Themed collection 2024 Organic Chemistry Frontiers HOT articles

From conventional to dual Co/photoredox mediated reductive coupling of alkynes and alkenes
The conventional and more recent metallaphotoredox-mediated reductive Co-catalysis for the coupling of olefins and alkynes is highlighted demonstrating the great potential of these approaches for the creation of advanced and complex synthons.

Org. Chem. Front., 2025,12, 1326-1339
https://doi.org/10.1039/D4QO02143F
Catalytic asymmetric synthesis of chiral sulfilimines via S–C bond formation
In this article, recent advances in catalytic asymmetric synthesis of chiral sulfilimines from sulfenamides via S–C bond formation are highlighted.

Org. Chem. Front., 2025,12, 953-959
https://doi.org/10.1039/D4QO01963F
Integrating electrocatalysis with biocatalysis for asymmetric synthesis
This highlight presents recent advances in asymmetric synthesis that integrate electrocatalysis with biocatalysis and discusses future development directions in this field.

Org. Chem. Front., 2024,11, 7243-7248
https://doi.org/10.1039/D4QO01899K
Direct C(sp2)–H activation of allenes via hydrogen-atom transfer (HAT) in organic synthesis
The highlighted reports demonstrate the unique application of the direct HAT strategy to unlock new reactivity of allenes. With this novel method, a variety of structurally diverse allenes are efficiently workable.

Org. Chem. Front., 2024,11, 2108-2111
https://doi.org/10.1039/D3QO02132G
Cycloparaphenylene-based [2]catenanes: interlocking the carbon nanohoops
In this highlight, recent advances in the design and synthesis of novel cycloparaphenylene (CPP)-based [2]catenanes are presented, with an emphasis on the design strategy and further investigations on their attractive properties.
![Graphical abstract: Cycloparaphenylene-based [2]catenanes: interlocking the carbon nanohoops](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO00033A&imageInfo.ImageIdentifier.Year=2024)
Org. Chem. Front., 2024,11, 1490-1494
https://doi.org/10.1039/D4QO00033A
Recent advances in catalytic asymmetric condensation reactions for the synthesis of nitrogen containing compounds
Asymmetric synthesis of functionalized nitrogen-containing compounds via the direct condensation of amines with carbonyls, focusing on the desymmetrization or dynamic kinetic resolution, the de novo cycloaromatization and the Heyns rearrangement.

Org. Chem. Front., 2025,12, 960-974
https://doi.org/10.1039/D4QO02078B
Recent advances in the photocatalytic cleavage of C–C and C–N bonds associated with amines and their derivatives
The recent advances of the visible-light-mediated photocatalytic cleavage of C–C and C–N bonds associated with amines and their derivatives, which featuring mild conditions, unique selectivity, broad substrate scope and green manner, are reviewed.

Org. Chem. Front., 2025,12, 975-1000
https://doi.org/10.1039/D4QO02038C
Transition-metal-free approaches to access N-heterocycles and valuable intermediates from aryldiazonium salts
Development of novel and efficient synthetic methods to achieve valuable N-heterocycles from aryldiazonium salts has been an important research area in organic chemistry for several decades.

Org. Chem. Front., 2025,12, 256-327
https://doi.org/10.1039/D4QO01776E
Convergent synthetic approaches to polycyclic aromatic dicarboximides
Polycyclic aromatic dicarboximides (PADIs) as represented by naphthalene and perylene bis(dicarboximide) are useful molecules for a variety of functional materials and remain important synthetic targets for organic chemists.

Org. Chem. Front., 2025,12, 328-345
https://doi.org/10.1039/D4QO01828A
The reactivity of alkenyl boron reagents in catalytic reactions: recent advances and perspectives
Recent advances focusing on novel reactivity of alkenyl boron reagents in polar or radical pathways within catalytic reactions by employing transition metal catalysis, organocatalysis have been summarized and discussed.

Org. Chem. Front., 2024,11, 7249-7277
https://doi.org/10.1039/D4QO01703J
Hydrogen autotransfer with alcohols for alkylations
Iron/ruthenium complexes, akin to Knölker catalysts, for bond reduction and sustainable alkylation using alcohols are included in this review, along with advances in dehydrogenative coupling, hydrogen borrowing, and light-assisted catalysis.

Org. Chem. Front., 2024,11, 7278-7317
https://doi.org/10.1039/D4QO01626B
Manganese(III) acetate in organic synthesis: a review of the past decade
In this review, we summarize the latest developments and applications of Mn(OAc)3 in organic synthesis over the past decade, focusing on efforts to achieve milder reaction conditions while expanding the scope of synthesis possibilities.

Org. Chem. Front., 2024,11, 6850-6917
https://doi.org/10.1039/D4QO01499E
State-of-the-art strategies for Lewis acid-catalyzed strain-release cycloadditions of bicyclo[1.1.0]butanes (BCBs)
This review primarily focuses on the latest developments in Lewis acid-catalyzed strain-release cycloaddition reactions of BCBs.
![Graphical abstract: State-of-the-art strategies for Lewis acid-catalyzed strain-release cycloadditions of bicyclo[1.1.0]butanes (BCBs)](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO01741B&imageInfo.ImageIdentifier.Year=2024)
Org. Chem. Front., 2024,11, 6942-6957
https://doi.org/10.1039/D4QO01741B
Synthesis of C-glycosides enabled by palladium-catalyzed glycosylation reactions
This review highlights the recent progress in palladium-catalyzed C-glycosylation reactions for the synthesis of C-glycosides.

Org. Chem. Front., 2024,11, 6918-6941
https://doi.org/10.1039/D4QO01619J
Recent advances in the catalytic asymmetric construction of axially chiral azole-based frameworks
This review highlights recent advances in the atroposelective preparation of axially chiral azole derivatives, emphasizing mechanistic insights and synthetic applications in the development of this class of five-membered heterocyclic frameworks.

Org. Chem. Front., 2024,11, 6819-6849
https://doi.org/10.1039/D4QO01705F
Recent advances in asymmetric synthesis of chiral benzoheterocycles via Earth-abundant metal catalysis
This review mainly focus on the assembly of chiral benzoheterocycles, such as dihydrobenzofurans, chromans, indolines, tetrahydroquinolines, etc., via Earth-abundant metal-catalyzed (Cu, Fe, Ni, and Ti) asymmetric synthesis in the last few decades.

Org. Chem. Front., 2024,11, 6534-6557
https://doi.org/10.1039/D4QO01686F
Recent developments in the ring-opening transformations of gem-difluorocyclopropanes
This review summarizes the advances of gem-F2CPs as fluoroallylic synthons in transition metal-catalysed ring-opening/cross-coupling reactions via single, double or no C-F bond cleavage on the last two years.

Org. Chem. Front., 2024,11, 6518-6533
https://doi.org/10.1039/D4QO01583E
Synthesis and applications of fluorinated, polyfluoroalkyl- and polyfluoroaryl-substituted 1,2,3-triazoles
This review summarises methods to synthesise and apply fluorine- and polyfluoroalkyl/aryl-substituted 1,2,3-triazoles. Focus is devoted to recent examples and those where the properties of both the 1,2,3-triazole and fluorine groups are important.

Org. Chem. Front., 2024,11, 5938-5984
https://doi.org/10.1039/D4QO01334D
Post-functionalization of end-capped twistacenes with pyrene units
This review covers recent advances in post-functionalization of twistacenes to realize polyacenes, chiral nanographenes and their semiconductor properties, and emphasizing a promising future for constructing chiral nanotubes.

Org. Chem. Front., 2024,11, 5638-5669
https://doi.org/10.1039/D4QO01199F
Recent advances in late-stage monofluorination of natural products and their derivatives
In this review, we have discussed the monofluorination of natural products or their derivatives occurring via direct C–H functionalization, late-stage functionalization and late-stage functional group transformation from 2011–2023.

Org. Chem. Front., 2024,11, 5605-5637
https://doi.org/10.1039/D4QO01111B
Modern photo- and electrochemical approaches to aryl radical generation
This review describes recent advances in the generation of aryl radicals using light and electricity. Such modern techniques allow for efficient energy and resource utilization, thus providing more sustainable radical arylation methods.

Org. Chem. Front., 2024,11, 5232-5277
https://doi.org/10.1039/D4QO00999A
Vinyl radicals in transition metal-catalyzed organic transformations
We summarize the advancements in transition metal-catalyzed organic transformations involving vinyl radicals and provide a categorical overview based on the reaction types.

Org. Chem. Front., 2024,11, 5202-5231
https://doi.org/10.1039/D4QO00995A
Site-selective editing of carbohydrate scaffolds to access rare sugars enabled by photoinduced radical processes
Herein, we summarize recent achievements via photoinduced radical-mediated strategies that enable the precise modification of carbohydrates.

Org. Chem. Front., 2024,11, 4926-4933
https://doi.org/10.1039/D4QO00963K
Recent advancements in Ni/photoredox dual catalysis for Csp3–Csp3 cross-coupling reactions
This minireview highlights recent advancements in the past five years (since 2019) in Ni/photoredox dual catalysis for Csp3–Csp3 cross-coupling reactions.

Org. Chem. Front., 2024,11, 4913-4925
https://doi.org/10.1039/D4QO00887A
Recent advances in post Ugi-4CR dearomatizations for constructing spiro heterocycles
This review summarizes recent advances in the combination of Ugi-4CR and dearomatization for the synthesis of spirocycles.

Org. Chem. Front., 2024,11, 4895-4912
https://doi.org/10.1039/D4QO01060D
Photocatalytic C–P bond formation based on the reaction of carbon-centered radicals with phosphides
This review focuses on the synthesis of C–P bonds using carbon-centered radicals with phosphorous compounds of different valence states (PIII, PV, P4) under photocatalysis.

Org. Chem. Front., 2024,11, 4882-4894
https://doi.org/10.1039/D4QO00933A
Catalytic transformations of alkenes via nickelacycles
This review summarizes recent developments in nickel-catalyzed transformations of alkenes via nickelacycles, highlighting the advantages and challenges of this innovative strategy.

Org. Chem. Front., 2024,11, 4602-4623
https://doi.org/10.1039/D4QO00737A
Unveiling the beauty of cyclopropane formation: a comprehensive survey of enantioselective Michael initiated ring closure (MIRC) reactions
This review presents a comprehensive overview of the synthesis of cyclopropanes using enantioselective Michael Initiated Ring Closure (MIRC) reactions.

Org. Chem. Front., 2024,11, 4560-4601
https://doi.org/10.1039/D4QO00535J
Electroreduction strategy: a sustainable tool for the generation of aryl radicals
This review primarily focuses on the generation of aryl radicals via an electroreduction strategy, and systematically elaborates on synthetic applications, scope, and limitations of the substrates.

Org. Chem. Front., 2024,11, 4318-4342
https://doi.org/10.1039/D4QO00846D
Silanes as a versatile hydride source for Ni–H catalysis: a promising tool for π-hydro functionalization
Nickel-catalyzed hydrofunctionalization of π-substrates is a possibly effective method to synthesize several value-added molecular architectures. This review covers the NiH catalyzed hydrofunctionalization reactions of alkenes, alkynes and allenes.

Org. Chem. Front., 2024,11, 4290-4317
https://doi.org/10.1039/D4QO00860J
Precision single-atom editing: new frontiers in nitrogen insertion and substitution for the generation of N-heterocycles
This paper primarily focuses on the editing of nitrogen atoms, encompassing the insertion of N, substitution of C with N, and utilization of 15N in place of 14N for the construction of N-heterocycles.

Org. Chem. Front., 2024,11, 4041-4053
https://doi.org/10.1039/D4QO00812J
Unleashing the potentiality of metals: synergistic catalysis with light and electricity
This review provides a comprehensive overview of metal catalysis in photo-electrochemical systems, discussing reaction mechanisms and offering prospects for this triadic catalytic mode.

Org. Chem. Front., 2024,11, 4024-4040
https://doi.org/10.1039/D4QO00797B
Catalytic alkane C–H functionalization by carbene insertion into unactivated C(sp3)–H bonds
This review summarizes the developments in the C–H functionalization of saturated hydrocarbons (CnH2n+2 and CnH2n) through catalytic carbene insertion into their C–H bonds dated up to February 2024, based on the categories of the types of carbenes.

Org. Chem. Front., 2024,11, 3777-3799
https://doi.org/10.1039/D4QO00606B
Advancements in the synthesis of polyoxygenated oxepanes and thiepanes for applications to natural products
Oxepanes and thiepanes are central motifs and tenants of many biologically important molecules. This comprehensive review covers synthetic methods for their preparation and use in the total synthesis of polyoxygenated oxepane natural products.

Org. Chem. Front., 2024,11, 3729-3776
https://doi.org/10.1039/D4QO00380B
Site-selective C(sp3)–H functionalization of primary aliphatic amines
C–H bond functionalization represents one of the most efficient and straightforward strategies for diversifying existing molecules.

Org. Chem. Front., 2024,11, 3270-3280
https://doi.org/10.1039/D4QO00324A
Electrochemical switching in mechanically interlocked molecules (MIMs)
Mechanically interlocked molecules (MIMs) which include rotaxanes and catenanes are formed by the mechanical linking of two or more components and has the ability to switch between different states in the presence of an external stimuli.

Org. Chem. Front., 2024,11, 2954-2980
https://doi.org/10.1039/D4QO00061G
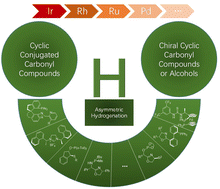
Recent advances in the metal-catalyzed asymmetric alkene hydrogenation of cyclic conjugated carbonyl compounds
This review summarizes the recent advances (2016–2023) in the stereoselective metal-catalyzed hydrogenation of cyclic α,β-unsaturated ketones, lactams and lactones since considerable developments were made. Where possible the application of these methodologies in synthesis is outlined.
Org. Chem. Front., 2024,11, 2934-2953
https://doi.org/10.1039/D4QO00227J
Review of application of the I2 and dimethyl sulfoxide combined reagent system to aryl methyl ketones for diverse transformations
The synthesis of small molecules and complex scaffolds is one of the most important topics in organic synthesis.

Org. Chem. Front., 2024,11, 2665-2692
https://doi.org/10.1039/D4QO00396A
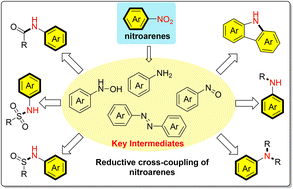
Recent progress in the synthesis of N-substituted arylamines by reductive cross-coupling of nitroarenes
This review describes the recent advances in different reaction types and catalytic systems for the construction of C–NAr and S–NAr bonds by nitroaromatic reductive cross-coupling.
Org. Chem. Front., 2024,11, 2638-2664
https://doi.org/10.1039/D4QO00059E
Catalytic intermolecular hydrofunctionalizations of ynamides
This review carefully summarizes the advances achieved in catalytic hydrofunctionalization of ynamides and is categorized by the bond formation type including C−C, C−X, C−O, C−N, C−S, C−P, C−Si, and C−Ge bonds.

Org. Chem. Front., 2024,11, 2351-2374
https://doi.org/10.1039/D4QO00301B
Advancement in the C–H bond alkylation of (hetero)arenes catalyzed by the most abundant transition metal–iron
Advancement in the direct C–H bond alkylation of arenes and heteroarenes using the catalysts based on the most abundant transition metal, iron, is summarized.

Org. Chem. Front., 2024,11, 2397-2417
https://doi.org/10.1039/D4QO00063C
Frustrated Lewis pair chemistry of alkynes
This review is focused on the chemistry of frustrated Lewis pairs (FLPs) with alkynes and surveys the range of stoichiometric and catalytic reactions enabled by this concept.

Org. Chem. Front., 2024,11, 2375-2396
https://doi.org/10.1039/D4QO00217B
Recent advances in N-heterocyclic carbene (NHC)-catalyzed fluorination and fluoroalkylation
Organofluorine compounds are of high value. NHC-catalyzed fluorination and fluoroalkylation have served as powerful and versatile vehicles for accessing the related organofluorines. This review focuses on the recent developments in this area.

Org. Chem. Front., 2024,11, 2112-2133
https://doi.org/10.1039/D3QO02098C
Recent advances in metal-free catalytic enantioselective higher-order cycloadditions
In this review, we briefly summarize the recent research progress in enantioselective higher-order cycloadditions, focusing on the preparation of medium-sized and multicyclic chiral molecules by metal-free catalysis.

Org. Chem. Front., 2024,11, 1824-1842
https://doi.org/10.1039/D4QO00017J
Recent advances in nickel-catalyzed propargylic substitution
This work summarizes recent progress in nickel-catalyzed propargylic substitution reactions, categorizing them based on the diverse types of nucleophiles involved, with a specific focus on exploring asymmetric transformations.

Org. Chem. Front., 2024,11, 1843-1857
https://doi.org/10.1039/D3QO02129G
Recent advances in hydrogen atom transfer induced C(sp3)–H functionalizations initiated by radical addition to alkynes
Recent advances in HAT-induced C(sp3)–H functionalizations triggered by radical addition to alkynes, including 5-exo-trig, 5-endo-trig, 4-exo-trig and 6-endo-trig cyclization cascades, and intermolecular functionalizations, are summarized.

Org. Chem. Front., 2024,11, 1232-1250
https://doi.org/10.1039/D3QO01876H
Advances in photoinduced radical–polar crossover cyclization (RPCC) of bifunctional alkenes
Photoinduced radical–polar crossover cyclization (RPCC) of bifunctional alkenes is of high synthetic utility to produce cyclic products with precise alkene design. This review summarizes the recent representative advances in the field of RPC.

Org. Chem. Front., 2024,11, 895-915
https://doi.org/10.1039/D3QO01929B
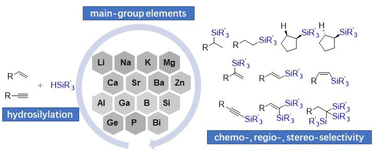
Nascent developments in main group element-catalyzed hydrosilylation and dehydrogenative silylation of alkenes and alkynes
Nascent developments in the main group element-catalyzed hydrosilylation of alkenes and alkynes.
Org. Chem. Front., 2024,11, 576-596
https://doi.org/10.1039/D3QO01777J
A cross-shaped organic framework: a multi-functional template arranging chromophores
The design and synthesis of an easily functionalizable cross-shaped organic framework is presented. The template is bis- and tetra-decorated with achiral chromophores, offering a platform to study fundamental properties such as chirality and FRET.

Org. Chem. Front., 2025,12, 1399-1408
https://doi.org/10.1039/D4QO01808G
Palladium-catalyzed dearomative Heck/[4 + 3] decarboxylative cyclization of indoles with α-oxocarboxylic acids via C–H activation
A palladium-catalyzed dearomative Heck/[4 + 3] decarboxylative cyclization of indoles with α-oxocarboxylic acids via C−H activation is reported, which offers an efficient avenue for assembling fused indolines containing a seven-membered ring.
![Graphical abstract: Palladium-catalyzed dearomative Heck/[4 + 3] decarboxylative cyclization of indoles with α-oxocarboxylic acids via C–H activation](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO02024C&imageInfo.ImageIdentifier.Year=2025)
Org. Chem. Front., 2025,12, 1122-1128
https://doi.org/10.1039/D4QO02024C
Organocatalytic enantioselective [2 + 2] cycloadditions towards chiral fused α-trifluoromethyl azetidines
Herein, we report straightforward access to chiral fused α-trifluoromethyl azetidines via organocatalytic asymmetric [2 + 2] cycloadditions. This method is characterized by mild reaction conditions, high efficiency, and excellent stereoselectivities.
![Graphical abstract: Organocatalytic enantioselective [2 + 2] cycloadditions towards chiral fused α-trifluoromethyl azetidines](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO01942C&imageInfo.ImageIdentifier.Year=2025)
Org. Chem. Front., 2025,12, 1129-1136
https://doi.org/10.1039/D4QO01942C
In situ generated aza-π-allylpalladium zwitterions as nucleophilic triggers for enantioselective [4 + 2] cycloaddition with electron-deficient alkenes toward functionalized chiral piperidines
Aza-π-allylpalladium zwitterions, in situ generated from methylene cyclic carbamates, are used as nucleophilic triggers for enantioselective [4 + 2] cycloaddition with electron-deficient alkenes, affording structurally diverse chiral piperidines.
![Graphical abstract: In situ generated aza-π-allylpalladium zwitterions as nucleophilic triggers for enantioselective [4 + 2] cycloaddition with electron-deficient alkenes toward functionalized chiral piperidines](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO02058H&imageInfo.ImageIdentifier.Year=2025)
Org. Chem. Front., 2025,12, 1115-1121
https://doi.org/10.1039/D4QO02058H
Iminonaphthofuranone synthesis via multiple-component cyclization of 2-naphthols using only molecular oxygen
A multiple-component oxidative cyclization of 2-naphthols, anilines, and ethyl glyoxylate is successfully achieved using only molecular oxygen. The products exhibit a wide spectrum of colors, adjustable from pale orange to dark purple.

Org. Chem. Front., 2025,12, 1108-1114
https://doi.org/10.1039/D4QO01939C
Transformation of a benzocorrole isomer into pyrrole-containing polycyclic molecules via copper-mediated cleavage and annulation
Copper-facilitated oxidative ring cleavage, followed by ring fusion of a corrole isomer, produced a pyrrole-embedded polycyclic molecule. This molecule demonstrates near-infrared absorption and possesses p-type semiconducting properties.

Org. Chem. Front., 2025,12, 717-724
https://doi.org/10.1039/D4QO02145B
(BO)2-doped tetrathia[7]helicenes: synthesis and property-change induced by “BO bond inversion”
(BO)2-helicene 2 was prepared and compared to its isomer 1 featuring reverted BO-bonds: spectroscopic and theoretical studies showed as “BO-inversion” is a tool to improve optoelectronic properties.
![Graphical abstract: (BO)2-doped tetrathia[7]helicenes: synthesis and property-change induced by “BO bond inversion”](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO01897D&imageInfo.ImageIdentifier.Year=2025)
Org. Chem. Front., 2025,12, 725-735
https://doi.org/10.1039/D4QO01897D
Impact of boron desymmetrization on supramolecular polymerization of BODIPY dyes
We introduce boron desymmetrization of BODIPY dyes as a new molecular design strategy towards controlled self-assembly pathways in supramolecular polymerization.

Org. Chem. Front., 2025,12, 414-421
https://doi.org/10.1039/D4QO01848F
Nickel-catalyzed reductive 1,2-alkylarylation of alkenes via a 1,5-hydrogen atom transfer (HAT) cascade
A nickel-catalyzed 1,2-alkylarylation of alkenes has been developed, leveraging amidyl radical-triggered 1,5-HAT with electrophiles as functionalizing reagents.

Org. Chem. Front., 2025,12, 57-63
https://doi.org/10.1039/D4QO01875C
Intermediate diradical character and thermal cis–trans isomerization of near-infrared absorbing thionated squaraine dyes
Near-infrared absorbing squaraine dyes incorporating 4-membered thionated oxocarbon and chalcogenopyrylium moieties exhibit intermediate diradical character, which impacts thermal cis–trans isomerization.

Org. Chem. Front., 2025,12, 42-47
https://doi.org/10.1039/D4QO01722F
Unravelling the different pathways of cyclohexene oxidation via a peroxyl radical generated from tert-butyl hydroperoxide (TBHP) by various metal salts
Two different mechanisms were obtained in the selective oxidation of cyclohexene catalyzed by CuCl2 and VCl3 respectively in presence of TBHP. The two opposed mechanisms are mainly caused by the roles of the t-BuOO˙ radical in catalytic oxidations.

Org. Chem. Front., 2025,12, 48-56
https://doi.org/10.1039/D4QO01681E
Iron-catalyzed thiolation of C(sp3)–H with sulfonyl chlorides via photoinduced ligand-to-metal charge transfer
A photoinduced FeCl3/HCl synergistic catalytic method enables C(sp3)–S bond formation via reductive cross-coupling of activated/inert C(sp3)–H and aryl/alkyl sulfonyl chlorides through ligand-to-metal charge transfer (LMCT).

Org. Chem. Front., 2024,11, 7001-7010
https://doi.org/10.1039/D4QO01488J
Pyrrolylsulfonium salts: stable, accessible and versatile pseudohalides for Stille couplings
Pyrroles functionalised with a thianthrenium or diphenylsulfonium group undergo Stille couplings with aryl or alkynyl stannanes in good yields, avoiding the issues of instability that can hamper the use of the analogous pyrrolyl halides.

Org. Chem. Front., 2024,11, 7011-7026
https://doi.org/10.1039/D4QO01793E
δ-Amination of alkyl alcohols via energy transfer photocatalysis
We present a metal-free photocatalytic method for the preparation of 1,4-iminoalcohols from bifunctional N-oximes, which are readily available from alcohol feedstocks.

Org. Chem. Front., 2024,11, 7037-7043
https://doi.org/10.1039/D4QO01522C
Deoxygenative 1,3-carbophosphination of allylic alcohols enabled by manganese pincer catalyst
The first manganese-catalyzed three-component coupling between secondary alcohol, allylic alcohol, and phosphine via borrowing hydrogen is described. This four-stage, one-pot reaction afforded ε-hydroxy phosphines in a step-economical fashion.

Org. Chem. Front., 2024,11, 7027-7036
https://doi.org/10.1039/D4QO01652A
Cobalt-catalyzed stereoselective synthesis of chiral gem-difluorocyclopropanes with vicinal stereocenters
We present a cobalt-catalyzed regio-, diastereo-, and enantioselective hydroalkylation of gem-difluorocyclopropenes, accessing chiral gem-difluorocyclopropanes with vicinal stereocenters.

Org. Chem. Front., 2024,11, 6617-6626
https://doi.org/10.1039/D4QO01585A
Cobalt-catalyzed enantioselective reductive addition of ketimine with cyclopropyl chloride to construct chiral amino esters bearing cyclopropyl fragments
Co-catalyzed asymmetric reductive addition of ketimine with cyclopropyl chloride has been realized to access diverse chiral amino esters bearing cyclopropyl fragments with broad functional group tolerance and excellent enantioselectivities.

Org. Chem. Front., 2024,11, 6311-6318
https://doi.org/10.1039/D4QO01474J
Rh(III)-catalyzed regioselective C–H activation/[3 + 2] cyclization of KHAs with iodonium ylides accessing pyrimido[1,2-a]indole derivatives
We present a protocol for the regioselective construction of three kinds of functionalized pyrrole derivatives, i.e., PIIDOs 3–5 from HKAs with iodonium ylides promoted by AgSbF6vs. NaSbF6 additives in different solvents (acetone vs. DCE).
![Graphical abstract: Rh(iii)-catalyzed regioselective C–H activation/[3 + 2] cyclization of KHAs with iodonium ylides accessing pyrimido[1,2-a]indole derivatives](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4QO01383B&imageInfo.ImageIdentifier.Year=2024)
Org. Chem. Front., 2024,11, 6048-6054
https://doi.org/10.1039/D4QO01383B
Synthesis of β-amino acid derivatives via photoredox-catalyzed radical cross–coupling of anilines with diazo compounds
A photoredox-catalyzed radical cross–coupling protocol for the synthesis of β-amino acid derivatives has been developed. This atom-economic protocol enables the synthesis of a range of β-amino acid esters in moderate to excellent yields.

Org. Chem. Front., 2024,11, 6042-6047
https://doi.org/10.1039/D4QO01396D
Nickel/ligand loading-controlled divergent and selective coupling between redox-active methylenecyclopropanes and ArBr
Diverse alkyne and dibenzylethylene derivatives were obtained via selective coupling of redox-active MCPs and ArBr under Ni/photoredox catalysis. The nickel/ligand loading tuning method played a crucial role in this divergent synthesis protocol.

Org. Chem. Front., 2024,11, 5720-5730
https://doi.org/10.1039/D4QO01346H
A HAT process of N-pyridyl radical cations for the synthesis of benzophenone-type bioisosteres
A photochemical method for generating alkyl radicals and N-pyridyl radical cations from Katritzky salts and application of the radicals in functionalization of [1.1.1]propellane to generate benzophenone-type bioisosteres was reported.

Org. Chem. Front., 2024,11, 5712-5719
https://doi.org/10.1039/D4QO01338G
Bioinspired intramolecular spirocyclopropanation of quinones as an interrupted photoredox process
Small molecules, such as trimethylamine N-oxide, interrupt intramolecular photoreactions of quinones by proton transfer and allow to convert a variety of quinones to spirocyclopropanes in a catalytic, diastereoselective, and atom-conserving manner.

Org. Chem. Front., 2024,11, 5703-5711
https://doi.org/10.1039/D4QO01291G
About this collection
Read our on-going collection of the hottest research published as advance article from Organic Chemistry Frontiers in 2024. These articles are recommended by reviewers as being of significant novelty and interest. Congratulations to all the authors whose articles are featured!