Themed collection Editors' Collection: Fluorine chemistry in medicinal chemistry and chemical biology

Recent advances in direct trifluoromethylation of olefinic C–H bonds
The aim of this review is to provide a comprehensive overview of the direct trifluoromethylation of olefinic C–H bonds with special attention on the mechanistic aspects of the reactions.

RSC Adv., 2019,9, 27625-27639
https://doi.org/10.1039/C9RA04170B
Recent advances in the synthesis of fluorinated hydrazones
Fluorinated compounds are widely used in pharmaceuticals, agrochemicals, and materials.
RSC Adv., 2018,8, 17110-17120
https://doi.org/10.1039/C8RA02533A
Translational incorporation of modified phenylalanines and tyrosines during cell-free protein synthesis
E. coli wild-type translational machinery utilizes a range of nonproteinogenic amino acids for protein synthesis with incorporation levels greater than 95%.

RSC Adv., 2020,10, 11013-11023
https://doi.org/10.1039/D0RA00655F
Rhenium(I) complexation–dissociation strategy for synthesising fluorine-18 labelled pyridine bidentate radiotracers
A novel fluorine-18 radiolabelling method employing rhenium(I) mediation is described herein. In less than 1 minute, fluorine-18 labelled complexes and ligands were synthesised in greater than 80% and 60% radiochemical yields (RCY), respectively.

RSC Adv., 2020,10, 8853-8865
https://doi.org/10.1039/D0RA00318B
2′-Fluoro-c-di-GMP as an oral vaccine adjuvant
2′-F-c-di-GMP was synthesized through the modified H-phosphonate chemistry. 2′-F-c-di-GMP was found to be an effective mucosal vaccine adjuvant, both intranasally and orally.
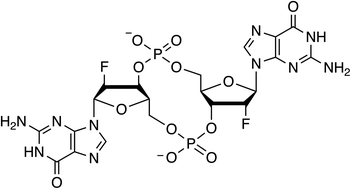
RSC Adv., 2019,9, 41481-41489
https://doi.org/10.1039/C9RA08310C
3(2H)-pyridazinone derivatives: a new scaffold for novel plant activators
Due to the emergence of drug resistance, pesticide residue and environmental contamination, it is important to develop novel eco-friendly strategies to protect plants.
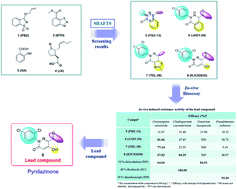
RSC Adv., 2019,9, 36204-36207
https://doi.org/10.1039/C9RA06892A
Combinatorial synthesis and biological evaluations of (E)-β-trifluoromethyl vinylsulfones as antitumor agents
(E)-β-Trifluoromethyl vinylsulfones is accomplished through a reaction of alkynes, Togni reagent, and sodium benzenesulfinates under metal-free conditions. p-Acetylphenyl in R1 has the best activities against several tumor cells.
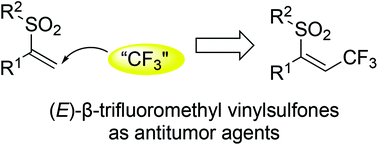
RSC Adv., 2019,9, 31474-31482
https://doi.org/10.1039/C9RA06368D
Synthesis, structural studies and biological properties of some phosphono-perfluorophenylalanine derivatives formed by SNAr reactions
Several novel phosphono-perfluorophenylalanine derivatives, as mimetics of phenylalanine, were synthesized by subjecting diethyl (2-(perfluorophenyl)-1-(phenylamino)ethyl)-phosphonate to SNAr reactions with thiols, amines and phenols.

RSC Adv., 2019,9, 24117-24133
https://doi.org/10.1039/C9RA03982A
Fluorination and chlorination effects on quinoxalineimides as an electron-deficient building block for n-channel organic semiconductors
Three fluorinated or chlorinated quinoxalineimide units (QI-1F, QI-2F, and QI-2Cl) have been designed and developed.

RSC Adv., 2019,9, 10807-10813
https://doi.org/10.1039/C9RA02413A
Fully automated peptide radiolabeling from [18F]fluoride
A fully automated approach for peptide radiolabeling from [18F]fluoride, provides an efficient process that is amenable to GMP production of [18F]F-peptide radiotracers.
![Graphical abstract: Fully automated peptide radiolabeling from [18F]fluoride](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C8RA10541C&imageInfo.ImageIdentifier.Year=2019)
RSC Adv., 2019,9, 8638-8649
https://doi.org/10.1039/C8RA10541C
19 F multiple-quantum coherence NMR spectroscopy for probing protein–ligand interactions
An NMR experiment is presented that allows identification of weak binders typically found in early stages of drug discovery programs.

RSC Adv., 2018,8, 40687-40692
https://doi.org/10.1039/C8RA09296F
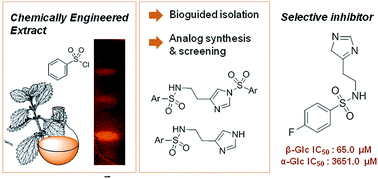
N α -arylsulfonyl histamines as selective β-glucosidase inhibitors
A selective β-glucosidase inhibitor was discovered using the chemically engineered extracts approach.
RSC Adv., 2018,8, 36209-36218
https://doi.org/10.1039/C8RA06625F
Chemoenzymatic synthesis of polypeptides in neat 1,1,1,2-tetrafluoroethane solvent
Chemoenzymatic polypeptide synthesis offers several advantages over chemical or other biological routes, however, the use of aqueous-based media suffers from reverse hydrolysis reactions that challenge peptide chain propagation.

RSC Adv., 2018,8, 35936-35945
https://doi.org/10.1039/C8RA06657D
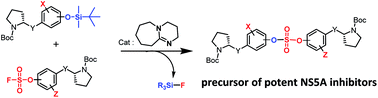
Sulfur(VI) fluoride exchange as a key reaction for synthesizing biaryl sulfate core derivatives as potent hepatitis C virus NS5A inhibitors and their structure–activity relationship studies
SuFEx chemistry was utilized for the design and synthesis of potent HCV NS5A inhibitors featuring a biaryl sulfate core.
RSC Adv., 2018,8, 31803-31821
https://doi.org/10.1039/C8RA05471A
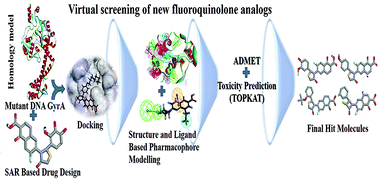
Design and virtual screening of novel fluoroquinolone analogs as effective mutant DNA GyrA inhibitors against urinary tract infection-causing fluoroquinolone resistant Escherichia coli
Design and development of novel fluoroquinolones analogs using target (mutant DNA GyrA), ligand-based SAR and virtual screening techniques.
RSC Adv., 2018,8, 23629-23647
https://doi.org/10.1039/C8RA01854E
Trifluoromethylated proline analogues as efficient tools to enhance the hydrophobicity and to promote passive diffusion transport of the L-prolyl-L-leucyl glycinamide (PLG) tripeptide
The incorporation of trifluoromethylated proline analogues in the tripeptide PLG enhances its hydrophobicity and promotes passive diffusion transport.

RSC Adv., 2018,8, 14597-14602
https://doi.org/10.1039/C8RA02511H
Click chemistry approaches to expand the repertoire of PEG-based fluorinated surfactants for droplet microfluidics
Click-synthesis of fluorinated surfactants for droplet microfluidics.
RSC Adv., 2018,8, 12960-12974
https://doi.org/10.1039/C8RA01254G
Design, synthesis, insecticidal activity and 3D-QSR study for novel trifluoromethyl pyridine derivatives containing an 1,3,4-oxadiazole moiety
Novel trifluoromethyl pyridine derivatives bearing 1,3,4-oxadiazole whereas synthesized, their which showed good insecticidal activity; a 3D-QSAR model with good predictability was is proposed.

RSC Adv., 2018,8, 6306-6314
https://doi.org/10.1039/C8RA00161H
About this collection
This collection, guest-edited by Professor Norio Shibata (Nagoya Institute of Technology), features articles published in the journal on fluorine chemistry related to medicinal chemistry and chemical biology.
Organofluorine compounds are revealed in the extensive use of key materials in diverse industrial areas of pharmaceuticals, agrochemicals, specialty materials, and polymers. In particular, the high demand for organofluorine compounds on the drug market has been evidenced by the sharp increase in the number of fluoro-pharmaceuticals approved each year. Fluorine-related papers published in the journal span over various research fields, including synthetic methodology, medicinal chemistry, chemical biology, and materials science.
We hope you will enjoy one of the states of the art of fluorine chemistry by this collection.