Two-dimensional halogen-bonded organic frameworks based on the tetrabromobenzene-1,4-dicarboxylic acid building molecule†
Abstract
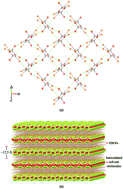
Two-dimensional (2D) halogen-bonded organic frameworks were readily engineered by strong and directional effects of the primary Br⋯O and the secondary Br⋯π halogen bonding interactions from the tetrabromobenzene-1,4-dicarboxylic acid (H2Br4BDC) building molecule involving 100% supramolecular yields. The 2D assembly can function as a host layered framework for the intercalation of various guest solvents including acetone (ATN), ethanol (EtOH), dimethyl sulfoxide (DMSO), and ethylene glycol (EG) resulting in a 1 : 2 host : guest complexation stoichiometry viz. H2Br4BDC·2S (S = ATN (1ATN), EtOH (2EtOH), DMSO (3DMSO), and EG (4EG)). All the solvates show remarkable similarities in their 2D layered sheets and the bilayer distance significantly responds to the size, shape, molecular conformation, and strength of the hydrogen bonding capability of the intercalated solvent molecules. The transition between solvate formation and desolvation was found to be facile and reversible upon the desolvation–resolvation process. The estimated Br⋯O halogen bonding energy of the solvates is in the −0.6 to −1.7 kcal mol−1 range, which was determined by quantum-mechanical calculations based on density functional theory (DFT) calculations. Furthermore, to quantitatively identify the host–guest intermolecular interactions of these solvates, they were visually compared by Hirshfeld surface analysis.
- This article is part of the themed collection: Halogen Bonding in Crystal Engineering Editor’s collection


 Please wait while we load your content...
Please wait while we load your content...