Themed collection Direct C-H Functionalization in Method Development and Late Stage Functionalization

Combining enzymes and organometallic complexes: novel artificial metalloenzymes and hybrid systems for C–H activation chemistry
This review describes the advances in the design and application of novel artificial metalloenzymes in C–H activation reactions.
Org. Biomol. Chem., 2019,17, 7114-7123
https://doi.org/10.1039/C9OB01091B
Late stage functionalization of heterocycles using hypervalent iodine(III) reagents
Iodine(III) reagents represent a powerful tool for the late-stage functionalization of a variety of heterocycles through a range of transformations.
Org. Biomol. Chem., 2019,17, 6326-6341
https://doi.org/10.1039/C9OB00694J
Visible light-mediated organophotocatalyzed C–H bond functionalization reactions
This review provides a current overview of the recent developments in the visible light mediated organophotocatalyzed C–H bond functionalization methodologies.
Org. Biomol. Chem., 2019,17, 5475-5489
https://doi.org/10.1039/C9OB00834A
Recent advances in metal-free aerobic C–H activation
An overview of recent reactions based on the metal-free, dioxygen-induced, C–H activation of various radical precursors.
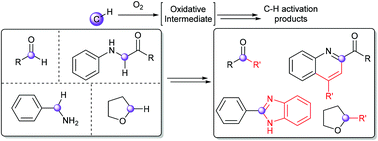
Org. Biomol. Chem., 2019,17, 2865-2872
https://doi.org/10.1039/C9OB00339H
Solvent-free ruthenium-catalysed triflate coupling as a convenient method for selective azole-o-C–H monoarylation
A convenient ruthenium-catalysed N-directed C–H monoarylation of arylpyrazoles with a good selectivity of up to 96% is reported.
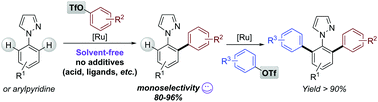
Org. Biomol. Chem., 2019,17, 5916-5919
https://doi.org/10.1039/C9OB00806C
Metal-free late-stage C(sp2)–H functionalization of N-aryl amines with various sodium salts
Metal-free consecutive C(sp2)–X (X = Cl, Br, S, N) bond formations of N-aryl amines (cyclic, fused, carbamate, and aminium radicals) were achieved under mild conditions using PIFA and simple nonharmful sodium salts.
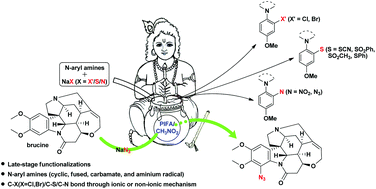
Org. Biomol. Chem., 2020,18, 450-464
https://doi.org/10.1039/C9OB02217A
Mild C–H functionalization of alkanes catalyzed by bioinspired copper(II) cores
Three new copper(II) coordination compounds with aminoalcohol and carboxylate type ligands were self-assembled, fully characterized, and applied as efficient homogeneous catalysts for the mild oxidation and carboxylation of saturated hydrocarbons.
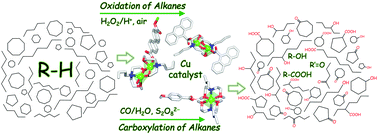
Org. Biomol. Chem., 2019,17, 7706-7714
https://doi.org/10.1039/C9OB01442J
Ruthenium(II)-catalysed selective C(sp2)–H bond benzoxylation of biologically appealing N-arylisoindolinones
A general site- and regio-selective aromatic C–H bond benzoxylation reaction using biologically-relevant isoindolinones as weak directing groups has been developed.
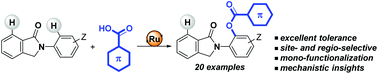
Org. Biomol. Chem., 2019,17, 7517-7525
https://doi.org/10.1039/C9OB01122F
A computational study on the identity of the active catalyst structure for Ru(II) carboxylate assisted C–H activation in acetonitrile
Density Functional Theory (DFT) calculations using a consistent methodology accounting for solvation, dispersion and thermal effects have been used to study C–H activation of the simple directing group substrate 2-phenylpyridine (a-H).
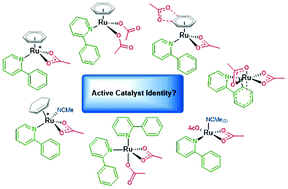
Org. Biomol. Chem., 2019,17, 6678-6686
https://doi.org/10.1039/C9OB01092K
Dehydrogenative C(sp3)–H bond functionalization of tetrahydroisoquinolines mediated by organic oxidants under mild conditions
DDQ and IBX are effective SET oxidants of N-aryl-protected and unprotected THIQs. Mannich and Strecker type functionalizations ensued in an overall CDC reaction.
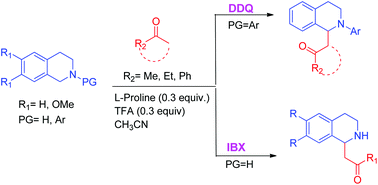
Org. Biomol. Chem., 2019,17, 6420-6425
https://doi.org/10.1039/C9OB01090D
Synthesis of fluorenyl alcohols via cooperative palladium/norbornene catalysis
Catalytic synthesis of fluorenols by synchronization of two stoichiometric reactions: (a) Pd(II)-mediated oxidation of alcohols to ketones and (b) Pd(0)/norbornene-catalyzed reaction of aryl iodides with ortho-bromoacetophenones.
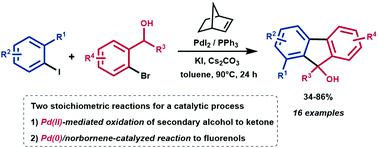
Org. Biomol. Chem., 2019,17, 6165-6173
https://doi.org/10.1039/C9OB01085H
Manganese(III) acetate-mediated direct C(sp2)–H-sulfonylation of enamides with sodium and lithium sulfinates
A Mn(OAc)3 mediated oxidative C(sp2)–H sulfonylation of enamides and encarbamates with sodium and lithium sulfinates is reported.
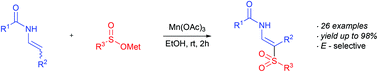
Org. Biomol. Chem., 2019,17, 5538-5544
https://doi.org/10.1039/C9OB00825J
Late-stage C–H amination of abietane diterpenoids
Application of Rh(II)-catalyzed C–H amination reactions allows the selective late-stage diversification of abietane diterpenoids.
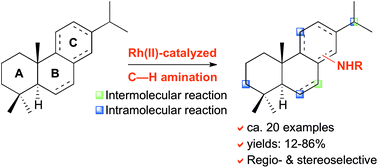
Org. Biomol. Chem., 2019,17, 4736-4746
https://doi.org/10.1039/C9OB00272C
Investigations of the generality of quaternary ammonium salts as alkylating agents in direct C–H alkylation reactions: solid alternatives for gaseous olefins
C–H alkylation reactions using short chain olefins as alkylating agents could be operationally simplified on the lab scale by using quaternary ammonium salts as precursors for these gaseous reagents.
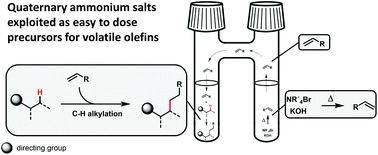
Org. Biomol. Chem., 2019,17, 4024-4030
https://doi.org/10.1039/C9OB00243J
Ru(II)-Catalysed synthesis of (1H)-isothiochromenes by oxidative coupling of benzylthioethers with internal alkynes
(1H)-Isothiochromenes have been prepared by Ru-catalysed oxidative coupling of benzylthioethers with internal alkynes.
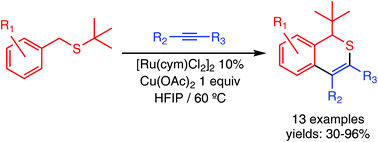
Org. Biomol. Chem., 2019,17, 2542-2547
https://doi.org/10.1039/C8OB03201G