Themed collection Sustainable synthesis and catalysis – Chemical Science symposium collection

Reductive aminations by imine reductases: from milligrams to tons
IRED-catalyzed reductive aminations have progressed from mg to ton scale, through advances in enzyme discovery, protein engineering and process biocatalysis.

Chem. Sci., 2022,13, 4697-4713
https://doi.org/10.1039/D2SC00124A
Synthetic half-reactions
This perspective on reactivity introduces Synthetic Half-Reactions (SHRs) as a way to analyze chemical transformations.
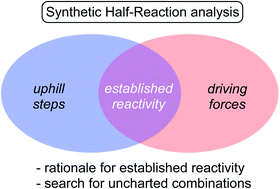
Chem. Sci., 2020,11, 12423-12427
https://doi.org/10.1039/D0SC03876H
Homogeneous catalytic C(sp3)–H functionalization of gaseous alkanes
The conversion of light alkanes into bulk chemicals is becoming an important challenge as it effectively avoids the use of prefunctionalized alkylating reagents.
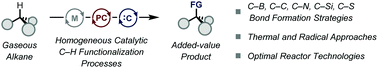
Chem. Commun., 2021,57, 9956-9967
https://doi.org/10.1039/D1CC04073A
Hexafluoroisopropanol: the magical solvent for Pd-catalyzed C–H activation
Among numerous solvents available for chemical transformations, 1,1,1,3,3,3-hexafluoro-2-propanol (popularly known as HFIP) has attracted enough attention of the scientific community in recent years.
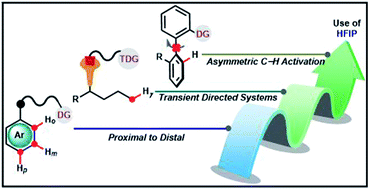
Chem. Sci., 2021,12, 3857-3870
https://doi.org/10.1039/D0SC06937J
Scalable biocatalytic C–H oxyfunctionalization reactions
Biocatalytic methods for selective C–H oxyfunctionalization reactions are rapidly emerging and hold significant potential to streamline complex molecule synthesis. This review highlights key advances in this area developed within the past decade.

Chem. Soc. Rev., 2020,49, 8137-8155
https://doi.org/10.1039/D0CS00440E
Reactive intermediates for interactome mapping
This tutorial review describes enabling methods for determining biomolecular interactions in live cells through the use of in situ generated reactive intermediates.
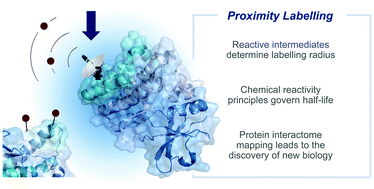
Chem. Soc. Rev., 2021,50, 2911-2926
https://doi.org/10.1039/D0CS01366H
Photochemical organocatalytic enantioselective radical γ-functionalization of α-branched enals
Reported herein is a rare example of asymmetric catalytic functionalisation of enals at the remote γ-position, proceeding via a radical path.
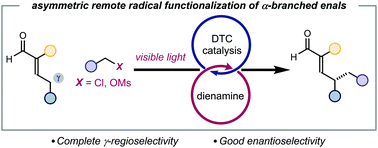
Chem. Commun., 2022,58, 6072-6075
https://doi.org/10.1039/D2CC01638A
Towards depeptidized aminoboronic acid derivatives through the use of borylated iminium ions
Herein, we use α-boryl iminium intermediates to access progressively depeptidized branched β-aminoboronic acids. We show the interaction of these compounds with carbohydrates and demonstrate their potential as synthetic building blocks.

Chem. Commun., 2022,58, 5033-5036
https://doi.org/10.1039/D2CC00659F
Direct arylation of N-heterocycles enabled by photoredox catalysis
A photoredox catalytic system was developed to construct N-heterobiaryls via direct arylation from readily accessible substrates. While phenols act as both coupling partner and proton donor, regular arenes were also applicable with HFIP as a solvent.
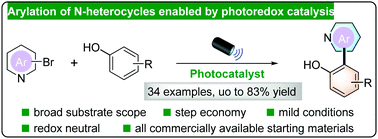
Chem. Commun., 2022,58, 4392-4395
https://doi.org/10.1039/D2CC01212J
Secondary-sphere modification in proline catalysis: old friend, new connection
Proline catalysis sparked not only the golden age of organocatalysis, but also the design of elaborate proline derivatives; instead, we propose to modify organocatalysts in situ under reaction conditions.
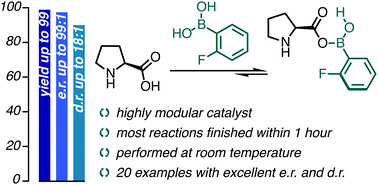
Chem. Commun., 2022,58, 1950-1953
https://doi.org/10.1039/D1CC05589E
Formic acid disproportionation into formaldehyde triggered by vanadium complexes with iridium catalysis under mild conditions in N-methylation
Renewable HCO2H disproportionation into CH2O triggered by vanadium complexes with iridium catalysis under mild conditions in N-methylation is discovered. The gram-scale application of in situ generated CH2O by HCO2H disproportionation is demonstrated.
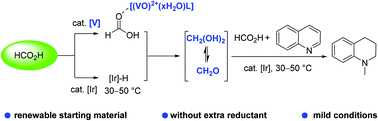
Green Chem., 2021,23, 2918-2924
https://doi.org/10.1039/D1GC00275A
A facile and regioselective multicomponent synthesis of chiral aryl-1,2-mercaptoamines in water followed by monoamine oxidase (MAO-N) enzymatic resolution
A microwave assisted multicomponent protocol on water allows the regioselective synthesis of chiral aryl-1,2-mercaptoamines. The enzymatic resolution of the racemates leads to enantioenriched products.
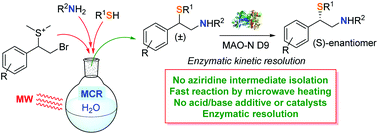
Org. Biomol. Chem., 2019,17, 8982-8986
https://doi.org/10.1039/C9OB01962F
Photoinduced decarboxylative azidation of cyclic amino acids
The direct decarboxylative azidation of cyclic α-amino acids has been achieved via visible light-mediated organo-photoredox catalysis.

Org. Biomol. Chem., 2019,17, 1839-1842
https://doi.org/10.1039/C8OB02702A
Ligand-redox assisted nickel catalysis toward stereoselective synthesis of (n+1)-membered cycloalkanes from 1,n-diols with methyl ketones
A homogeneous nickel catalyst is described that forms (n+1)-membered cycloalkane rings from ketones and 1,n-diols following a radical-promoted pathway.
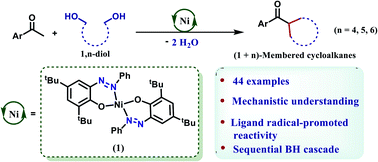
Chem. Sci., 2021,12, 14217-14223
https://doi.org/10.1039/D1SC04261K
Synthesis and enantioseparation of chiral Au13 nanoclusters protected by bis-N-heterocyclic carbene ligands
A series of chiral Au13 nanoclusters were synthesized via the direct reduction of achiral dinuclear Au(I) halide complexes ligated by ortho-xylyl-linked bis-N-heterocyclic carbene (NHC) ligands.
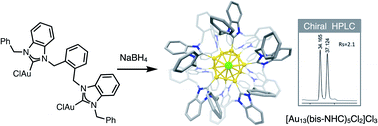
Chem. Sci., 2021,12, 10436-10440
https://doi.org/10.1039/D1SC03076K
Benzylic C–H isocyanation/amine coupling sequence enabling high-throughput synthesis of pharmaceutically relevant ureas
A copper-based catalyst system composed of commercially available reagents enables C–H isocyanation with exquisite (hetero)benzylic site selectivity, enabling high-throughput access to pharmaceutically relevant ureas via coupling with amines.
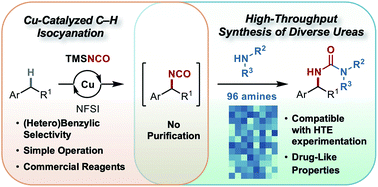
Chem. Sci., 2021,12, 10380-10387
https://doi.org/10.1039/D1SC02049H
A case of chain propagation: α-aminoalkyl radicals as initiators for aryl radical chemistry
Aminoalkyl radicals can be used as both initiators and chain-carriers for the conversion of aryl halides into the corresponding radicals. This approach by-passes the requirement for strongly reducing photocatalysts.
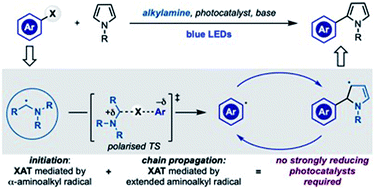
Chem. Sci., 2020,11, 12822-12828
https://doi.org/10.1039/D0SC04387G
Direct regioisomer analysis of crude reaction mixtures via molecular rotational resonance (MRR) spectroscopy
Direct analyses of crude reaction mixtures have been carried out using molecular rotational resonance (MRR) spectroscopy, allowing identification and quantification of major and minor components without sample purification or reference standards.
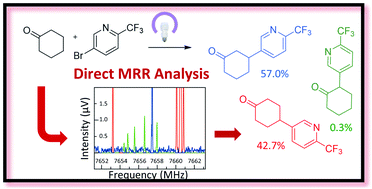
Chem. Sci., 2020,11, 6332-6338
https://doi.org/10.1039/D0SC01853H
Photochemical generation of acyl and carbamoyl radicals using a nucleophilic organic catalyst: applications and mechanism thereof
An organic catalyst uses low-energy photons to generate acyl and carbamoyl radicals upon activation of the corresponding chlorides via a nucleophilic acyl substitution path. The synthetic potential and the mechanism of this strategy are discussed.
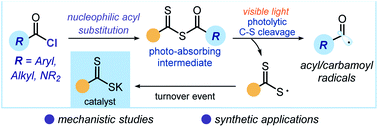
Chem. Sci., 2020,11, 6312-6324
https://doi.org/10.1039/D0SC02313B
Electrochemical C–H oxygenation and alcohol dehydrogenation involving Fe-oxo species using water as the oxygen source
The well-known [(TAML)Fe(OH2)]− complex undergoes proton-coupled oxidation to an Fe-oxo species that supports electrochemical C–H oxidation and alcohol dehydrogenation.
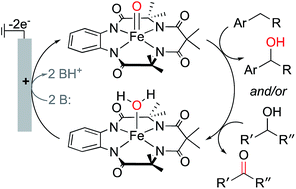
Chem. Sci., 2019,10, 7542-7548
https://doi.org/10.1039/C9SC02609F
Mechanistic investigation into the C(sp3)–H acetoxylation of morpholinones
The study of a selective palladium(II)-catalyzed C(sp3)–H acetoxylation reaction on a class of cyclic alkyl amines is reported.
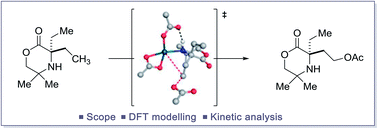
Chem. Sci., 2019,10, 83-89
https://doi.org/10.1039/C8SC03434F
About this collection
Sustainable synthesis and catalysis is the theme of this years’ Chemical Science symposium. We are excited to welcome the community back to our home in London, and virtually, for the fourth symposium in this series. To celebrate, we have selected articles recently published by the speakers and committee to showcase their work in RSC journals.
We hope you enjoy reading these articles and look forward to welcoming you to London either in-person or virtually in November!