Themed collection Benchmark Experiments for Numerical Quantum Chemistry

Benchmark experiments for numerical quantum chemistry
This themed collection addresses the question of numerical quantum chemistry benchmarking in its many facets, with a focus on experiment-driven practices.

Phys. Chem. Chem. Phys., 2023,25, 26415-26416
https://doi.org/10.1039/D3CP90186F
Benchmarks for transition metal spin-state energetics: why and how to employ experimental reference data?
Experimental data provide reliable benchmarks for computed spin-state energetics.

Phys. Chem. Chem. Phys., 2023,25, 30800-30820
https://doi.org/10.1039/D3CP03537A
Noncovalently bound molecular complexes beyond diatom–diatom systems: full-dimensional, fully coupled quantum calculations of rovibrational states
The methodological advances made in recent years have significantly extended the range and dimensionality of noncovalently bound molecular complexes for which full-dimensional quantum calculations of their rovibrational states are feasible.
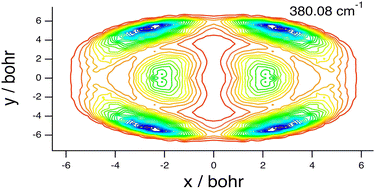
Phys. Chem. Chem. Phys., 2022,24, 24655-24676
https://doi.org/10.1039/D2CP04005K
Setting up the HyDRA blind challenge for the microhydration of organic molecules
The first theory blind challenge addressing the effect of microsolvation on water vibrations is launched.

Phys. Chem. Chem. Phys., 2022,24, 11442-11454
https://doi.org/10.1039/D2CP01119K
Connections between the accuracy of rotational constants and equilibrium molecular structures
Rotational constants and equilibrium structures are strongly connected. To meet the 0.1% accuracy in the computation of the former, an accuracy of 0.0005–0.001 Å is required for bond lengths.

Phys. Chem. Chem. Phys., 2023,25, 1421-1429
https://doi.org/10.1039/D2CP04706C
On the vibrations of formic acid predicted from first principles
In this article, we review recent first principles, anharmonic studies on the molecular vibrations of gaseous formic acid in its monomer form.
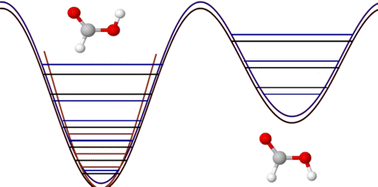
Phys. Chem. Chem. Phys., 2022,24, 28109-28120
https://doi.org/10.1039/D2CP04417J
The computational road to reactivity scales
Reactivity scales are powerful research tools. This tutorial shows how to create and use them on the computer.
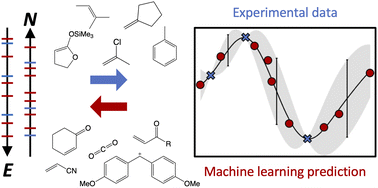
Phys. Chem. Chem. Phys., 2023,25, 2717-2728
https://doi.org/10.1039/D2CP03937K
The transferability limits of static benchmarks
Since the errors of quantum chemical methods can strongly vary across chemical space, the transferability of traditional benchmarks is limited. This can be overcome by quantifying the uncertainty of quantum chemical results in a system-focused way.
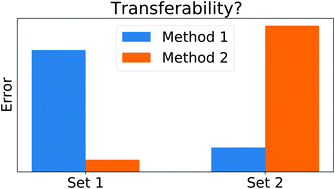
Phys. Chem. Chem. Phys., 2022,24, 14692-14698
https://doi.org/10.1039/D2CP01725C
The first HyDRA challenge for computational vibrational spectroscopy
A joint community effort to critically evaluate quantum chemical approaches to the prediction of vibrational shifts of hydrates in the gas phase.

Phys. Chem. Chem. Phys., 2023,25, 22089-22102
https://doi.org/10.1039/D3CP01216F
Sub 20 cm−1 computational prediction of the CH bond energy – a case of systematic error in computational thermochemistry
Benchmarking state-of-the-art computations of D0(CH) with Active Thermochemical Tables reveals a systematic error in prior high-level computations.

Phys. Chem. Chem. Phys., 2023,25, 21162-21172
https://doi.org/10.1039/D2CP03964H
How do density functionals affect the Hirshfeld atom refinement?
The X–H bonds obtained from Hirshfeld atom refinements elongate with Hartree–Fock exchange in contrast to geometry optimizations. These results suggest new venues in benchmarking density functionals with respect to experimental crystallographic data.
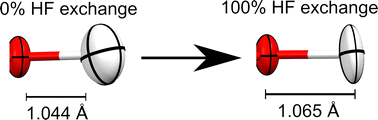
Phys. Chem. Chem. Phys., 2023,25, 12702-12711
https://doi.org/10.1039/D2CP04098K
Hydrogen sharing between two nitroxyl radicals in the gas phase and other microsolvation effects on the infrared spectrum of a bulky hydroxylamine
Despite OH stretch excitation, the hydrogen atom between two TEMPO radicals does not tunnel fast enough to show up as a splitting in the supersonic jet infrared spectrum.

Phys. Chem. Chem. Phys., 2023,25, 11324-11330
https://doi.org/10.1039/D3CP01156A
Improved modeling of anharmonicity for furan microsolvation
1D-hindered rotor profiles are corrected for coupled cluster energies at stationary points. Probability density functions at each energy level allow to resolve different conformations within the scan.
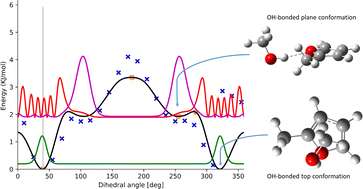
Phys. Chem. Chem. Phys., 2023,25, 11316-11323
https://doi.org/10.1039/D2CP03907A
Subtle hydrogen bond preference and dual Franck–Condon activity – the interesting pairing of 2-naphthol with anisole
In contrast to standard DFT predictions, 2-naphthol is shown to dock on the oxygen of anisole, with excitation-dependent angular geometry.
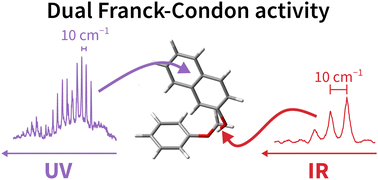
Phys. Chem. Chem. Phys., 2023,25, 10427-10439
https://doi.org/10.1039/D3CP00160A
Benchmarking quantum chemical methods for accurate gas-phase structure predictions of carbonyl compounds: the case of ethyl butyrate
High-resolution spectroscopy techniques play a pivotal role to validate and benchmark methods from quantum chemistry. This is crucial for small esters which exhibit a soft-degree of freedom around the C–C bond in proximity to the carbonyl moiety.
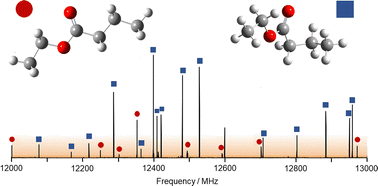
Phys. Chem. Chem. Phys., 2023,25, 7688-7696
https://doi.org/10.1039/D2CP05774C
A rotational investigation of the three isomeric forms of cyanoethynylbenzene (HCC-C6H4-CN): benchmarking experiments and calculations using the “Lego brick” approach
Benchmarking experiments and calculations using the “Lego brick” approach on cyanoethynylbenzene isomers.

Phys. Chem. Chem. Phys., 2023,25, 6397-6405
https://doi.org/10.1039/D2CP04825F
On the accuracy of orbital based multi-level approaches for closed-shell transition metal chemistry
We studied the accuracy of projection-based WFT-in-DFT embedding and LCCSD(T0):LMP2 embedding for predicting reaction energies and barriers of typical, closed-shell transition-metal-based reactions.

Phys. Chem. Chem. Phys., 2023,25, 4635-4648
https://doi.org/10.1039/D2CP05056K
Hierarchical analysis of the target-based scoring function modification for the example of selected class A GPCRs
Targeted scoring function for different levels of biological hierarchy of selected GPCRs, leads to improvement in molecular docking predictive power.
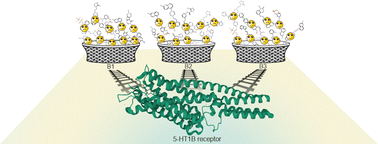
Phys. Chem. Chem. Phys., 2023,25, 3513-3520
https://doi.org/10.1039/D2CP04671G
Benchmarking the quadrupolar coupling tensor for chlorine to probe weak-bonding interactions
Rotational spectroscopy relies on quantum chemical calculations to interpret hyperfine splitting.
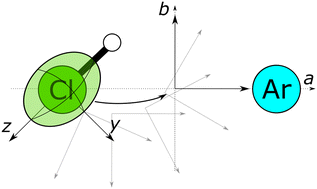
Phys. Chem. Chem. Phys., 2023,25, 2420-2429
https://doi.org/10.1039/D2CP04067K
Cation-responsive cavity expansion of valinomycin revealed by cryogenic ion trap infrared spectroscopy
The change in the cavity size of valinomycin in response to the size of the metal ion is revealed by cryogenic ion trap infrared spectroscopy.
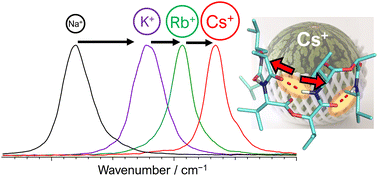
Phys. Chem. Chem. Phys., 2023,25, 1075-1080
https://doi.org/10.1039/D2CP04570B
Large 31P-NMR enhancements in liquid state dynamic nuclear polarization through radical/target molecule non-covalent interaction
Large 31P-NMR enhancements are observed with DNP in PPh3 doped with BDPA radical, while they are reduced when a nitroxide radical or triphenylphosphine-oxide are used instead. This is due to different non-covalent radical/target molecule interactions.
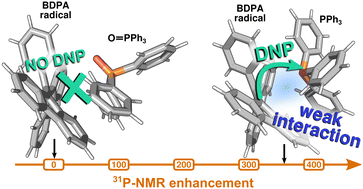
Phys. Chem. Chem. Phys., 2023,25, 822-828
https://doi.org/10.1039/D2CP04092A
Raising the benchmark potential of a simple alcohol-ketone intermolecular balance
A simple asymmetric ketone directs solvent molecules to the shorter end and this subtle preference is useful for theory benchmarking.
Phys. Chem. Chem. Phys., 2023,25, 384-391
https://doi.org/10.1039/D2CP05141A
Benchmarking acetylthiophene derivatives: methyl internal rotations in the microwave spectrum of 2-acetyl-5-methylthiophene
The time required for recording survey scans of the microwave spectrum of the title molecule with two inequivalent methyl internal rotors was significantly reduced with the help of adequate spectral analysis skill and quantum chemical benchmarking.

Phys. Chem. Chem. Phys., 2023,25, 509-519
https://doi.org/10.1039/D2CP03897H
Adsorption and keto–enol-tautomerisation of butanal on Pd(111)
Stabilization of normally unstable enol form of carbonyl compounds on surfaces is realized via intermolecular interactions.
Phys. Chem. Chem. Phys., 2022,24, 29480-29494
https://doi.org/10.1039/D2CP04398J
High-dimensional neural network potentials for accurate vibrational frequencies: the formic acid dimer benchmark
A high-dimensional neural network potential has been developed to study the harmonic and anharmonic frequencies of the formic acid dimer with coupled cluster accuracy.
Phys. Chem. Chem. Phys., 2022,24, 29381-29392
https://doi.org/10.1039/D2CP03893E
Quantum versus classical unimolecular fragmentation rate constants and activation energies at finite temperature from direct dynamics simulations
We show that ring polymer molecular dynamics is able to include nuclear quantum effects in unimolecular dissociation simulations. In this way, rate constants directly take into account both quantum statistics and anharmonic behavior.
Phys. Chem. Chem. Phys., 2022,24, 29357-29370
https://doi.org/10.1039/D2CP03809A
Benchmark of a functional-group database for distributed polarizability and dipole moment in biomolecules
Application of group polarizability database towards better prediction of electric moments and electrostatic properties of biomolecules.

Phys. Chem. Chem. Phys., 2022,24, 29495-29504
https://doi.org/10.1039/D2CP04052B
Reliable prediction of association (free) energies of supramolecular complexes with heavy main group elements – the HS13L benchmark set
We introduce a set of 13 supramolecular complexes featuring diverse non-covalent interactions with heavy main group elements (Zn, As, Se, Te, Br, I), high charges (−2 up to +4), and large systems with up to 266 atoms (HS13L).
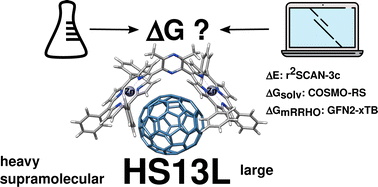
Phys. Chem. Chem. Phys., 2022,24, 28831-28843
https://doi.org/10.1039/D2CP04049B
Theoretical study of the CO2–O2 van der Waals complex: potential energy surface and applications
A four-dimensional-potential energy surface (4D-PES) of the atmospherically relevant CO2–O2 van der Waals complex is generated using ab initio methodology. Induced complexation shifts and second virial coefficient are also presented.
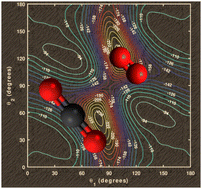
Phys. Chem. Chem. Phys., 2022,24, 28984-28993
https://doi.org/10.1039/D2CP04101D
Examining the gas-phase homodimers of 3,3,3-trifluoro-1,2-epoxypropane using quantum chemistry and microwave spectroscopy
Quantum chemistry and rotational spectroscopy work hand-in-hand in the characterization of a potential chiral tag.
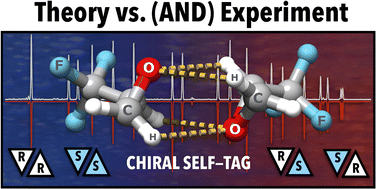
Phys. Chem. Chem. Phys., 2022,24, 28495-28505
https://doi.org/10.1039/D2CP04663F
Accuracy of quantum chemistry structures of chiral tag complexes and the assignment of absolute configuration
The absolute configuration of a molecule can be established by analysis of molecular rotational spectra of the analyte complexed with a small chiral molecule of known configuration.
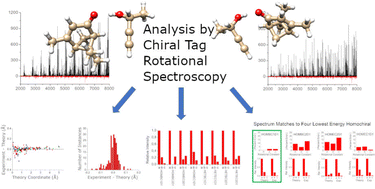
Phys. Chem. Chem. Phys., 2022,24, 27705-27721
https://doi.org/10.1039/D2CP04060C
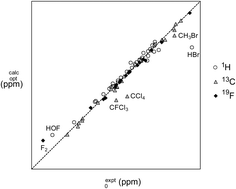
A new double-reference correction scheme for accurate and efficient computation of NMR chemical shieldings
Our novel correction procedure yields high-accuracy DFT predictions of absolute NMR shieldings and enables outliers due to relativistic effects or manifestly inadequate modelling of electron correlation to be easily and unambiguously identified.
Phys. Chem. Chem. Phys., 2022,24, 27055-27063
https://doi.org/10.1039/D2CP03992C
The many forms of alpha-methoxy phenylacetic acid in the gas phase: flexibility, internal dynamics, and their intramolecular interactions
Five conformers of the flexible molecule alpha-methoxy phenylacetic acid were identified using rotational spectroscopy. The conformational landscape, internal dynamics, and intramolecular interactions were investigated.

Phys. Chem. Chem. Phys., 2022,24, 27312-27320
https://doi.org/10.1039/D2CP03962A
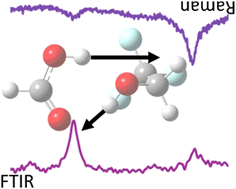
Coupled proton vibrations between two weak acids: the hinge complex between formic acid and trifluoroethanol
Raman and FTIR spectra of an acid–alcohol complex show complementary signatures from acidic and alcoholic OH stretching, proving its existence.
Phys. Chem. Chem. Phys., 2022,24, 26449-26457
https://doi.org/10.1039/D2CP04176F
Interactions of limonene with the water dimer
The interactions of limonene with the water dimer have been characterised through the identification of seven different isomers.

Phys. Chem. Chem. Phys., 2022,24, 26529-26538
https://doi.org/10.1039/D2CP04174J
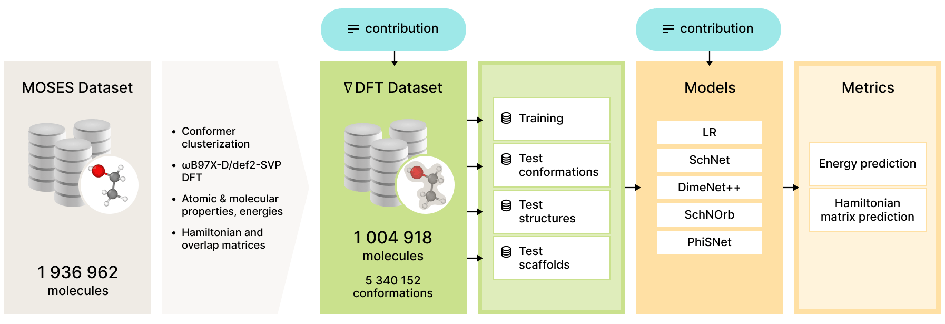
nablaDFT: Large-Scale Conformational Energy and Hamiltonian Prediction benchmark and dataset
In this work we present nablaDFT, the new dataset and benchmark for the Density Functional Theory Hamiltonian and energy prediction. We provide data for over 1 million different molecules and over 5 million conformations and baseline models for both tasks.
Phys. Chem. Chem. Phys., 2022,24, 25853-25863
https://doi.org/10.1039/D2CP03966D
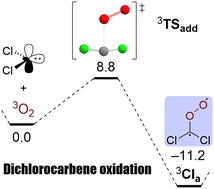
The activated reaction of dichlorocarbene with triplet molecular oxygen
The reaction of dichlorocarbene and molecular oxygen in their respective ground states is hindered by a considerable barrier rendering a diffusion controlled formation of the resulting Criegee intermediate unlikely.
Phys. Chem. Chem. Phys., 2022,24, 25834-25841
https://doi.org/10.1039/D2CP03889G
Halide–propene complexes: validated DSD-PBEP86-D3BJ calculations and photoelectron spectroscopy
Anion photoelectron spectroscopy has been used to determine the electron binding energies of the X−⋯C3H6 (X = Cl, Br, I) complexes.
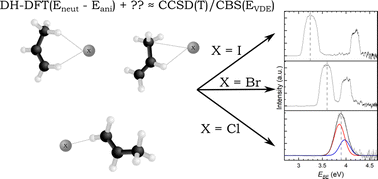
Phys. Chem. Chem. Phys., 2022,24, 25842-25852
https://doi.org/10.1039/D2CP03796C
Cryogenic IR and UV spectroscopy of isomer-selected cytosine radical cation
The UV photodissociation of cryogenic-cooled isomer-selected cytosine–silver complex leads to the production of cytosine radical cation without isomerization.

Phys. Chem. Chem. Phys., 2022,24, 25182-25190
https://doi.org/10.1039/D2CP03953B
A litmus test for the balanced description of dispersion interactions and coordination chemistry of lanthanoids
The influence of long-range interactions on the structure of complexes of Eu(III) with four 9-hydroxy-phenalen-1-one ligands (HPLN) and one alkaline earth metal dication [Eu(PLN)4AE]+ (AE: Mg, Ca, Sr, and Ba) is analyzed.
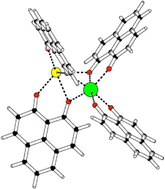
Phys. Chem. Chem. Phys., 2022,24, 25106-25117
https://doi.org/10.1039/D2CP01414A
Towards high-resolution X-ray scattering as a probe of electron correlation
We demonstrate that X-ray scattering can be used as a probe of electron–electron correlation.

Phys. Chem. Chem. Phys., 2022,24, 24542-24552
https://doi.org/10.1039/D2CP02933B
How change in chirality prevents β-amyloid type interaction in a protonated cyclic dipeptide dimer
The protonated dimer of cyclo (Phe-His) is studied in a ion trap by IR spectroscopy. A β-sheet interaction exists in cyclo (LPhe-LHis) and not in cyclo (LPhe-DHis). A change in chirality of one of the residues thus prevents amyloid-type clustering.

Phys. Chem. Chem. Phys., 2022,24, 19783-19791
https://doi.org/10.1039/D2CP03110H
Can domain-based local pair natural orbitals approaches accurately predict phosphorescence energies?
A DLPNO-CCSD(T)-based protocol allows the accurate calculation of phosphorescence energies for aromatic compounds. The figure shows the computed and experimental spectra for a carbazolyl dicyanobenzene photocatalyst.
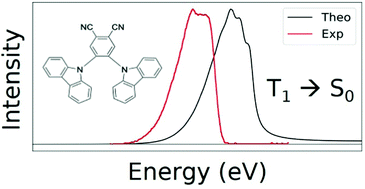
Phys. Chem. Chem. Phys., 2022,24, 14228-14241
https://doi.org/10.1039/D2CP01623K
Performance of GFN1-xTB for periodic optimization of metal organic frameworks
We benchmark GFB-xTB for periodic geometry optimisations of CoRE-MOF structures and find both local and global geometry well conserved.
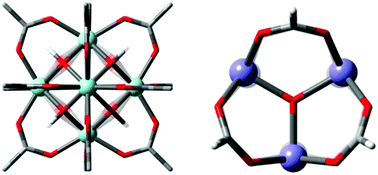
Phys. Chem. Chem. Phys., 2022,24, 10906-10914
https://doi.org/10.1039/D2CP00184E
About this collection
Benchmarking is a core activity in the development of quantum chemical methods. This is something which is recognized across the different fields of application but the scope of benchmarking and the processes behind it are often times a matter of discussion. Theory-based benchmarking is an accepted standard, however this is not an end-all solution, given that theoretical predictions have to at some point be verified experimentally. This raises totally new questions and challenges. It requires major effort from both sides to foster the fruitful interplay between experiment and theory and to improve the predictive power of numerical quantum approaches based on experimental data.
Looking into the future, it also warrants greater care in the way data is managed and shared among communities, as well as building pressure on the reproducibility of both experimental and simulated data. This themed collection of papers will be dedicated to these different facets of experimental benchmarking activities and their consequences for quantum chemical predictions. It is meant to inspire new approaches and targets for the productive interleaving of in silico and in vitro studies of molecular systems.
Guest Edited by: Ricardo Mata (University of Göttingen), Martin Suhm (University of Göttingen) and Anne Zehnacker (University of Paris – Saclay)