Ruthenium(ii)-catalysed selective C(sp2)–H bond benzoxylation of biologically appealing N-arylisoindolinones†
Abstract
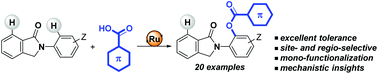
Site- and regio-selective aromatic C–H bond benzoxylations were found to take place using biologically appealing N-arylisoindolinones under ruthenium(II) catalysis in the presence of (hetero)aromatic carboxylic acid derivatives as coupling partners. Besides the presence of two potential C(sp2)–H sites available for functionalization in the substrates, exclusive ortho selectivity was achieved in the phenyl ring attached to the nitrogen atom. Notably, the reactions occurred in a selective manner as only mono-functionalized products were formed and they tolerated a large number of functional chemical groups. The ability of the cyclic tertiary amide within the isoindolinone skeleton to act as a weak directing group in order to accommodate six-membered ring ruthenacycle intermediates appears to be the key to reach such high levels of selectivity. In contrast, the more sterically demanding cyclic imides were unreactive under identical reaction conditions.
- This article is part of the themed collections: Synthetic methodology in OBC and Direct C-H Functionalization in Method Development and Late Stage Functionalization


 Please wait while we load your content...
Please wait while we load your content...