variety of activated heteroaryl bromides, chlorides and iodides were described.
![Graphical abstract: Bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]−: synthesis, characterization and its application for trifluoromethylation of activated heteroaryl bromides, chlorides and iodides](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00527G&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 2324-2328
https://doi.org/10.1039/C9QO00527G
Synthesis and antiproliferative activities of OSW-1 analogues bearing 2-acylamino-xylose residues
We synthesized 38 OSW-1 analogues with 2-acylamino xylose residues and found that the antitumor activities could be greatly enhanced.
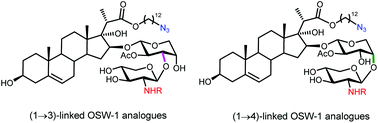
Org. Chem. Front., 2019,6, 2385-2391
https://doi.org/10.1039/C9QO00462A
Nickel-catalyzed highly regioselective hydrocyanation of alkenes with Zn(CN)2
The first general and regioselective nickel-catalyzed hydrocyanation of terminal alkenes with Zn(CN)2 using an air-stable and inexpensive nickel(II) salt as the precatalyst has been developed.

Org. Chem. Front., 2019,6, 2037-2042
https://doi.org/10.1039/C9QO00396G
Rh(I)-Catalyzed intramolecular [3 + 2] cycloaddition reactions of yne-vinylidenecyclopropanes
A Rh(I)-catalyzed intramolecular [3 + 2] cycloaddition reaction of yne-vinylidenecyclopropanes has been developed, providing fused [6.5]-bicyclic products in moderate to good yields.
![Graphical abstract: Rh(i)-Catalyzed intramolecular [3 + 2] cycloaddition reactions of yne-vinylidenecyclopropanes](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00343F&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 1816-1820
https://doi.org/10.1039/C9QO00343F
Pd-Catalyzed coupling reaction of cyclobutanols with propargylic carbonates
Pd-Catalyzed ring opening coupling reaction of cyclobutanols with propargylic carbonates afforded δ-allenyl ketones efficiently.
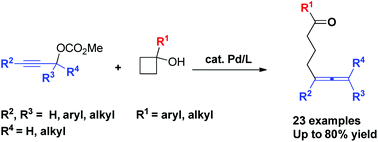
Org. Chem. Front., 2019,6, 1757-1761
https://doi.org/10.1039/C9QO00192A
Optically active N-alkyl aziridines via stereospecific reductive cyclization of α-mesylated acetamides
An efficient method for the synthesis of optically active N-alkyl aziridines has been realized for the first time by stereospecific reductive cyclization of optically active α-mesylated acetamides. A series of optically active N-alkyl aziridines are prepared in moderate to good yields and excellent ees.
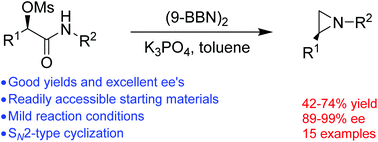
Org. Chem. Front., 2018,5, 2723-2727
https://doi.org/10.1039/C8QO00774H
Rhodium-catalyzed highly diastereoselective intramolecular [4 + 2] cycloaddition of 1,3-disubstituted allene-1,3-dienes
RhCl(PPh3)3-catalyzed [4 + 2] intramolecular cycloaddition of allene-1,3-dienes afforded cis-fused [3.4.0]-bicyclic products with three chiral centers in good yields with excellent chemo- and diastereoselectivity.
![Graphical abstract: Rhodium-catalyzed highly diastereoselective intramolecular [4 + 2] cycloaddition of 1,3-disubstituted allene-1,3-dienes](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C8QO00650D&imageInfo.ImageIdentifier.Year=2018)
Org. Chem. Front., 2018,5, 2680-2684
https://doi.org/10.1039/C8QO00650D
Palladium-catalyzed reductive electrocarboxylation of allyl esters with carbon dioxide
Palladium-catalyzed regioselective electrocarboxylation of homostyrenyl acetates with CO2 has been successfully developed, providing α-aryl carboxylic acids with good selectivity and yield.
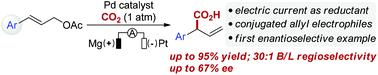
Org. Chem. Front., 2018,5, 2244-2248
https://doi.org/10.1039/C8QO00507A
Palladium-catalyzed intermolecular dearomatic allenylation of hydrocycloalk[b]indoles with 2,3-allenyl carbonates
A palladium-catalyzed allenylation of hydrocycloalk[b]indoles with 2,3-allenyl carbonates provides functionalized allenes containing an indoline.
![Graphical abstract: Palladium-catalyzed intermolecular dearomatic allenylation of hydrocycloalk[b]indoles with 2,3-allenyl carbonates](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C8QO00163D&imageInfo.ImageIdentifier.Year=2018)
Org. Chem. Front., 2018,5, 1664-1669
https://doi.org/10.1039/C8QO00163D
Catalyst-controlled synthesis of 4-amino-isoquinolin-1(2H)-one and oxazole derivatives
A facile synthetic method for the preparation of oxazole and 4-amino-isoquinolin-1(2H)-one derivatives has been developed from the reaction of N-(pivaloyloxy)-amides and ynamides by using different metal catalysts.
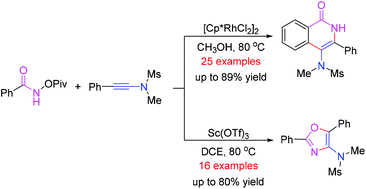
Org. Chem. Front., 2018,5, 1466-1470
https://doi.org/10.1039/C8QO00125A
Tri- and di-fluoroethylation of alkenes by visible light photoredox catalysis
The photoredox-catalyzed solvent-dependent tri-/di-fluoroethylation of alkenes with sulfonium salts (Ph2S+CH2RF TfO−) (RF = CF3 or HCF2) is described.
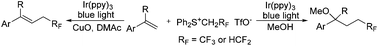
Org. Chem. Front., 2018,5, 1452-1456
https://doi.org/10.1039/C8QO00082D
Base-promoted [3 + 3] cyclization of cyclopropenones and cyclopropenethiones with amides for the synthesis of 6H-1,3-oxazin-6-ones and 6H-1,3-thiazin-6-ones
A facile synthesis of 6H-1,3-oxazin-6-ones and 6H-1,3-thiazin-6-ones has been achieved in the base-promoted [3 + 3] cyclization of cyclopropenones and cyclopropenethiones with amides.
![Graphical abstract: Base-promoted [3 + 3] cyclization of cyclopropenones and cyclopropenethiones with amides for the synthesis of 6H-1,3-oxazin-6-ones and 6H-1,3-thiazin-6-ones](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C8QO00091C&imageInfo.ImageIdentifier.Year=2018)
Org. Chem. Front., 2018,5, 1267-1271
https://doi.org/10.1039/C8QO00091C
Transition metal-free aminofluorination of β,γ-unsaturated hydrazones: base-controlled regioselective synthesis of fluorinated dihydropyrazole and tetrahydropyridazine derivatives
Various base-controlled regioselective reactions of β,γ-unsaturated hydrazones with Selectfluor were achieved under transition metal-free conditions.
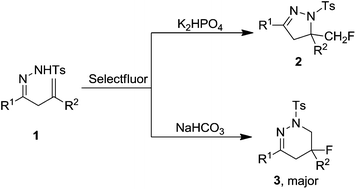
Org. Chem. Front., 2018,5, 1155-1159
https://doi.org/10.1039/C7QO01105A
Copper(II)-catalyzed trifluoromethylation of iodoarenes using Chen's reagent
The utility of Chen's reagent in trifluoromethylation revisited.

Org. Chem. Front., 2018,5, 1143-1147
https://doi.org/10.1039/C8QO00025E
Photoinduced hydroxylperfluoroalkylation of styrenes
“No Catalyst is Better” is confirmed in the hydroxylperfluoroalkylation of styrenes with perfluoroalkyl iodides under irradiation by visible light.

Org. Chem. Front., 2018,5, 1045-1048
https://doi.org/10.1039/C7QO00946A
Organocatalytic asymmetric synthesis of cornolactones A and B, and formal synthesis of brasoside and littoralisone
The asymmetric total or formal syntheses of cornolactone A, cornolactone B, brasoside and littoralisone were accomplished in short steps.
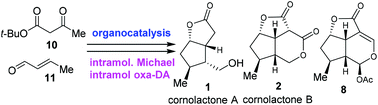
Org. Chem. Front., 2018,5, 358-360
https://doi.org/10.1039/C7QO00843K
Tunable regiodivergent phosphine-catalyzed [3 + 2] cycloaddition of alkynones and trifluoroacetyl phenylamides
The regiodivergent phosphine-catalyzed [3 + 2] cycloaddition of 2-(2,2,2-trifluoroacetyl)phenylamides and alkynones have been achieved through adjusting the acidities of substrates, changing reaction temperature and addition of protic additive H2O. The detailed mechanism was systematically studied.
![Graphical abstract: Tunable regiodivergent phosphine-catalyzed [3 + 2] cycloaddition of alkynones and trifluoroacetyl phenylamides](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C7QO00512A&imageInfo.ImageIdentifier.Year=2017)
Org. Chem. Front., 2017,4, 2392-2402
https://doi.org/10.1039/C7QO00512A
Concise asymmetric total synthesis of (−)-patchouli alcohol
An asymmetric total synthesis of (−)-patchouli alcohol was realized featuring an enantioselective organocatalytic [4 + 2] approach to the [2.2.2] bicyclic core.
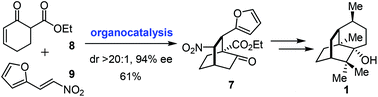
Org. Chem. Front., 2017,4, 2031-2033
https://doi.org/10.1039/C7QO00459A
Fe-Catalyzed insertion of fluoromethylcarbenes generated from sulfonium salts into X–H bonds (X = Si, C, P)
The Fe-catalyzed insertion of fluoromethylcarbenes including trifluoromethylcarbene and difluoromethylcarbene into X–H (X = Si, C and P) bonds is described.
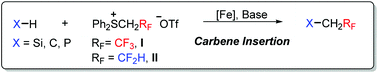
Org. Chem. Front., 2017,4, 1917-1920
https://doi.org/10.1039/C7QO00430C
Identifying a cobalt catalyst for highly selective hydrosilylation of allenes
An efficient method of cobalt-catalyzed allene-hydrosilylation is developed. The less-substituted C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond was reacted and the reaction demonstrated Z-stereoselectivity.
C bond was reacted and the reaction demonstrated Z-stereoselectivity.
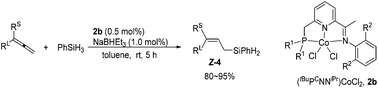
Org. Chem. Front., 2017,4, 1829-1832
https://doi.org/10.1039/C7QO00497D
Efficient trifluoromethylation via the cyclopropanation of allenes and subsequent C–C bond cleavage
Trifluoromethyl has been efficiently incorporated into organic skeletons via allene cyclization and C–C bond cleavage.
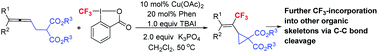
Org. Chem. Front., 2017,4, 1762-1767
https://doi.org/10.1039/C7QO00419B
Pincer cobalt complex-catalyzed Z-selective hydrosilylation of terminal alkynes
Co-Catalyzed Z-selective hydrosilylation of terminal alkynes produces (Z)-β-vinylsilanes, which are further converted to (Z)-disubstituted alkenes by Pd-catalyzed cross couplings.
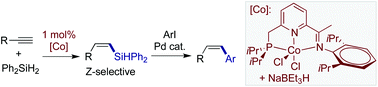
Org. Chem. Front., 2017,4, 1517-1521
https://doi.org/10.1039/C7QO00250E
Copper-catalyzed highly selective approach to 2-boroallylic silanes from allenylsilanes
A highly selective copper-catalyzed borylcupration of 1,2-allenylsilanes affords 2-borylallylsilanes with many synthetically attractive functional groups by applying BIPHEP as the ligand has been developed.
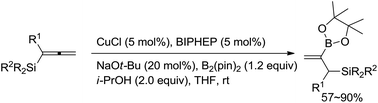
Org. Chem. Front., 2017,4, 1261-1265
https://doi.org/10.1039/C7QO00135E
Enantioselective synthesis of naturally occurring isoquinoline alkaloids: (S)-(−)-trolline and (R)-(+)-oleracein E
The highly efficient asymmetric synthesis of a pair of enantiomers, (S)-(−)-trolline and (R)-(+)-oleracein E, has been achieved in five steps.
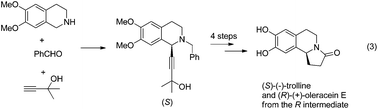
Org. Chem. Front., 2017,4, 958-966
https://doi.org/10.1039/C7QO00062F
Synthesis of largely π-extended naphthalenediimides via C–H activation towards highly soluble and narrow band-gap organic optoelectronic materials
Largely π-extended naphthalenediimides were synthesized by using efficient palladium catalyzed C–H/C–H homocouplings, with the yields of up to 94%.
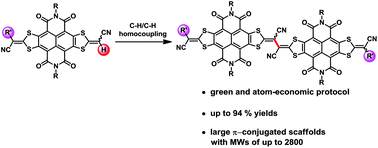
Org. Chem. Front., 2017,4, 823-827
https://doi.org/10.1039/C7QO00061H
Palladium-catalyzed asymmetric allylic amination: enantioselective synthesis of chiral α-methylene substituted β-aminophosphonates
Palladium catalyzed asymmetric allylic amination of 2-diethylphosphonate-substituted allylic acetates in the presence of SKP ligands afforded the corresponding chiral β-aminophosphonates in good yields, high regioselectivities, and excellent enantioselectivities.

Org. Chem. Front., 2017,4, 271-276
https://doi.org/10.1039/C6QO00597G
A facile approach for the trifluoromethylthiolation of methylenecyclopropanes
Trifluoromethylthiolation of methylenecyclopropanes (MCPs) has been developed by using AgSCF3/Na2S2O8 as a trifluoromethylthiolation source (SCF3) to give trifluoromethylthiolated 1,2-dihydronaphthalene derivatives in moderate to good yields.
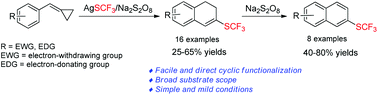
Org. Chem. Front., 2017,4, 86-90
https://doi.org/10.1039/C6QO00536E
Molecular engineering of thiostrepton via single “base”-based mutagenesis to generate side ring-derived variants
Six thiostrepton (TSR) analogs, siomycin (SIO) and SIO-Dha2Ser were produced in the engineered TSR-producing strain by using a single “base”-based mutagenesis strategy.
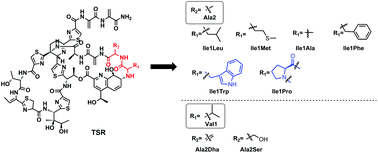
Org. Chem. Front., 2016,3, 1254-1258
https://doi.org/10.1039/C6QO00320F
Base-induced synthesis of N-dialkylaminomethyl-2H-1,2,3-triazoles from N-sulfonyl-1,2,3-triazoles
A facile synthetic method to access N-dialkylaminomethyl-2H-1,2,3-triazoles has been developed from N-sulfonyl-1,2,3-triazoles by a base induced reaction. Moreover, 4-phenyl-1H-1,2,3-triazoles can also give the same product even in the absence of a base at high temperature.
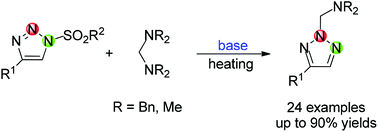
Org. Chem. Front., 2016,3, 744-748
https://doi.org/10.1039/C6QO00102E
Concurrent modifications of the C-terminus and side ring of thiostrepton and their synergistic effects with respect to improving antibacterial activities
Five new C-terminally methylated TSR derivatives that varied in side-ring structure were obtained via the chemical feeding of quinaldic acid analogs to a double-mutant strain ΔtsrB/T.
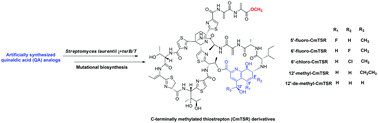
Org. Chem. Front., 2016,3, 496-500
https://doi.org/10.1039/C5QO00433K
Synthesis of 1,1-diboronate esters by cobalt-catalyzed sequential hydroboration of terminal alkynes
Co-catalyzed sequential hydroborations of alkyl and aryl alkynes with pinacolborane produce 1,1-diboronate esters with high selectivity and yields.
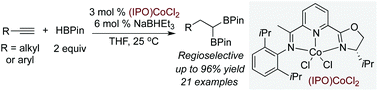
Org. Chem. Front., 2016,3, 434-438
https://doi.org/10.1039/C5QO00426H
A concise total synthesis of sespenine, a structurally unusual indole terpenoid from Streptomyces
A ten-step (the longest linear sequence) total synthesis of sespenine was accomplished.
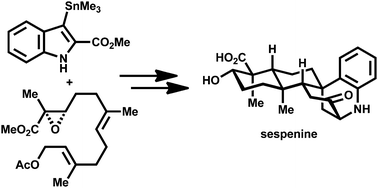
Org. Chem. Front., 2016,3, 368-374
https://doi.org/10.1039/C5QO00416K
Asymmetric total synthesis of Lycopodium alkaloids α-obscurine, N-desmethyl-α-obscurine, β-obscurine and N-desmethyl-β-obscurine
We report herein the asymmetric total synthesis of α-obscurine (1), β-obscurine (2), N-desmethyl-α-obscurine (3), and N-desmethyl-β-obscurine (4).
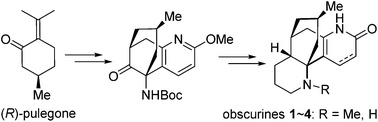
Org. Chem. Front., 2016,3, 62-65
https://doi.org/10.1039/C5QO00355E
Divergent synthesis of indole-fused polycycles via Rh(II)-catalyzed intramolecular [3 + 2] cycloaddition and C–H functionalization of indolyltriazoles
Intramolecular formal [3 + 2] cycloaddition and C–H functionalization of N-sulfonyl triazoles with indoles have been developed to synthesize potential indole-fused polycycles.
![Graphical abstract: Divergent synthesis of indole-fused polycycles via Rh(ii)-catalyzed intramolecular [3 + 2] cycloaddition and C–H functionalization of indolyltriazoles](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C5QO00216H&imageInfo.ImageIdentifier.Year=2015)
Org. Chem. Front., 2015,2, 1516-1520
https://doi.org/10.1039/C5QO00216H
Cu-Mediated 2,2,2-trifluoroethylation of terminal alkynes using 1,1-dichloro-2,2,2-trifluoroethane (HCFC-123)
Cu-Promoted coupling reactions of HCFC-123 with terminal alkynes give trifluoroethylated internal alkynes.
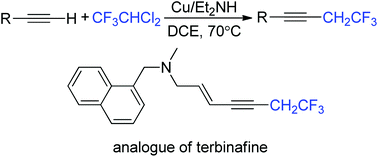
Org. Chem. Front., 2015,2, 1379-1387
https://doi.org/10.1039/C5QO00210A
Efficient synthesis of P-chiral biaryl phosphonates by stereoselective intramolecular cyclization
A series of P-chiral biaryl phosphonates were efficiently synthesized from diaryl 2-bromo arylphosphonates in high yields (up to 92%) and good enantioselectivities (up to 88% ee) through a palladium-catalyzed desymmetric intramolecular cyclization with a P-chiral biaryl monophosphorus ligand.
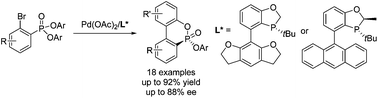
Org. Chem. Front., 2015,2, 1342-1345
https://doi.org/10.1039/C5QO00142K
Chemoselective and fast decarboxylative allylation by photoredox catalysis under mild conditions
A chemoselective and fast decarboxylative allylation is developed by photoredox catalysis to build C(sp3)–allyl bonds under mild conditions.
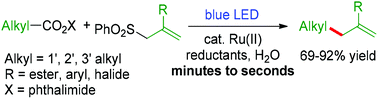
Org. Chem. Front., 2015,2, 1352-1355
https://doi.org/10.1039/C5QO00187K
Enantioselective nickel-catalyzed alkylative alkyne–aldehyde cross-couplings
An efficient asymmetric nickel-catalyzed alkylative alkyne–aldehyde cross-coupling is developed by employing BI-DIME as the P-chiral monophosphorus ligand, allowing rapid access to a series of chiral tetra-substituted olefinic allylic alcohols in high yields and good to excellent ees.
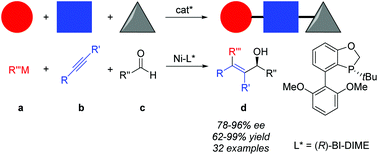
Org. Chem. Front., 2015,2, 1322-1325
https://doi.org/10.1039/C5QO00148J
Copper-mediated oxidative difluoromethylation of terminal alkynes with TMSCF2H
A copper-mediated oxidative coupling of terminal alkynes and TMSCF2H has been described for the preparation of various difluoromethylated alkynes.
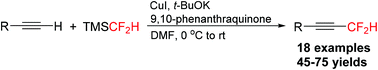
Org. Chem. Front., 2015,2, 1022-1025
https://doi.org/10.1039/C5QO00186B
Pd(II)-catalyzed intermolecular enantioselective hydroamination of styrenes
A Pd-catalyzed intermolecular asymmetric hydroamination of styrenes was developed with pyridine-oxazoline as the chiral ligand.
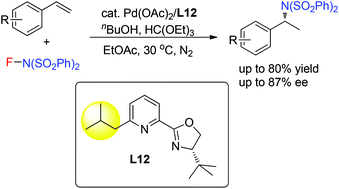
Org. Chem. Front., 2015,2, 819-822
https://doi.org/10.1039/C5QO00096C
Efficient catalytic enantioselective Nazarov cyclizations of divinyl ketoesters
An efficient catalytic enantioselective Nazarov cyclization of divinyl ketoesters is developed using a BOX/Cu(II) catalyst, affording various optically active cyclopent-2-enone esters.
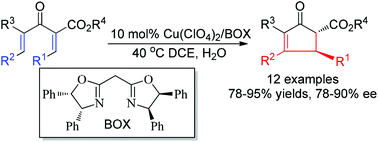
Org. Chem. Front., 2015,2, 811-814
https://doi.org/10.1039/C5QO00099H
Enantioselective synthesis of 4,5,6,7-tetrahydroindoles via olefin cross-metathesis/intramolecular Friedel–Crafts alkylation reaction of pyrroles
A sequential catalysis involving olefin cross-metathesis/asymmetric intramolecular Friedel–Crafts alkylation of pyrrole derivatives has been developed.
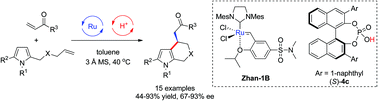
Org. Chem. Front., 2015,2, 476-480
https://doi.org/10.1039/C5QO00034C
Synthesis of monolateral and bilateral sulfur-heterocycle fused naphthalene diimides (NDIs) from monobromo and dibromo NDIs
Monolateral and bilateral sulfur-heterocycle fused naphthalene diimides (NDIs) are successfully synthesized from monobromo and dibromo NDIs via nucleophilic aromatic substitution reactions and the subsequent oxidative aromatization processes.
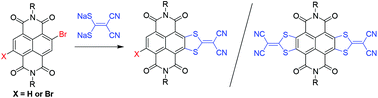
Org. Chem. Front., 2015,2, 372-377
https://doi.org/10.1039/C4QO00252K
Copper(I)-catalyzed enantioselective hydroboration of cyclopropenes: facile synthesis of optically active cyclopropylboronates
Copper(I)-catalyzed enantioselective hydroboration of 3-aryl substituted cyclopropene-3-carboxylate is described, providing chiral cyclopropylboronates with excellent enantioselectivities (89–95% ee) in moderate to high yields (55–86%).
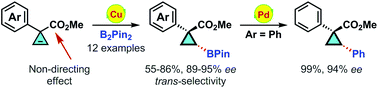
Org. Chem. Front., 2014,1, 1116-1122
https://doi.org/10.1039/C4QO00157E
A sidearm-assisted phosphine for catalytic ylide intramolecular cyclopropanation
The first phosphine-catalyzed cyclopropanation reaction via covalent ylide catalysis has been realized with high efficiency in the presence of sidearm-assisted phosphine catalysts.
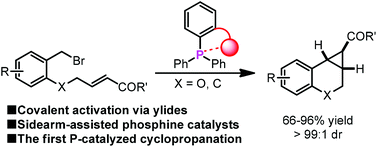
Org. Chem. Front., 2014,1, 1035-1039
https://doi.org/10.1039/C4QO00232F
Two- and three-coordinate formal iron(I) compounds featuring monodentate aminocarbene ligands
Monodentate aminocarbene ligands, both NHC and cAAC, are capable of stabilizing low-coordinate iron(I) compounds.

Org. Chem. Front., 2014,1, 1040-1044
https://doi.org/10.1039/C4QO00175C
Copper-mediated difluoromethylation of electron-poor aryl iodides at room temperature
The direct difluoromethylation of electron-poor aryl iodides, as well as heteroaryl and β-styryl iodides, using TMSCF2H at ambient temperature was developed.
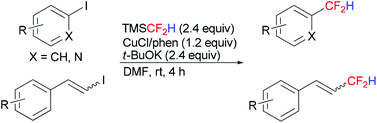
Org. Chem. Front., 2014,1, 774-776
https://doi.org/10.1039/C4QO00153B
Palladium-catalyzed cascade cyclization for the construction of spiro-N,O-acetals
A facile Pd-catalyzed cascade cyclization of N-alkenylamine and pyruvic acids has been developed to construct spiro-N,O-acetals efficiently.
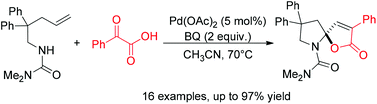
Org. Chem. Front., 2014,1, 289-293
https://doi.org/10.1039/C4QO00002A
Asymmetric hydrogenation of α-arylacrylic and β-arylbut-3-enoic acids catalyzed by a Rh(I) complex of a monodentate secondary phosphine oxide ligand
Catalytic asymmetric hydrogenation of α-arylacrylic and β-arylbut-3-enoic acids using Rh(I) complexes of secondary phosphine oxides provided the corresponding chiral acids with up to 97% ee.
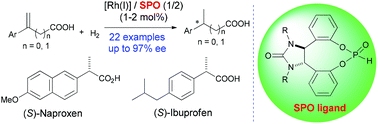
Org. Chem. Front., 2014,1, 155-160
https://doi.org/10.1039/C3QO00042G
N-alkylation of indole via ring-closing metathesis/isomerization/Mannich cascade under ruthenium/chiral phosphoric acid sequential catalysis
A sequential catalysis by combining a Ru catalyst with chiral phosphoric acid has been utilized for N-alkylation of indole.
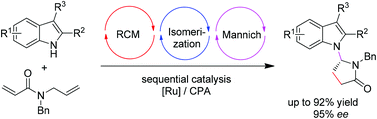
Org. Chem. Front., 2014,1, 39-43
https://doi.org/10.1039/C3QO00008G
Transition-metal-free, room-temperature radical azidofluorination of unactivated alkenes in aqueous solution
Transition-metal-free radical azidofluorination of unactivated alkenes was successfully achieved in aqueous solution at room temperature with excellent regioselectivity and functional group compatibility.

Org. Chem. Front., 2014,1, 100-104
https://doi.org/10.1039/C3QO00037K
The remarkable role of solvent in reaction mechanism studies by electrospray mass spectrometry
The solvent assistance technologies in detecting reactive intermediates in complicated reaction solutions using electrospray mass spectrometry were summarized and discussed.
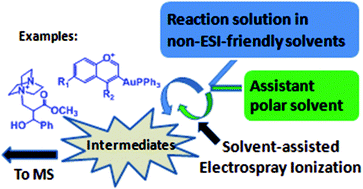
Org. Chem. Front., 2015,2, 990-994
https://doi.org/10.1039/C5QO00154D
Difluoromethyl 2-pyridyl sulfone: a versatile carbonyl gem-difluoroolefination reagent
The gem-difluoroolefination of carbonyl compounds with difluoromethyl 2-pyridyl sulfone (2-PySO2CF2H) has been summarized in this tutorial account.
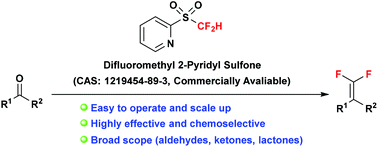
Org. Chem. Front., 2015,2, 163-168
https://doi.org/10.1039/C4QO00291A
The highly enantioselective catalytic aza-Morita–Baylis–Hillman reaction
Chiral phosphine, amine or metal catalyzed asymmetric aza-MBH reactions of various imines with different Michael acceptors in recent decades have been discussed and the perspectives offered by these new developments have also been demonstrated.
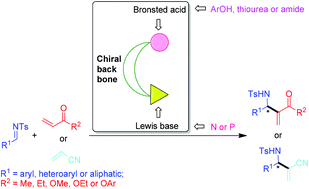
Org. Chem. Front., 2014,1, 587-595
https://doi.org/10.1039/C4QO00026A
About this collection
Organic Chemistry Frontiers is delighted to commemorate the 70th anniversary of Shanghai Institute of Organic Chemistry (SIOC) with this special web collection. SIOC is a co-owner of Organic Chemistry Frontiers, together with the Chinese Chemical Society and the Royal Society of Chemistry. In honour of its 70th anniversary, we selected 70 articles from over 200 papers that SIOC researchers have published into Organic Chemistry Frontiers since the journal was launched. Articles in the collection are free to read until the end of October.