Halogen and chalcogen-bonding interactions in sulphur-rich π-electron acceptors†
Abstract
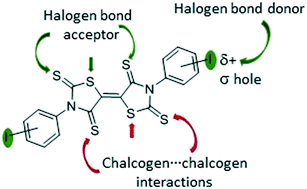
In order to explore the feasibility of generating halogen bonding interactions between sulphur-rich π-electron acceptors, we prepared three bithiazolidinylidene derivatives substituted by iodine atoms, namely 3,3′-bis(iodophenyl)bithiazolidinylidene-2,4,2′,4′-tetrathione (BIP-BTTT). Sulphur and iodine heteroatoms were introduced to the skeleton of the acceptor molecule to induce chalcogen⋯chalcogen and halogen bonding interactions. Both interactions can be evidenced by X-ray diffraction studies in the synthetic precursors as well as in the acceptors themselves.
- This article is part of the themed collection: Halogen Bonding in Crystal Engineering Editor’s collection


 Please wait while we load your content...
Please wait while we load your content...