Themed collection Editor's choice - Paolo Melchiorre

Visual kinetic analysis
Naked-eye comparison of reaction profiles is a powerful tool to extract mechanistic information – FAQ, video tutorial and templated examples included.
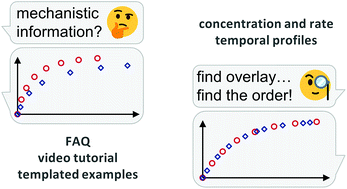
Chem. Sci., 2019,10, 348-353
https://doi.org/10.1039/C8SC04698K
Predictive and mechanistic multivariate linear regression models for reaction development
The utilization of physical organic molecular descriptors for the quantitative description of reaction outcomes in multivariate linear regression models is demonstrated as an effective tool for a priori prediction and mechanistic interrogation.
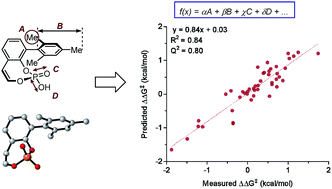
Chem. Sci., 2018,9, 2398-2412
https://doi.org/10.1039/C7SC04679K
A transition-metal-free & diazo-free styrene cyclopropanation
A visible-light-enabled, operationally simple cyclopropanation reaction of styrenes with excellent functional group tolerance and chemoselectivity via carbenoid-like radicals.

Chem. Sci., 2019,10, 9374-9379
https://doi.org/10.1039/C9SC02749A
Pd-catalyzed site-selective C(sp2)–H radical acylation of phenylalanine containing peptides with aldehydes
A novel Pd-catalyzed δ-C(sp2)–H functionalization reaction with readily available aldehydes towards the assembly of non-proteogenic acylated Phe-containing oligopeptides is presented.
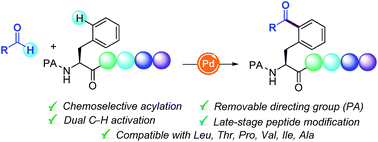
Chem. Sci., 2019,10, 8872-8879
https://doi.org/10.1039/C9SC03425K
Oxidant speciation and anionic ligand effects in the gold-catalyzed oxidative coupling of arenes and alkynes
The mechanism of the gold-catalyzed oxidative cross-coupling of arenes and alkynes has been studied in detail combining stoichiometric experiments with putative reaction intermediates and DFT calculations.

Chem. Sci., 2019,10, 8411-8420
https://doi.org/10.1039/C9SC02372K
Bio-inspired synthesis of xishacorenes A, B, and C, and a new congener from fuscol
In this manuscript, we describe the conversion of fuscol, a natural product isolated in 1978, to xishacorenes A, B, and C, isolated in 2017, as well as a previously unreported congener, which we have named xishacorene D.

Chem. Sci., 2019,10, 7788-7791
https://doi.org/10.1039/C9SC02572C
Oxidative organocatalysed enantioselective coupling of indoles with aldehydes that forms quaternary carbon stereocentres
The first organocatalysed, metal-free cross-nucleophile coupling of indoles with α-branched aldehydes forming acyclic stereoselective quaternary carbon centres is presented.
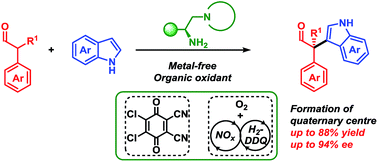
Chem. Sci., 2019,10, 3586-3591
https://doi.org/10.1039/C9SC00196D
Catalytic asymmetric allylation of aldehydes with alkenes through allylic C(sp3)–H functionalization mediated by organophotoredox and chiral chromium hybrid catalysis
We have developed a catalytic asymmetric nucleophilic allylation of aldehydes using simple alkenes as pronucleophiles without relying on stoichiometric metals.
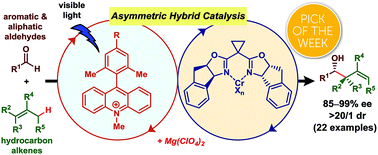
Chem. Sci., 2019,10, 3459-3465
https://doi.org/10.1039/C8SC05677C
Catalytic radical difluoromethoxylation of arenes and heteroarenes
The first visible light photocatalytic generation and utilization of the OCF2H radical for direct (hetero)aryl C–H difluoromethoxylation at room temperature.
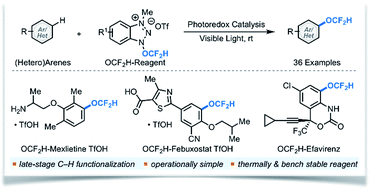
Chem. Sci., 2019,10, 3217-3222
https://doi.org/10.1039/C8SC05390A
Synthesis of 18F-difluoromethylarenes using aryl boronic acids, ethyl bromofluoroacetate and [18F]fluoride
Herein, we report the radiosynthesis of 18F-difluoromethylarenes via the assembly of three components, a boron reagent, ethyl bromofluoroacetate, and cyclotron-produced non-carrier added [18F]fluoride.
![Graphical abstract: Synthesis of 18F-difluoromethylarenes using aryl boronic acids, ethyl bromofluoroacetate and [18F]fluoride](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C8SC05096A&imageInfo.ImageIdentifier.Year=2019)
Chem. Sci., 2019,10, 3237-3241
https://doi.org/10.1039/C8SC05096A
A modular approach to prepare enantioenriched cyclobutanes: synthesis of (+)-rumphellaone A
A modular synthesis of enantioenriched polyfunctionalized cyclobutanes was developed and applied to the synthesis of (+)-rumphellaone A.
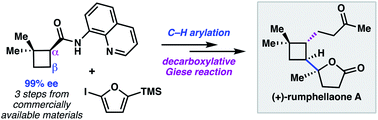
Chem. Sci., 2019,10, 2315-2319
https://doi.org/10.1039/C8SC05444D
Photoredox-mediated remote C(sp3)–H heteroarylation of free alcohols
We report an efficient and economical method for remote δ C(sp3)–H heteroarylation of free aliphatic alcohols using a hypervalent iodine PFBI-OH oxidant under photoredox catalysis.
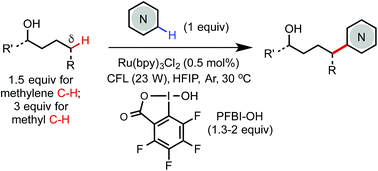
Chem. Sci., 2019,10, 688-693
https://doi.org/10.1039/C8SC04134B
A quantitative reactivity scale for electrophilic fluorinating reagents
Through kinetic studies we provide a quantitative reactivity scale for ten electrophilic fluorination reagents.
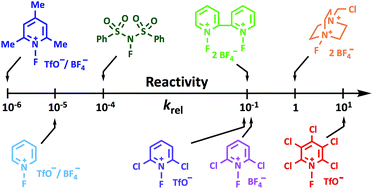
Chem. Sci., 2018,9, 8692-8702
https://doi.org/10.1039/C8SC03596B
Catalytic enantioselective radical coupling of activated ketones with N-aryl glycines
Asymmetric H-bonding catalysis as a viable strategy for enantioselective radical coupling of ketones is demonstrated.

Chem. Sci., 2018,9, 8094-8098
https://doi.org/10.1039/C8SC02948B
Diastereoselective C–H carbonylative annulation of aliphatic amines: a rapid route to functionalized γ-lactams
A palladium(II)-catalysed C(sp3)–H carbonylation of free(NH) secondary aliphatic amines to 2-pyrrolidinones is described.

Chem. Sci., 2018,9, 7628-7633
https://doi.org/10.1039/C8SC02855A
N-Heterocyclic carbenes as chiral Brønsted base catalysts: a highly diastereo- and enantioselective 1,6-addition reaction
Highly diastereo- and enantioselective 1,6-addition of β-keto amides to p-quinone methides catalyzed by N-heterocyclic carbenes via ion-pair interaction is developed.
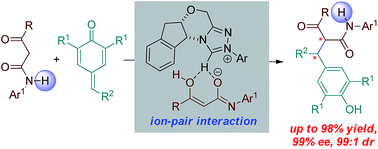
Chem. Sci., 2018,9, 6446-6450
https://doi.org/10.1039/C8SC02138D
Fine-tuned organic photoredox catalysts for fragmentation-alkynylation cascades of cyclic oxime ethers
Fine-tuned organic photoredox catalysts are introduced for the metal-free alkynylation of alkylnitrile radicals generated via oxidative ring opening of cyclic alkylketone oxime ethers.
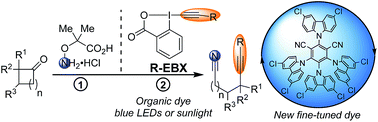
Chem. Sci., 2018,9, 5883-5889
https://doi.org/10.1039/C8SC01818A
Radical difluoromethylthiolation of aromatics enabled by visible light
A metal-catalyst-free and redox-neutral innate difluoromethylthiolation method enabled by visible light is described. This protocol employs a shelf-stable and readily available electrophilic difluoromethylthiolating reagent, PhSO2SCF2H, and converts a broad spectrum of arenes and heteroarenes to difluoromethylthioethers without noble metals and stoichiometric amount of additives.
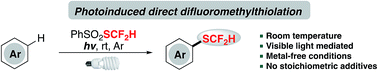
Chem. Sci., 2018,9, 5781-5786
https://doi.org/10.1039/C8SC01669K
Redox and photocatalytic properties of a NiII complex with a macrocyclic biquinazoline (Mabiq) ligand
Upon irradiation with visible light, the NiII complex 1 is converted to the reduced form 2 in the presence of NEt3. The Ni-based photosensitizer catalyzes the cyclization of a bromoalkyl-substituted indole.

Chem. Sci., 2018,9, 3313-3317
https://doi.org/10.1039/C7SC05320G
Super electron donors derived from diboron
The combination of diboron, pyridine and base serves as an efficient source of structurally well-defined super electron donors.

Chem. Sci., 2018,9, 2711-2722
https://doi.org/10.1039/C8SC00008E
About this collection
In 2019, Chemical Science welcomed Professor Paolo Melchiorre to the journal as an Associate Editor, handling papers in the areas of organic synthesis and catalysis, mainly utilising photochemical and radical reactivity patterns.
Paolo has looked back over recently published Chemical Science papers and has selected some outstanding articles on the theme of catalysis and synthetic methodology. We hope you enjoy reading through this selection.