Themed collection Celebrating the 2019 Nobel Prize in Chemistry

Electrochemical energy storage in a sustainable modern society
Oxide/polymer separator membranes allow alternative strategies for Li-ion and Na-ion batteries storing electrical energy for the grid.
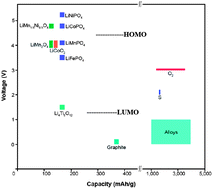
Energy Environ. Sci., 2014,7, 14-18
https://doi.org/10.1039/C3EE42613K
Room-temperature liquid metal and alloy systems for energy storage applications
Liquid metals and alloy systems that feature inherent deformability, high electronic conductivity, and superior electrochemical properties have enabled further development of next-generation energy storage devices.
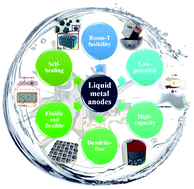
Energy Environ. Sci., 2019,12, 2605-2619
https://doi.org/10.1039/C9EE01707K
Inorganic nanomaterials for batteries
The availability of inorganic materials at the nano-dimension opens up opportunities for advanced battery designs and architectures. However, such materials must be chosen carefully to avoid deleterious side-reactions.
Dalton Trans., 2008, 5424-5431
https://doi.org/10.1039/B806372A
Batteries for electric road vehicles
The dependence of modern society on the energy stored in a fossil fuel is not sustainable.
Dalton Trans., 2018,47, 645-648
https://doi.org/10.1039/C7DT03026F
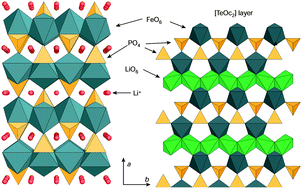
What can we learn about battery materials from their magnetic properties?
Magnetic studies help better understand battery materials.
J. Mater. Chem., 2011,21, 9865-9875
https://doi.org/10.1039/C1JM00024A
Some transition metal (oxy)phosphates and vanadium oxides for lithium batteries
These electrochemically active phosphate tunnel structures, with one-dimensional lithium diffusion and different magnetic properties, are prime candidates for lithium batteries.
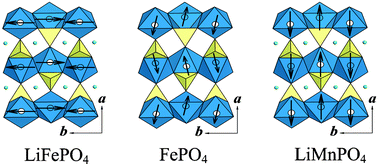
J. Mater. Chem., 2005,15, 3362-3379
https://doi.org/10.1039/B501961C
A chemistry and material perspective on lithium redox flow batteries towards high-density electrical energy storage
This review summarizes the latest advances and challenges from a chemistry and material perspective on Li-redox flow batteries that combine the synergistic features of Li-ion batteries and redox flow batteries towards large-scale high-density energy storage systems.

Chem. Soc. Rev., 2015,44, 7968-7996
https://doi.org/10.1039/C5CS00289C
Spinel materials for high-voltage cathodes in Li-ion batteries
The success of lithium-ion batteries in small-scale applications translates to large-scale applications, with an important impact in the future of the environment by improving energy efficiency and reduction of pollution.

RSC Adv., 2014,4, 154-167
https://doi.org/10.1039/C3RA45706K
Lithium–oxygen batteries: bridging mechanistic understanding and battery performance
Fundamental and practical challenges of Li–O2 batteries are evaluated to provide new insights into the state of understanding, and to highlight promising research directions for the development of stable, efficient and rechargeable high-energy Li–O2 systems.
Energy Environ. Sci., 2013,6, 750-768
https://doi.org/10.1039/C3EE23966G
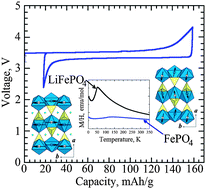
Development and challenges of LiFePO4 cathode material for lithium-ion batteries
In this review, we focus on LiFePO4 and discuss its structure, synthesis, electrochemical behavior, mechanism, and the problems encountered in its application.
Energy Environ. Sci., 2011,4, 269-284
https://doi.org/10.1039/C0EE00029A
Enabling multi-electron reaction of ε-VOPO4 to reach theoretical capacity for lithium-ion batteries
Synthesizing highly crystalline nano-sized ε-VOPO4 particles is the key to achieve theoretical capacity of 2 Li+ intercalation in lithium-ion batteries.
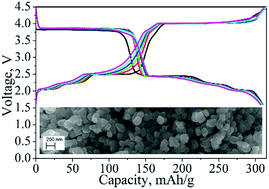
Chem. Commun., 2018,54, 7802-7805
https://doi.org/10.1039/C8CC02386G
A new approach for recycling waste rubber products in Li–S batteries
Vulcanized rubber products contain polymer backbones crosslinked with sulfur to improve mechanical strength.
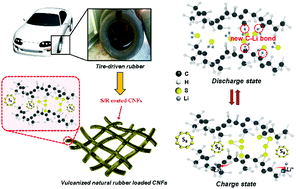
Energy Environ. Sci., 2017,10, 86-90
https://doi.org/10.1039/C6EE02770A
Enhanced Li+ ion transport in LiNi0.5Mn1.5O4 through control of site disorder
Site disorder not only enhances Li+ ion transport in LiNi0.5Mn1.5O4 lattice, but also fundamentally changes the phase transition pathway during electrochemical reactions.
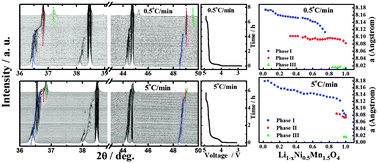
Phys. Chem. Chem. Phys., 2012,14, 13515-13521
https://doi.org/10.1039/C2CP43007J
Prussian blue: a new framework of electrode materials for sodium batteries
Prussian blue and its analogues consisting of different transition-metal ions (Fe, Mn, Ni, Cu, Co and Zn) have been synthesized at room temperature.
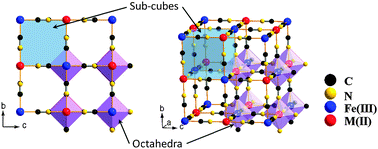
Chem. Commun., 2012,48, 6544-6546
https://doi.org/10.1039/C2CC31777J
Micropores-in-macroporous gel polymer electrolytes for alkali metal batteries
Micropores-in-macroporous polymer membranes containing an immobilized-liquid electrolyte enable dendrite-free alkali metal batteries.
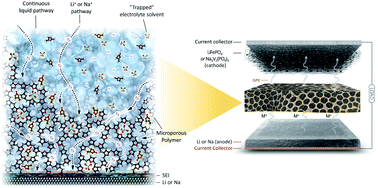
Sustainable Energy Fuels, 2020,4, 177-189
https://doi.org/10.1039/C9SE00690G
Rational synthesis and electrochemical performance of LiVOPO4 polymorphs
A thorough study on the stability of LiVOPO4 polymorphs to determine which is the most promising for Li-ion batteries.
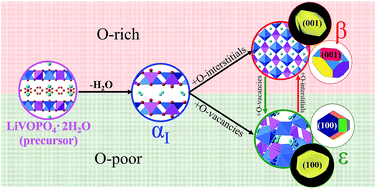
J. Mater. Chem. A, 2019,7, 8423-8432
https://doi.org/10.1039/C8TA12531G
Role of disorder in limiting the true multi-electron redox in ε-LiVOPO4
Ball-milling-induced disorder and defects impede multi-electron redox in ε-LiVOPO4 and trigger side reactions.
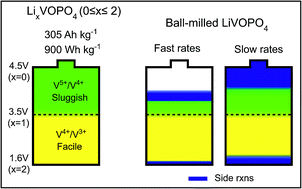
J. Mater. Chem. A, 2018,6, 20669-20677
https://doi.org/10.1039/C8TA06469E
Identifying the chemical and structural irreversibility in LiNi0.8Co0.15Al0.05O2 – a model compound for classical layered intercalation
Anisotropic disorder along the c-axis results from static disorder.
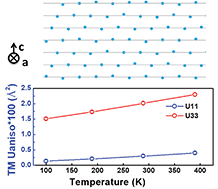
J. Mater. Chem. A, 2018,6, 4189-4198
https://doi.org/10.1039/C7TA10829J
Comparison of the polymorphs of VOPO4 as multi-electron cathodes for rechargeable alkali-ion batteries
VO6/VO5–PO4 frameworks govern electrochemical performance in VOPO4 polymorphs.
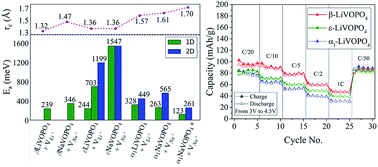
J. Mater. Chem. A, 2017,5, 17421-17431
https://doi.org/10.1039/C7TA04558A
Eldfellite, NaFe(SO4)2: an intercalation cathode host for low-cost Na-ion batteries
The mineral eldfellite, NaFe(SO4)2, is characterized as a potential cathode for a Na-ion battery that can be fabricated in charged-state.
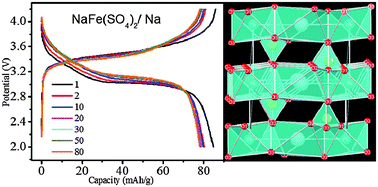
Energy Environ. Sci., 2015,8, 3000-3005
https://doi.org/10.1039/C5EE02274F
Sulfur encapsulated in porous hollow CNTs@CNFs for high-performance lithium–sulfur batteries
A novel carbon-sulfur nanoarchitecture with a high Brunauer–Emmett–Teller (BET) specific surface area of ~80 m2 g−1 and a total pore volume of ~0.2cm3 g−1 shows a high capacity of ~ 700 mAh g−1 at 1 C and 520 mAh g−1 at 5 C after 100 cycles, which makes it a superior cathode material for a rechargeable Li–S battery.

J. Mater. Chem. A, 2014,2, 10126-10130
https://doi.org/10.1039/C4TA01823K
Optimizing Li+ conductivity in a garnet framework
The garnet-related oxides with the general formula Li7−xLa3Zr2−xTaxO12 (0 ≤ x ≤ 1) were prepared by conventional solid-state reaction.
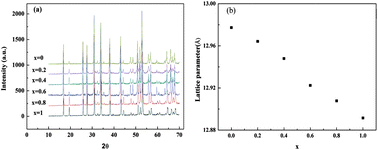
J. Mater. Chem., 2012,22, 15357-15361
https://doi.org/10.1039/C2JM31413D
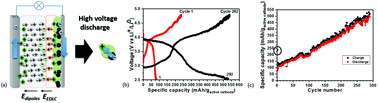
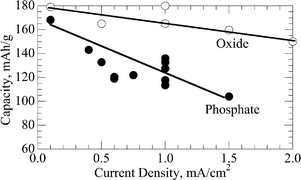
Rechargeable alkali-ion cathode-flow battery
Schematic cell of rechargeable alkali-ion cathode-flow battery.

J. Mater. Chem., 2011,21, 10113-10117
https://doi.org/10.1039/C0JM04222F
The synthesis, characterization and electrochemical behavior of the layered LiNi0.4Mn0.4Co0.2O2 compound
The lithium electrochemical cell LiNi0.4Mn0.4Co0.2O2 gave the highest reversible capacity of the mixed transition metal layered compounds studied.
J. Mater. Chem., 2004,14, 214-220
https://doi.org/10.1039/B309834F