Themed collection In memory of Peter Day

Ion–radical salts: a new type of molecular ferrimagnet
Ion–radical salts composed of organo-sulfur donor cations and 3d-complex anions show long-range ferrimagnetic order.
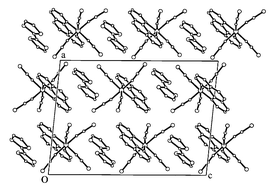
J. Mater. Chem., 2005,15, 23-25
https://doi.org/10.1039/B412696N
Chiral conducting salts of BEDT-TTF containing a single enantiomer of tris(oxalato)chromate(III) crystallised from a chiral solvent
Two polymorphs of the chiral conductor (BEDT-TTF)3NaCr(C2O4)3·CH3NO2 have been crystallised from racemic tris(oxalato)chromate(III) in a chiral electrolyte.
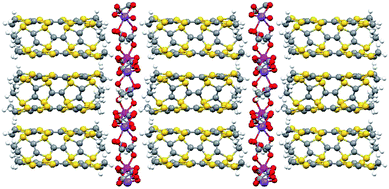
J. Mater. Chem., 2010,20, 2738-2742
https://doi.org/10.1039/B920224B
A molecular charge transfer salt of BEDT-TTF containing a single enantiomer of tris(oxalato)chromate(III) crystallised from a chiral solvent
The first chiral BEDT-TTF-tris(oxalato)metallate(III) salt has been crystallised from a solution containing the racemic anion in the chiral solvent (R)-(−)-carvone.
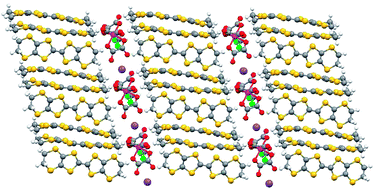
CrystEngComm, 2010,12, 1369-1372
https://doi.org/10.1039/B916136H
Novel enantiopure bis(pyrrolo)tetrathiafulvalene donors exhibiting chiral crystal packing arrangements
Two novel enantiopure bis(pyrrolo[3,4-d])tetrathiafulvalene derivatives, substrates for preparing chiral conducting materials, show chiral crystal packing arrangements in which successive layers are rotated in accordance with an exact or approximate 43 axis.
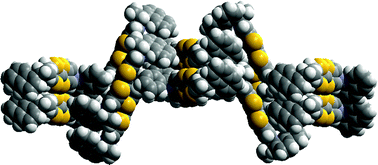
CrystEngComm, 2009,11, 993-996
https://doi.org/10.1039/B822009N
Metallic molecular crystals containing chiral or racemic guest molecules
Metallic charge-transfer salts of BEDT-TTF containing both racemic (R/S)- and chiral (S)-sec-phenethyl alcohol as guest molecules have been crystallised. Their structures and physical properties are compared.
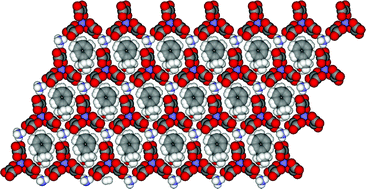
CrystEngComm, 2007,9, 865-867
https://doi.org/10.1039/B709558A
Superstructures of donor packing arrangements in a series of molecular charge transfer salts
A series of three semiconducting BEDT-TTF charge transfer salts with tris(oxalato)metallate anions have unit cells that uniquely contain layers of donors packed in both the α and β″ packing motifs.
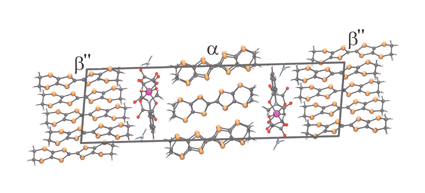
Chem. Commun., 2004, 18-19
https://doi.org/10.1039/B310683G
The first molecular charge transfer salt containing proton channels
Chem. Commun., 2001, 1462-1463
https://doi.org/10.1039/B103877J
Contrasting crystal packing arrangements in triiodide salts of radical cations of chiral bis(pyrrolo[3,4-d])tetrathiafulvalenes
Triiodides are arranged in head to tail pairs, infinite lines or a castellated arrangement in the radical cation salts of chiral bis(N(1-arylethyl))pyrrolo-tetrathiafulvalene salts.
![Graphical abstract: Contrasting crystal packing arrangements in triiodide salts of radical cations of chiral bis(pyrrolo[3,4-d])tetrathiafulvalenes](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C5CE01430A&imageInfo.ImageIdentifier.Year=2015)
CrystEngComm, 2015,17, 7354-7362
https://doi.org/10.1039/C5CE01430A
Synthesis of bis(ethylenedithio)tetrathiafulvalene (BEDT-TTF) derivatives functionalised with two, four or eight hydroxyl groups
A range of polyhydroxylated BEDT-TTF derivatives is described including a single diastereomer bearing four –CH2OH groups, and a single enantiomer bearing four CH(OH)CH2OH groups and containing eight stereogenic centres.
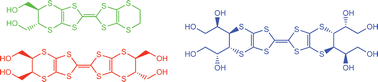
Org. Biomol. Chem., 2007,5, 3172-3182
https://doi.org/10.1039/B709823E
Multi-layered molecular charge-transfer salts containing alkali metal ions
Two new BEDT-TTF charge-transfer salts, novel examples of multi-layered stacking, demonstrate the potential of such structures for combining properties such as electronic and ionic conduction with chirality and magnetism.
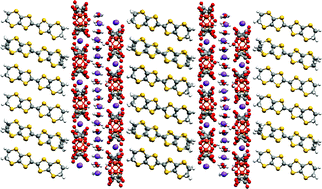
J. Mater. Chem., 2007,17, 3324-3329
https://doi.org/10.1039/B702442H
Synthetic strategies to chiral organosulfur donors related to bis(ethylenedithio)tetrathiafulvalene
Syntheses are reported for three enantiopure organosulfur donors which are substrates for preparing chiral electroactive materials.
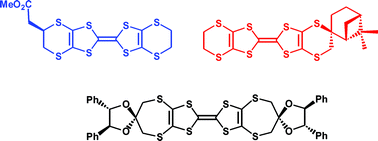
Org. Biomol. Chem., 2005,3, 2155-2166
https://doi.org/10.1039/B502437D
Synthesis, structure and magnetic properties of organic-intercalated bimetallic molecular-based ferrimagnets (n-CnH2n+1)PPh3MIIFeIII(C2O4)3, (MII = Mn, Fe; n = 3–7)
Increasing the alkyl chain length from C3 to C7 increases the interlayer spacing in (n-CnH2n+1)PPh3MIIFeIII(C2O4)3 ferrimagnets by 15% but Tc is unchanged.
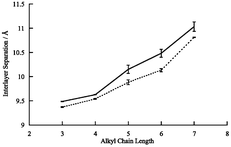
J. Chem. Soc., Dalton Trans., 2002, 1429-1434
https://doi.org/10.1039/B107057F
Preparation, X-ray crystal structures and properties of α-(BEDT-TTF)2[FeIII(phen)(NCS)4]·2CH2Cl2 and (BEDT-TTF)[CrIII(isoq)2(NCS)4] (phen = 1,10-phenanthroline ; isoq = isoquinoline )
New superconducting charge-transfer salts (BEDT-TTF)4[A·M(C2O4)3]·C6H5NO2
(A = H3O or NH4, M = Cr or Fe,
![Graphical abstract: New superconducting charge-transfer salts (BEDT-TTF)4[A·M(C2O4)3]·C6H5NO2 (A = H3O or NH4, M = Cr or Fe, BEDT-TTF = bis(ethylenedithio)tetrathiafulvalene)](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=B101134K&imageInfo.ImageIdentifier.Year=2001)
J. Mater. Chem., 2001,11, 2095-2101
https://doi.org/10.1039/B101134K
TTF based charge transfer salts of [Cr(NCS)4(phen)]−: bulk magnetic order and crystal structures of the TTF, TMTTF (tetramethyltetrathiafulvalene ) and TMTSF (tetramethyltetraselenafulvalene) derivatives
J. Chem. Soc., Dalton Trans., 2000, 2739-2744
https://doi.org/10.1039/B004140H
TTF based charge transfer salts of [M(NCS)4(C9H7N)2]− where M = Cr, Fe and C9H7N = isoquinoline ; observation of bulk ferrimagnetic order
J. Chem. Soc., Dalton Trans., 2000, 905-909
https://doi.org/10.1039/A910259K
Polymorphism based on molecular stereoisomerism in tris(oxalato) Cr(III) salts of bedt-ttf [bis(ethylenedithio)tetrathiafulvalene]
Synthesis, crystal structure and properties of the semiconducting molecular charge-transfer salt (bedt-ttf)2Ge(C2O4)3·PhCN [bedt-ttf=bis(ethylenedithio)tetrathiafulvalene]
New molecular superconductor containing paramagnetic chromium(iii) ions
Co-ordination complexes as organic–inorganic layer magnets
Quasi-one-dimensional bis(ethylenedithio)tetrathiafulvalene charge-
transfer salts with paramagnetic Group 6 anions
Molecular magnetic semiconductors, metals and superconductors:BEDT-TTF salts with magnetic anions
Structural properties of A2SnO4(A = Ba,Sr). A neutron diffraction study
Crystal structure and magnetic properties of the layer ferrimagnet N(n-C5H11)4MnIIFeIII(C2O4)3
Synthesis and magnetism of mixed valency [N(n-C4H9)4 or P(C6H5)4]CrIICrIII(C2O4)3
β″-(bedt-ttf)4[(H2O)Fe(C2O4)3]·PhCN: the first molecular superconductor containing paramagnetic metal ions
Molecular-based mixed valency ferrimagnets (XR4)FeIIFeIII(C2O4)3(X = N, P; R =,n-propyl, n-butyl, phenyl): anomalous negative magnetisation in the tetra-n-butylammonium derivative
Crystal structures and properties of two modifications of the molecular charge-transfer salt (BEDT-TTF)2Ag(CN)2[BEDT-TTF = bis(ethylenedithio)tetrathiafulvalene]
Feature article. Organic-intercalated halogenochromates(II): low-dimensional magnets
Crystal structures and physical properties of bis(ethylenedithio)-tetrathiafulvalene charge-transfer salts with FeX4–(X = Cl or Br) anions
A novel conducting charge-transfer salt: (BEDT-TTF)3Cl2·2H2O
Electronic spectrum of the manganate(V) ion in different host lattices
Charge transfer in mixed-valence solids. Part VIII. Contribution of valence delocalisation to the ferromagnetism of Prussian Blue
Bis(monoalkylammonium) tetrachlorochromates(II): a new series of two-dimensional ionic ferromagnets
Charge transfer in mixed valence solids. Part VII. Perturbation calculations of valence delocalization in iron(II,III) cyanides and silicates
Polarization, temperature dependence and absorption mechanism of the electronic transitions in some linear antiferromagnets
Charge transfer in mixed-valence solids. Part V. Semiconductivity of hexachloroantimonates(III,V)
The spectra of complexes of conjugated ligands. Part III. Charge-transfer spectra of bisnioximebis(substituted pyridine)iron(II) complexes
Charge transfer in mixed-valence solids. Part IV. Electronic spectra of hexachloroantimonates(III,V)
Some reactions of cobalt phthalocyanines
The vibrational spectra of some chloro-anions
Far-ultraviolet spectra of metal halide complexes
The spectra of complexes of conjugated ligands. Part I. Charge-transfer in phenanthroline complexes: energy shifts on substitution
The spectra of complexes of conjugated ligands. Part II. Charge-transfer in substituted phenanthroline complexes: intensities
740. Charge-transfer spectra of some inorganic complexes in solution
About this collection
Professor Peter Day, who sadly passed away on May 19, 2020 at the age of 81, was a true pioneer in the field of materials chemistry. During his career, he made a substantial contribution to the development of mixed-valence chemistry and molecular electronic materials. In the search for unusual magnetic and electron transport properties, his research centred on the synthesis and characterisation of molecular inorganic and metal-organic solids.
Peter Day has been very involved with the Journal of Materials Chemistry, leading the journal through the millennium as Scientific Advisory Editor. In this tribute to Peter Day, we have collated a number of his publications across the journals Chemical Communications, CrystEngComm, Dalton Transactions, Journal of the Chemical Society A: Inorganic, Physical, Theoretical, Journal of the Chemical Society, Faraday Transactions 2: Molecular and Chemical Physics, Journal of Materials Chemistry and Organic & Biomolecular Chemistry. The publications span Peter’s entire research career and we hope you enjoy revisiting some of his exceptional work, which even today continue to inspire new research.