Themed collection Sustainable synthesis

Recent approaches in the organocatalytic synthesis of pyrroles
Recent advances for the synthesis of pyrroles by employing organocatalysis are summarized in this review article.
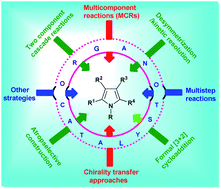
RSC Adv., 2021,11, 13585-13601
https://doi.org/10.1039/D1RA01690C
Copper-free Sonogashira cross-coupling reactions: an overview
The Sonogashira reaction is a cross-coupling reaction of a vinyl or aryl halide with a terminal alkyne to form a C–C bond.
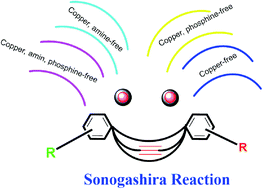
RSC Adv., 2021,11, 6885-6925
https://doi.org/10.1039/D0RA10575A
Microwave assisted synthesis of five membered nitrogen heterocycles
Microwave assisted synthesis of N-heterocycles with short reaction time, high yields and high product purities along with a decrease in the rate of by-product formation.

RSC Adv., 2020,10, 36031-36041
https://doi.org/10.1039/D0RA05150K
Cycloaddition of atmospheric CO2 to epoxides under solvent-free conditions: a straightforward route to carbonates by green chemistry metrics
The conversion of CO2 into value-added organic compounds has received more attention over recent years since this gas is one of the major greenhouse gases, as well as an abundant, inexpensive, nontoxic, nonflammable and renewable one-carbon (C1) resource.
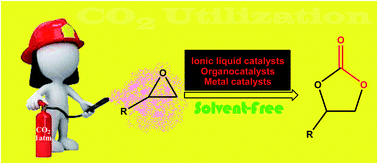
RSC Adv., 2019,9, 3884-3899
https://doi.org/10.1039/C8RA10233C
Recent advances of titanium dioxide (TiO2) for green organic synthesis
This review focuses on the recent development of TiO2 as a catalyst in green organic synthesis including in hydrodeoxygenation, hydrogenation, esterification/transesterification, the water–gas shift reaction, and visible light-induced organic transformation.

RSC Adv., 2016,6, 108741-108754
https://doi.org/10.1039/C6RA22894A
A review on the advancement of ether synthesis from organic solvent to water
Ethers have been synthesized by different protocols, this review aims to bring the attention of scientists working in the field of industrial chemistry, green chemistry, and catalysis towards the potential future of ether synthesis in micellar media.

RSC Adv., 2016,6, 69605-69614
https://doi.org/10.1039/C6RA12914E
Advancement in methodologies for reduction of nitroarenes
Recent advancement in reduction methods of nitroarenes are reviewed. The different methods are classified based on the source of hydrogen utilized during reduction and the mechanism involved in the reduction process.

RSC Adv., 2015,5, 83391-83407
https://doi.org/10.1039/C5RA10076C
Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions
This review highlights the recent advances using deep eutectic solvents (DESs), deep eutectic ionic liquids (DEILs), low-melting mixtures (LMMs) or low transition temperature mixtures (LTTMs) as green media as well as catalysts in organic reactions.
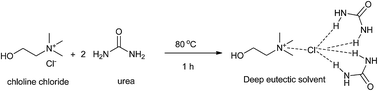
RSC Adv., 2015,5, 48675-48704
https://doi.org/10.1039/C5RA05746A
Horseradish peroxidase (HRP) as a tool in green chemistry
The horseradish peroxidase (HRP) potential in organic synthesis.
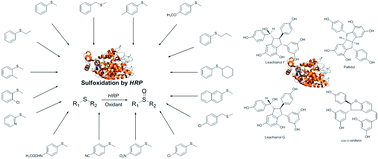
RSC Adv., 2014,4, 37244-37265
https://doi.org/10.1039/C4RA06094F
“The green side of the moon: ecofriendly aspects of organoselenium chemistry”
Key aspects for the development of a greener synthesis and the use of the organoselenium compounds.

RSC Adv., 2014,4, 31521-31535
https://doi.org/10.1039/C4RA04493B
Molecular iodine-catalyzed multicomponent reactions: an efficient catalyst for organic synthesis
Molecular iodine has broad transformation ability of functional groups and can be used widely in MCRs.
RSC Adv., 2013,3, 7182-7204
https://doi.org/10.1039/C3RA23461D
Mechanochemical polymerization – controlling a polycondensation reaction between a diamine and a dialdehyde in a ball mill
Mechanochemical polymerization displays a sustainable alternative to classical solvent-based approaches while also offering faster kinetics and an increased conversion.
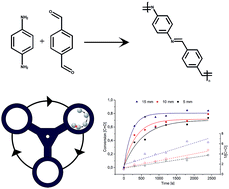
RSC Adv., 2016,6, 64799-64802
https://doi.org/10.1039/C6RA15677K
Green synthesis and characterisation of novel [1,3,4]thiadiazolo/benzo[4,5]thiazolo[3,2-a]pyrimidines via multicomponent reaction using vanadium oxide loaded on fluorapatite as a robust and sustainable catalyst
We synthesized material with different loading of vanadia on fluorapatite (V2O5/FAp), and excellent activity was showed for the synthesis of pyrimidine derivatives (90–97%) in ethanol medium for short reaction time.
![Graphical abstract: Green synthesis and characterisation of novel [1,3,4]thiadiazolo/benzo[4,5]thiazolo[3,2-a]pyrimidines via multicomponent reaction using vanadium oxide loaded on fluorapatite as a robust and sustainable catalyst](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0RA02298E&imageInfo.ImageIdentifier.Year=2020)
RSC Adv., 2020,10, 19803-19810
https://doi.org/10.1039/D0RA02298E
Synthesis of 3-selanylbenzo[b]furans promoted by SelectFluor®
This novel methodology provided a greener alternative to generate 3-substituted-benzo[b]furans mediate by Selectfluor® reagent. The formation of this electrophilic selenium species (RSe-F) was confirmed by heteronuclear NMR spectroscopy.
![Graphical abstract: Synthesis of 3-selanylbenzo[b]furans promoted by SelectFluor®](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0RA01907K&imageInfo.ImageIdentifier.Year=2020)
RSC Adv., 2020,10, 13975-13983
https://doi.org/10.1039/D0RA01907K
Pd nanoparticles on green support as dip-catalyst: a facile transfer hydrogenation of olefins and N-heteroarenes in water
A dip catalyst consisting of Pd nanoparticles immobilized on the surface of thin slices of jute-stem was fabricated. Its versatility as a hydrogen transfer agent for styrenyl, cyclic and unfunctionalized olefins and N-heterocycles was investigated.
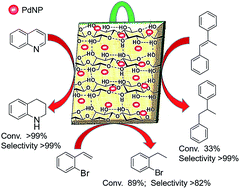
RSC Adv., 2019,9, 28199-28206
https://doi.org/10.1039/C9RA06285H
Metal- and photocatalyst-free synthesis of 3-selenylindoles and asymmetric diarylselenides promoted by visible light
A novel and sustainable procedure for the synthesis of 3-selenylindoles employing diorganyl diselenides and indoles or electron-rich arenes and promoted by visible light was developed.
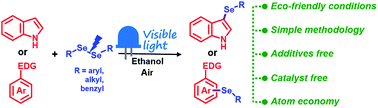
RSC Adv., 2019,9, 22685-22694
https://doi.org/10.1039/C9RA03642C
Environmentally benign and diastereoselective synthesis of 2,4,5-trisubstituted-2-imidazolines
A one-pot, membrane-assisted synthesis of 2,4,5-trisubstituted-2-imidazolines was developed, and substituent effects, reaction kinetics and mechanism by DFT were studied.
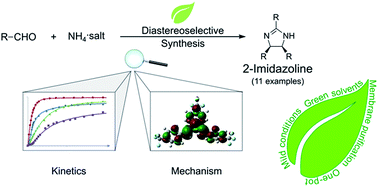
RSC Adv., 2017,7, 53278-53289
https://doi.org/10.1039/C7RA11827A
Laccase-catalyzed green synthesis and cytotoxic activity of novel pyrimidobenzothiazoles and catechol thioethers
Laccase-catalyzed green reaction between catechols and 2-thioxopyrimidin-4-ones delivers novel pyrimidobenzothiazoles and catechol thioethers with antiproliferative activities against HepG2 cell line.
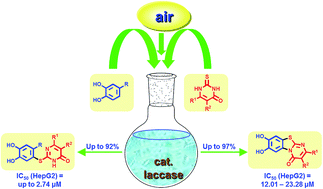
RSC Adv., 2017,7, 17427-17441
https://doi.org/10.1039/C6RA28102H
TBHP-promoted direct oxidation reaction of benzylic Csp3–H bonds to ketones
A metal-free oxidation system employing tert-butyl hydroperoxide (TBHP) has been developed for selective oxidation of structurally diverse benzylic sp3 C–H bonds.
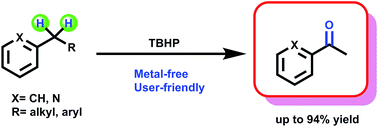
RSC Adv., 2017,7, 15176-15180
https://doi.org/10.1039/C7RA00352H
Solvent-free synthesis of 5-(aryl/alkyl)amino-1,2,4-triazines and α-arylamino-2,2′-bipyridines with greener prospects
A convenient approach towards 5-(aryl/alkyl)amino-1,2,4-triazines and α-arylamino-2,2′-bipyridines was developed following ipso-substitution of a cyano-group by amino groups and, finally, aza-Diels-Alder reaction with 1-morpholynocyclopentene.
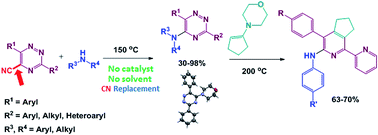
RSC Adv., 2017,7, 9610-9619
https://doi.org/10.1039/C6RA26305D
Introduction of taurine (2-aminoethanesulfonic acid) as a green bio-organic catalyst for the promotion of organic reactions under green conditions
Taurine (2-aminoethanesulfonic acid), a semi-essential amino acid that exists in the human body and numerous other living creatures, is used as a green bio-organic catalyst for the promotion of the Knoevenagel reaction between aldehydes and malononitrile.
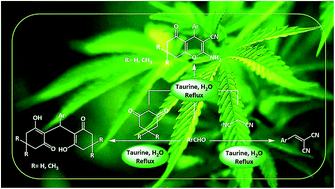
RSC Adv., 2016,6, 110190-110205
https://doi.org/10.1039/C6RA15432H
Green synthesis of tetrahydrobenzo[b]pyrans, pyrano[2,3-c]pyrazoles and spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles catalyzed by nano-structured diphosphate in water
Preparation of nanostructured diphosphate Na2CaP2O7. Avoiding of any acid catalysts. The prepared catalyst is reusable and eco-friendly. The method is easy and contains simple experimental procedure.
![Graphical abstract: Green synthesis of tetrahydrobenzo[b]pyrans, pyrano[2,3-c]pyrazoles and spiro[indoline-3,4′-pyrano[2,3-c]pyrazoles catalyzed by nano-structured diphosphate in water](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C6RA15800E&imageInfo.ImageIdentifier.Year=2016)
RSC Adv., 2016,6, 79128-79134
https://doi.org/10.1039/C6RA15800E
Poly(ethylene) glycols and mechanochemistry for the preparation of bioactive 3,5-disubstituted hydantoins
Mechanochemistry was effective for the preparation of 3,5-disubstituted hydantoins from α-amino methyl esters, using either 1,1′-carbonyldiimidazole (CDI) or alkyl isocyanates.
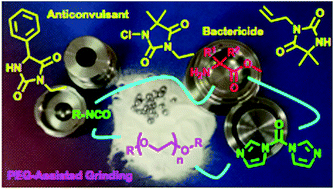
RSC Adv., 2016,6, 36978-36986
https://doi.org/10.1039/C6RA03222B
The first report on the eco-friendly synthesis of 5-substituted 1H-tetrazoles in PEG catalyzed by Cu(II) immobilized on Fe3O4@SiO2@L-arginine as a novel, recyclable and non-corrosive catalyst
Cu(II) immobilized on Fe3O4@SiO2@L-arginine as a novel nanostructured compound catalyzed the cycloaddition reaction of organic nitriles and sodium azide to produce 5-substituted 1H-tetrazoles.
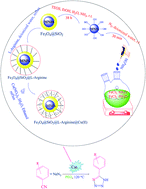
RSC Adv., 2016,6, 32653-32660
https://doi.org/10.1039/C6RA03023H
A highly efficient synthesis of dithiocarbamates in green reaction media
A deep eutectic solvent (DES) and polyethylene glycol (PEG) promoted the environmentally friendly and fast synthesis of dithiocarbamate derivatives via a one-pot, three-component condensation of an amine, carbon disulfide, and a variety of electrophilic reagents in high yields and short reaction times without organic solvents and tedious work-up.
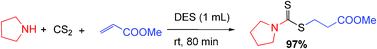
RSC Adv., 2012,2, 7413-7416
https://doi.org/10.1039/C2RA20615C
About this collection
We are very pleased to present our 10th Anniversary collection on Sustainable synthesis!Looking back over the last 10 years, we would like to showcase some of the very best articles that have been published in RSC Advances. Many of these papers have been cited hundreds of times, providing valuable advances for further research, and some continue to be among the journal’s most downloaded articles as of today.
We hope you enjoy our 10th Anniversary collection on Sustainable synthesis!