Themed collection (Photo)electrocatalysis for renewable energy

Introduction to (photo)electrocatalysis for renewable energy
(Photo)electrocatalysis holds the promise to enable the broad implementation of renewable energies. The articles highlighted in this issue emphasize advances in types and activity of catalysts and electrode materials for a variety of reactions and technologies.

Chem. Commun., 2021,57, 1540-1542
https://doi.org/10.1039/D0CC90530E
Single-atom catalysts for the oxygen evolution reaction: recent developments and future perspectives
Issues to be improved in the future for single-atom OER catalysts and related challenges.
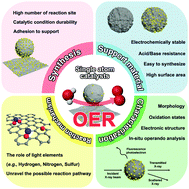
Chem. Commun., 2020,56, 12687-12697
https://doi.org/10.1039/D0CC04752J
Understanding active sites in molecular (photo)electrocatalysis through complementary vibrational spectroelectrochemistry
Highlighting vibrational spectroelectrochemistry for the investigation of synthetic molecular (photo) electrocatalysts for key energy conversion reactions.
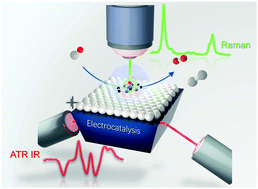
Chem. Commun., 2021,57, 2328-2342
https://doi.org/10.1039/D0CC07376H
Catalytic bias in oxidation–reduction catalysis
Under steady state conditions, the differential stability of reaction intermediates can alter the rate and the direction of a catalytic process regardless the overall underlying thermodynamic driving force.

Chem. Commun., 2021,57, 713-720
https://doi.org/10.1039/D0CC07062A
Electrocatalytic synthesis of organic carbonates
This article presents the latest research on electrosynthesis routes to organic carbonates. Different methods are discussed in an effort to understand the underlying processes and challenges.
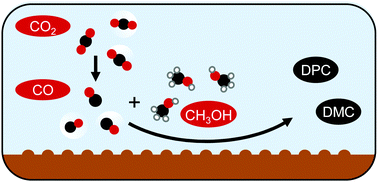
Chem. Commun., 2020,56, 13082-13092
https://doi.org/10.1039/D0CC04231E
Photoelectrocatalytic production of solar fuels with semiconductor oxides: materials, activity and modeling
Transition metal oxides keep on being excellent candidates as electrode materials for the photoelectrochemical conversion of solar energy into chemical energy.
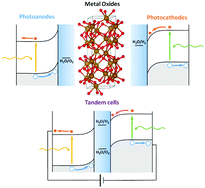
Chem. Commun., 2020,56, 12272-12289
https://doi.org/10.1039/D0CC04387G
Evolution of metal organic frameworks as electrocatalysts for water oxidation
The development of metal organic framework based water oxidation catalysts is discussed here in connection with various design strategies.

Chem. Commun., 2020,56, 11735-11748
https://doi.org/10.1039/D0CC03659E
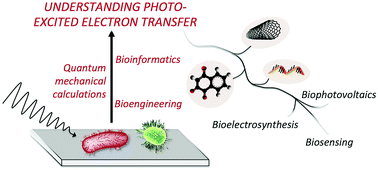
Advancing the fundamental understanding and practical applications of photo-bioelectrocatalysis
Combination of electrochemical methods, quantum mechanical calculations, bioinformatics, and bioengineering allows understanding the photoexcited electron transfer process and set the basis for artificially tuning photo-bioelectrocatalysis.
Chem. Commun., 2020,56, 8553-8568
https://doi.org/10.1039/D0CC02672G
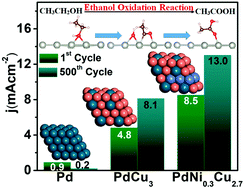
Tuning intermediate adsorption in structurally ordered substituted PdCu3 intermetallic nanoparticles for enhanced ethanol oxidation reaction
Structurally ordered Ni substituted PdCu3 nanoparticles were found to remarkably enhance the ethanol oxidation efficiency due to optimal adsorption of intermediates in close proximity.
Chem. Commun., 2021,57, 4508-4511
https://doi.org/10.1039/D0CC08075F
A nickel oxide-decorated in situ grown 3-D graphitic forest engrained carbon foam electrode for microbial fuel cells
Carbon foam was used as a substrate for NiO and growing carbon nanofibers. The synthesized NiO-CNF-CF electrode was successfully used as an efficient electrode for a microbial fuel cell.

Chem. Commun., 2021,57, 879-882
https://doi.org/10.1039/D0CC07303B
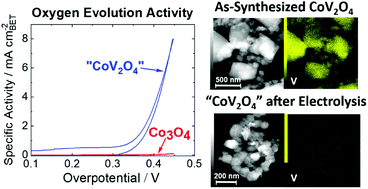
A CoV2O4 precatalyst for the oxygen evolution reaction: highlighting the importance of postmortem electrocatalyst characterization
Postmortem analysis of a CoV2O4 catalyst for oxygen evolution reveals the active species is likely a vanadium-free amorphous cobalt oxide.
Chem. Commun., 2021,57, 883-886
https://doi.org/10.1039/D0CC06513G
An efficient method to prepare supported bismuth nanoparticles as highly selective electrocatalyst for the conversion of CO2 into formate
We report a novel and upscalable method to synthesise small, highly dispersed Bi nanoparticles on activated carbon. This electrocatalyst displays outstanding selectivity (FE > 99% at −1.07 V vs. RHE) and stability in the conversion of CO2 to formate.

Chem. Commun., 2020,56, 14992-14995
https://doi.org/10.1039/D0CC06818G
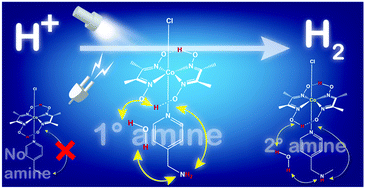
Probing the peripheral role of amines in photo- and electrocatalytic H2 production by molecular cobalt complexes
The photo- and electrocatalytic H2 production by the cobaloxime core improves in the presence of a primary amine group in the periphery.
Chem. Commun., 2020,56, 14841-14844
https://doi.org/10.1039/D0CC05786J
A dinuclear porphyrin-macrocycle as efficient catalyst for the hydrogen evolution reaction
A macrocyclic porphyrin complex comprising two nickel centres connected via redox mediating linkers gives rise to efficient HER catalysis.
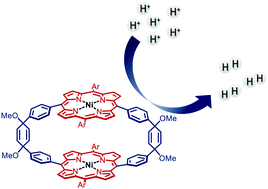
Chem. Commun., 2020,56, 14179-14182
https://doi.org/10.1039/D0CC05229A
Surface immobilized copper(I) diimine photosensitizers as molecular probes for elucidating the effects of confinement at interfaces for solar energy conversion
The ground- and excited-state properties of redox- and photo-active Cu(I) complexes immobilized in nanoporous AAO are responsive to pore diameter.

Chem. Commun., 2020,56, 12130-12133
https://doi.org/10.1039/D0CC05972B
Hydrogen evolution reaction mediated by an all-sulfur trinuclear nickel complex
A trinuclear nickel complex with S-based ligands is reported as a bio-inspired model of the [NiFe] hydrogenases' active site. DFT calculations indicate that thiolate and thioether functions are involved as proton relays in the H2 evolution mechanism.
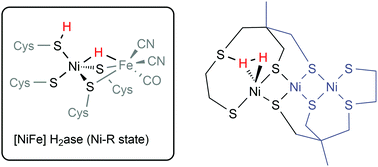
Chem. Commun., 2020,56, 11106-11109
https://doi.org/10.1039/D0CC04174B
Krypton-derivatization highlights O2-channeling in a four-electron reducing oxidase
Kr-derivatization and X-ray structures indicated O2-channel and gating-loop that prevent side-reaction in reduction of O2 to water in F420H2 oxidase.
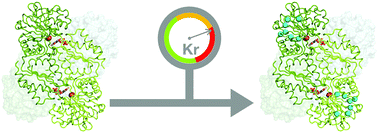
Chem. Commun., 2020,56, 10863-10866
https://doi.org/10.1039/D0CC04557H
A neural network-based approach to predicting absorption in nanostructured, disordered photoelectrodes
A neural network-based emulator can accurately and efficiently predict the spatial distribution of absorption throughout a nanoscale semiconductor in a disordered photoelectrode.
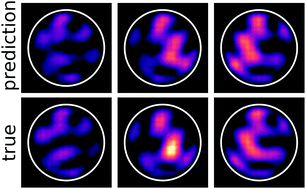
Chem. Commun., 2020,56, 10473-10476
https://doi.org/10.1039/D0CC04229C
Role of electrolyte composition on the acid stability of mixed-metal oxygen evolution catalysts
Acid stability in catalysts that promote the oxygen evolution reaction (OER) involves an interplay between electrolyte and catalyst composition, both of which must be judiciously selected in order to promote activity and durability.
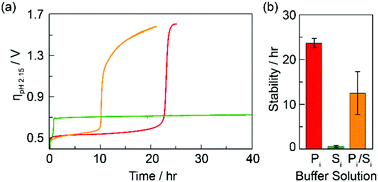
Chem. Commun., 2020,56, 10477-10480
https://doi.org/10.1039/D0CC03863F
Charge transfer in DHICA eumelanin-like oligomers: role of hydrogen bonds
The building blocks of eumelanin can be used as versatile material with enhanced charge transfer properties.
Chem. Commun., 2020,56, 10481-10484
https://doi.org/10.1039/D0CC04702C
Reactivation of sulfide-protected [FeFe] hydrogenase in a redox-active hydrogel
[FeFe] hydrogenases are highly active hydrogen conversion catalysts, whose oxygen sensitivity prevents their widespread application. Here, an oxygen-stable inactive form was reactivated in a redox hydrogel enabling its practical use under air.
![Graphical abstract: Reactivation of sulfide-protected [FeFe] hydrogenase in a redox-active hydrogel](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0CC03155K&imageInfo.ImageIdentifier.Year=2020)
Chem. Commun., 2020,56, 9958-9961
https://doi.org/10.1039/D0CC03155K
Reduction of carbon dioxide at a plasmonically active copper–silver cathode
Ethylene, CO, methane, formate, and allyl alcohol were selectively enhanced upon illumination of a copper–silver cathode during plasmon-enhanced electrochemical conversion.
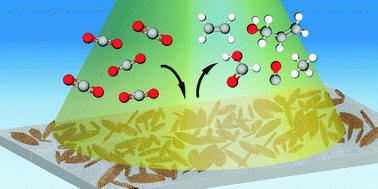
Chem. Commun., 2020,56, 9970-9973
https://doi.org/10.1039/D0CC03215H
Diazo coupling for surface attachment of small molecules to TiO2 nanoparticles
Interfacial diazo coupling of surface-bound amines with aromatics via a robust siloxane anchor installs various small molecules on TiO2 surface.
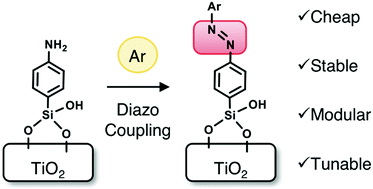
Chem. Commun., 2020,56, 9340-9343
https://doi.org/10.1039/D0CC03631E
Surprisingly big linker-dependence of activity and selectivity in CO2 reduction by an iridium(I) pincer complex
Here, we report the quantitative electroreduction of CO2 to CO by a PNP-pincer iridium(I) complex bearing amino linkers in DMF/water. The activity and selectivity of the reduction greatly depend on the choice of linker within the ligand.
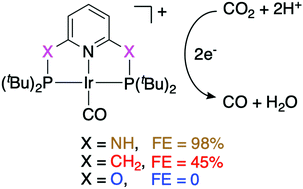
Chem. Commun., 2020,56, 9126-9129
https://doi.org/10.1039/D0CC03207G
Enhancing the activity of photocatalytic hydrogen evolution from CdSe quantum dots with a polyoxovanadate cluster
Adding metal-cluster hole scavengers enhances photocatalytic hydrogen production by quantum dots.
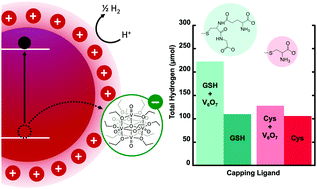
Chem. Commun., 2020,56, 8762-8765
https://doi.org/10.1039/D0CC03163A
Phosphorene-quantum-dot-interspersed few-layered MoS2 hybrids as efficient bifunctional electrocatalysts for hydrogen and oxygen evolution
0-D/2-D hybrids consisting of few-layered MoS2 nanosheets interspersed with phosphorene quantum dots show remarkable bifunctional electrocatalytic activity for oxygen and hydrogen evolution, with exceptional stability in alkaline conditions.
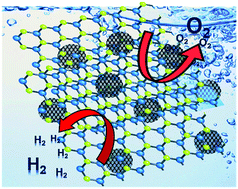
Chem. Commun., 2020,56, 8623-8626
https://doi.org/10.1039/D0CC03053H
Enhancement of the oxygen reduction reaction electrocatalytic activity of metallo-corroles using contracted cobalt(III) CF3-corrole incorporated in a high surface area carbon support
Molecular ORR catalysts based on metallo-corrole with the smallest meso-substituent reported to date, Co(III)CF3-corrole, was synthesized and compared to the well-studied Co(III)tpf-corrole when adsorbed on a high surface area carbon support.
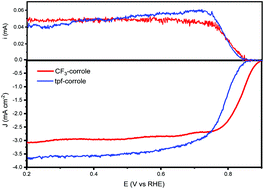
Chem. Commun., 2020,56, 8627-8630
https://doi.org/10.1039/D0CC03122D
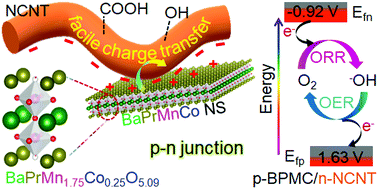
Charge transfer from perovskite oxide nanosheets to N-doped carbon nanotubes to promote enhanced performance of a zinc–air battery
Charge transfer from spatially connected double perovskite oxide nanosheets to N-doped carbon nanotubes facilitates oxygen activation in a stable zinc–air battery.
Chem. Commun., 2020,56, 8277-8280
https://doi.org/10.1039/D0CC03154B
Photoreforming of biomass in metal salt hydrate solutions
A combined process using LiBr metal salt hydrates and photoreforming converts cellulosic biomass into H2 and organic reaction products.
Chem. Commun., 2020,56, 5743-5746
https://doi.org/10.1039/D0CC01686A
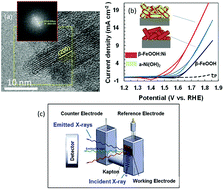
Operando X-ray absorption spectroscopy of hyperfine β-FeOOH nanorods modified with amorphous Ni(OH)2 under electrocatalytic water oxidation conditions
Operando X-ray absorption spectroscopy was employed to study an active electrocatalyst, hyperfine β-FeOOH nanorods (∅ 3 × 15 nm) surface-modified with amorphous Ni hydroxide.
Chem. Commun., 2020,56, 5158-5161
https://doi.org/10.1039/D0CC00692K