Identifying the chemical and structural irreversibility in LiNi0.8Co0.15Al0.05O2 – a model compound for classical layered intercalation†
Abstract
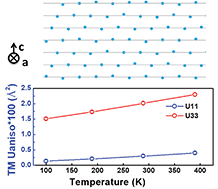
In this work, we extracted 95% of the electrochemically available Li from LiNi0.8Co0.15Al0.05O2 (NCA) by galvanostatically charging the NCA/MCMB full cell to 4.7 V. Joint powder X-ray and neutron diffraction (XRD & ND) studies were undertaken for NCA at highly charged states at the first cycle, and discharged states at different cycles. The results indicate that the bulk structure of NCA maintains the O3 structure up to the extraction of 0.90 Li per formula unit. In addition, we found that the transition metal layer becomes more disordered along the c-axis than along the a- and b-axes upon charging. This anisotropic disorder starts to develop no later than 4.3 V on charge and continues to grow until the end of charge. As Li is re-inserted during discharge, the structure that resembles the pristine NCA is recovered. The irreversible loss of Li and the migration of Ni to the Li layer have been quantified by the joint XRD and ND refinement and the results were further verified by solid state 7Li NMR and magnetic measurements. Our work clearly demonstrates that the NCA bulk retains a robust, single phase O3 structure throughout the wide delithiation range (up to 0.9 Li per formula unit of NCA) and is suitable for higher energy density usage with proper modifications.
- This article is part of the themed collection: Celebrating the 2019 Nobel Prize in Chemistry


 Please wait while we load your content...
Please wait while we load your content...