Themed collection Halogen Bonding in Crystal Engineering Editor’s collection

Crystallization-induced room-temperature phosphorescence in fumaramides
Novel fumaramides exhibit room-temperature phosphorescence in the solid state once molecular design and positioning of the carbonyl and bromine atoms allow for the formation of strong intermolecular halogen bonding interactions.
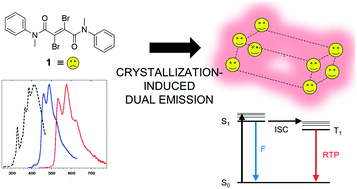
CrystEngComm, 2020,22, 7782-7785
https://doi.org/10.1039/D0CE01253J
Halogen-bonded cocrystals of donepezil with perfluorinated diiodobenzenes
Donepezil, an active pharmaceutical ingredient with several different acceptor sites for halogen bonding has successfully been cocrystallized with two perfluorinated halogen bond donors.
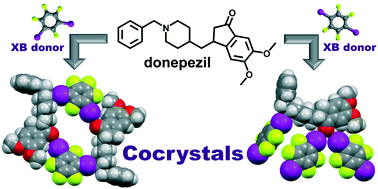
CrystEngComm, 2020,22, 5573-5577
https://doi.org/10.1039/D0CE01065K
A self-assembled nanotube supported by halogen bonding interactions
Upper-rim halogenation of a calix[4]arene modulates the packing of self-assembled nanotubes through the formation of complementary halogen bonding interactions.

CrystEngComm, 2019,21, 786-790
https://doi.org/10.1039/C8CE01824C
Halogen bonding at the wet interfaces of an amyloid peptide structure
Halogenation is a promising tool to stabilize – through halogen bonds – the wet interface of amyloid structures.
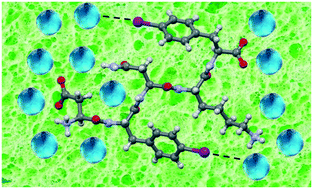
CrystEngComm, 2018,20, 5321-5326
https://doi.org/10.1039/C8CE01205A
Crystal engineering of coordination-polymer-based iodine adsorbents using a π-electron-rich polycarboxylate aryl ether ligand
This work revealed that the synergy of microporous channels and convergent arrangements of halogen bonding and charge-transfer interaction sites within coordination polymers facilitated the iodine adsorption process.
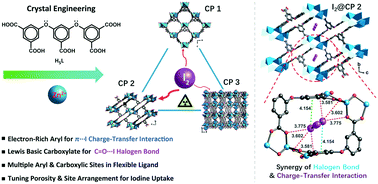
CrystEngComm, 2020,22, 6612-6619
https://doi.org/10.1039/D0CE01004A
The combination of halogen and hydrogen bonding: a versatile tool in coordination chemistry
4-Iodo-N-(4-pyridyl)benzamide (INPBA) and four derived coordination complexes were synthesized in order to explore the combination of halogen and hydrogen bonding interactions in coordination chemistry.
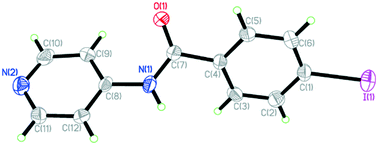
CrystEngComm, 2020,22, 6010-6018
https://doi.org/10.1039/D0CE00832J
Halogen bonded metal bis(dithiolene) 2D frameworks
Short and directional I⋯S halogen bonding interactions in iodinated bis(dithiolene) complexes lead to the crystallization of 2D or 3D anionic frameworks.
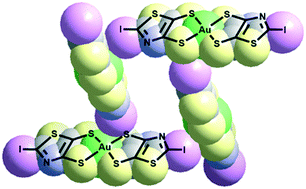
CrystEngComm, 2020,22, 3579-3587
https://doi.org/10.1039/D0CE00512F
Anion templated crystal engineering of halogen bonding tripodal tris(halopyridinium) compounds
Crystal engineering of halogen bonding tripodal receptors is found to be highly dependent on solvent and choice of anion.
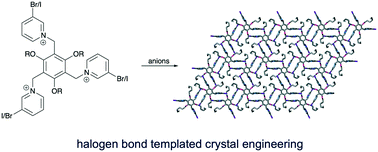
CrystEngComm, 2020,22, 2526-2536
https://doi.org/10.1039/D0CE00241K
Two-dimensional halogen-bonded organic frameworks based on the tetrabromobenzene-1,4-dicarboxylic acid building molecule
2D halogen-bonded organic frameworks were readily engineered by strong and directional effects of the primary Br⋯O and the secondary Br⋯π halogen bonding interactions from the tetrabromobenzene-1,4-dicarboxylic acid building molecule involving 100% supramolecular yields.
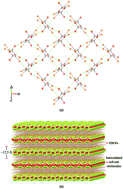
CrystEngComm, 2020,22, 24-34
https://doi.org/10.1039/C9CE01140D
Halogen bonding in 5-iodo-1-arylpyrazoles investigated in the solid state and predicted by solution 13C-NMR spectroscopy
Halogen bonding as important directional forces in the supramolecular structure of iodinated 1-arylpyrazoles.

CrystEngComm, 2019,21, 7085-7093
https://doi.org/10.1039/C9CE01263J
Regioselective [2 + 2] cycloaddition reaction within a pair of polymorphic co-crystals based upon halogen bonding interactions
A pair of photoreactive polymorphic co-crystals that undergo a regioselective solid-state [2 + 2] cycloaddition reaction that yields exclusively the head-to-tail photoproduct is reported.
![Graphical abstract: Regioselective [2 + 2] cycloaddition reaction within a pair of polymorphic co-crystals based upon halogen bonding interactions](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9CE01379B&imageInfo.ImageIdentifier.Year=2019)
CrystEngComm, 2019,21, 6671-6675
https://doi.org/10.1039/C9CE01379B
Probing non-covalent interactions driving molecular assembly in organo-electronic building blocks
One co-crystal structure characterized to identify and quantify various non-covalent interactions with spectroscopy, X-ray crystallography and density functional theory computations.
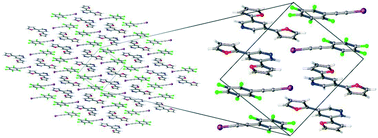
CrystEngComm, 2019,21, 3151-3157
https://doi.org/10.1039/C9CE00219G
Halogen and chalcogen-bonding interactions in sulphur-rich π-electron acceptors
Sulphur and iodine heteroatoms on the acceptor skeleton induce chalcogen⋯chalcogen and halogen-bonding interactions.
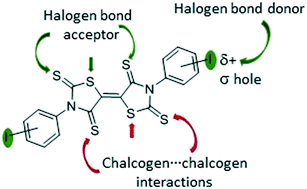
CrystEngComm, 2019,21, 1934-1939
https://doi.org/10.1039/C8CE02046A
Organic molecular tessellations and intertwined double helices assembled by halogen bonding
Crystalline polymorphs featuring halogen-bonded single-component supramolecular polygonal tessellations, a network of 41- and 43-double helices, and intertwined 31 and 32meso-helices.
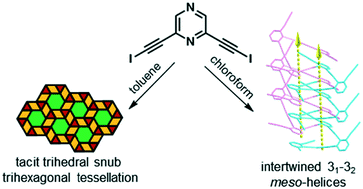
CrystEngComm, 2019,21, 1130-1136
https://doi.org/10.1039/C8CE02133C
Organometallic halogen bond acceptors: directionality, hybrid cocrystal precipitation, and blueshifted CO ligand vibrational band
The halogen bonding (XB) in metal carbonyls blueshifts the νCO band and XB directionality is dictated by XB-accepting atom hybridization.
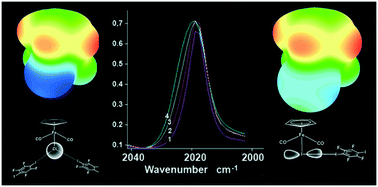
CrystEngComm, 2018,20, 2258-2266
https://doi.org/10.1039/C7CE02185B
About this collection
The halogen bond is the attractive interaction between an electrophilic region associated with a halogen atom in a molecular entity and a nucleophilic region in another, or the same, molecular entity. Since its rediscovery in the early 2000s, the field of halogen bonding has boomed, becoming one of the most used chemical interactions in crystal engineering. This Editor’s collection, guest edited by Pierangelo Metrangolo, Chair of the CrystEngComm Editorial Boardand one of the modern-day fathers of halogen bonding, highlights recent contributions to CrystEngComm that are broadly focused on halogen bonding in crystal engineering. The selection of articles showcases how the field has developed from the design of crystal structures to the development of functional materials. The wide range of halogen bond applications covers phosphorescent materials, pharmaceutical co-crystals and peptides, organic electronics, coordination frameworks, where the halogen bonds either drive the formation of the network, or stabilize it as a secondary interaction, or is used to bind hosted guests. Halogen bonding has become a mature field of research but it’s rediscovery has sparked interest towards other lesser known interactions, which are foreseen to become prominent in the future, such as chalcogen, pnictogen, and tetrel bonds.
This collection is dedicated to Professor Giuseppe Resnati on the occasion of his 65th birthday.