A new approach for recycling waste rubber products in Li–S batteries†
Abstract
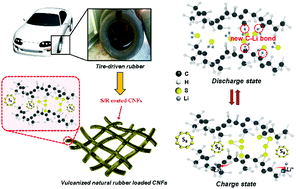
Vulcanized rubber products contain polymer backbones crosslinked with sulfur to improve mechanical strength. Burning of waste rubber products emits toxic gases, and recycling of rubber by breaking the C–S bond with costly reagents and heat is also haunted by environmental concerns. The crosslinked polymers can be extracted chemically at room temperature and, by a simple solution-drop method, used to prepare a bifunctional cathode layer on the cathode current collector of a Li–S battery. The C–S bond of the crosslinking sulfur can be broken reversibly in a discharge/recharge cycle to provide a sulfur source, and the broken carbon bond can capture a lithium polysulfide soluble in a liquid organic electrolyte by forming a C–Li–S bond during discharge. During charge, the Li is extracted and the C–S bond is reformed. However, added sulfur powder is also needed in the cathode of a Li–S battery. The data also provide a low-cost way to recycle waste rubber electrochemically.
- This article is part of the themed collections: 2017 Energy and Environmental Science HOT articles and Celebrating the 2019 Nobel Prize in Chemistry

 Please wait while we load your content...
Please wait while we load your content...