Themed collection 2019 Organic Chemistry Frontiers HOT articles

Synthesis of polycyclic aromatic hydrocarbons by palladium-catalysed [3 + 3] annulation
A new [3 + 3] annulation method for the synthesis of polycyclic aromatic hydrocarbons (PAHs) from two smaller aromatic fragments is reported. Packing structures for four products were obtained and relation to that of parent perylene was discussed.
![Graphical abstract: Synthesis of polycyclic aromatic hydrocarbons by palladium-catalysed [3 + 3] annulation](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D0QO00968G&imageInfo.ImageIdentifier.Year=2020)
Org. Chem. Front., 2020,7, 2925-2930
https://doi.org/10.1039/D0QO00968G
Photocatalytic decarboxylative [2 + 2 + 1] annulation of 1,6-enynes with N-hydroxyphthalimide esters for the synthesis of indene-containing polycyclic compounds
An efficient photoredox-mediated [2 + 2 + 1] annulation of 1,6-enynes with N-hydroxyphthalimide esters was reported for the synthesis of spiro and non-spiro indene-containing polycyclic frameworks.
![Graphical abstract: Photocatalytic decarboxylative [2 + 2 + 1] annulation of 1,6-enynes with N-hydroxyphthalimide esters for the synthesis of indene-containing polycyclic compounds](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO01166H&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 3834-3838
https://doi.org/10.1039/C9QO01166H
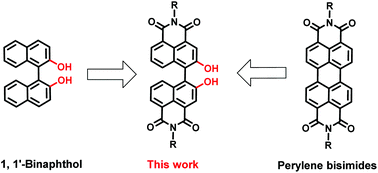
1,1′-Bi(2-naphthol-4,5-dicarboximide)s: blue emissive axially chiral scaffolds with aggregation-enhanced emission properties
Novel electron-deficient 1,1′-binaphthol derivatives bearing two dicarboximide groups at the peri-positions show intense blue luminescence and aggregation-enhanced emission characteristics.
Org. Chem. Front., 2019,6, 3731-3740
https://doi.org/10.1039/C9QO01090D
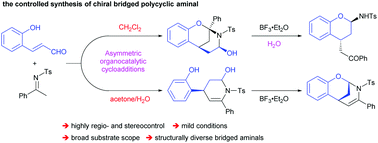
Asymmetric organocatalyzed reaction sequence of 2-hydroxy cinnamaldehydes and acyclic N-sulfonyl ketimines to construct diverse chiral bridged polycyclic aminals
2-Hydroxy cinnamaldehydes and acyclic N-sulfonyl ketimines were used as multisite substrates in an iminium catalysis triggered sequential process, leading to diverse chiral bridged polycyclic aminals in a highly regio- and stereoselective manner.
Org. Chem. Front., 2019,6, 3725-3730
https://doi.org/10.1039/C9QO01116A
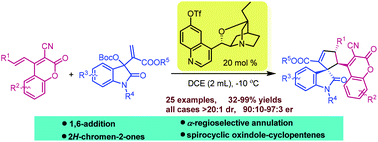
Asymmetric domino 1,6-addition/annulation reaction of 3-cyano-4-alkenyl-2H-chromen-2-ones with isatin-derived MBH carbonates: enantioselective synthesis of 3,3′-cyclopentenylspirooxindoles bearing 2H-chromen-2-ones
An asymmetric domino 1,6-addition/annulation reaction of 3-cyano-4-alkenyl-2H-chromen-2-ones with isatin-derived MBH carbonates was achieved by using a modified cinchona alkaloid as a catalyst.
Org. Chem. Front., 2019,6, 3342-3347
https://doi.org/10.1039/C9QO00890J
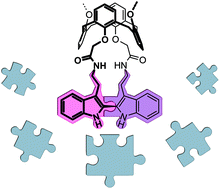
Constructing bridged multifunctional calixarenes by intramolecular indole coupling
2,2′-Bisindole bridges can be easily created at (thia)calixarene cores providing the molecules with multiple functionalities for application in supramolecular chemistry.
Org. Chem. Front., 2019,6, 3327-3341
https://doi.org/10.1039/C9QO00859D
Minisci C–H alkylation of N-heteroarenes with aliphatic alcohols via β-scission of alkoxy radical intermediates
A generally applicable method for Minisci C–H alkylation of N-heteroarenes with aliphatic alcohols via β-scission of alkoxy radical intermediates under photoredox-catalyzed conditions.
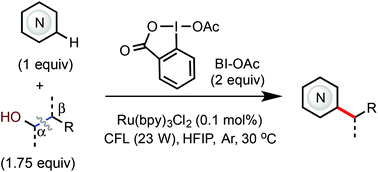
Org. Chem. Front., 2019,6, 3205-3209
https://doi.org/10.1039/C9QO00786E
A novel chiral DMAP–thiourea bifunctional catalyst catalyzed enantioselective Steglich and Black rearrangement reactions
A novel chiral DMAP–thiourea bifunctional catalyst has been prepared and applied in the highly enantioselective Steglich and Black rearrangement reactions.
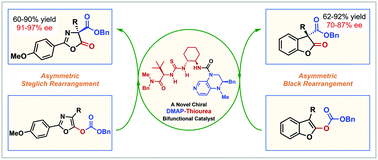
Org. Chem. Front., 2019,6, 2624-2629
https://doi.org/10.1039/C9QO00691E
Ligandless nickel-catalyzed transfer hydrogenation of alkenes and alkynes using water as the hydrogen donor
The first general route for nickel-catalyzed transfer hydrogenation reaction of alkenes and alkynes using water as the hydrogen source has been developed.
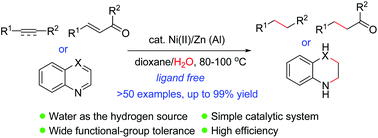
Org. Chem. Front., 2019,6, 2619-2623
https://doi.org/10.1039/C9QO00616H
Bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]−: synthesis, characterization and its application for trifluoromethylation of activated heteroaryl bromides, chlorides and iodides
The synthesis and characterization of a bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]− and its reactions with a variety of activated heteroaryl bromides, chlorides and iodides were described.
![Graphical abstract: Bistrifluoromethylated organocuprate [Ph4P]+[Cu(CF3)2]−: synthesis, characterization and its application for trifluoromethylation of activated heteroaryl bromides, chlorides and iodides](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00527G&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 2324-2328
https://doi.org/10.1039/C9QO00527G
Creation of bispiro[pyrazolone-3,3′-oxindoles] via a phosphine-catalyzed enantioselective [3 + 2] annulation of the Morita–Baylis–Hillman carbonates with pyrazoloneyldiene oxindoles
A [3 + 2] annulation between the Morita–Baylis–Hillman (MBH) carbonates and pyrazoloneyldiene oxindoles catalyzed by (S)-SITCP has been developed.
![Graphical abstract: Creation of bispiro[pyrazolone-3,3′-oxindoles] via a phosphine-catalyzed enantioselective [3 + 2] annulation of the Morita–Baylis–Hillman carbonates with pyrazoloneyldiene oxindoles](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00471H&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 2210-2214
https://doi.org/10.1039/C9QO00471H
Trifluoromethyl-substituted selenium ylide: a broadly applicable electrophilic trifluoromethylating reagent
An easily available, air/moisture insensitive and broadly applicable electrophilic reagent trifluoromethyl-substituted selenium ylide 1a and its reactions with a variety of nucleophiles including β-ketoesters and silyl enol ethers, aryl/heteroaryl boronic acids, electron-rich heteroarenes and sulfinates were described.

Org. Chem. Front., 2019,6, 2205-2209
https://doi.org/10.1039/C8QO01249K
Quantification of the hydrophobic effect using water-soluble super aryl-extended calix[4]pyrroles
We report the quantification of the hydrophobic effect using a model system based on water-soluble super aryl-extended calix[4]pyrrole receptors and a series of pyridyl N-oxide derivatives, bearing a non-polar para-substituent, as guests.
![Graphical abstract: Quantification of the hydrophobic effect using water-soluble super aryl-extended calix[4]pyrroles](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C9QO00171A&imageInfo.ImageIdentifier.Year=2019)
Org. Chem. Front., 2019,6, 1738-1748
https://doi.org/10.1039/C9QO00171A
A pore-expanded supramolecular organic framework and its enrichment of photosensitizers and catalysts for visible-light-induced hydrogen production
A 3.6 nm-pore SOF is constructed, which adsorbs both photosensitizers and polyoxometallates for visible light-induced proton reduction to produce H2.
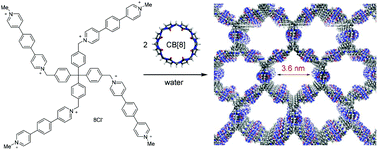
Org. Chem. Front., 2019,6, 1698-1704
https://doi.org/10.1039/C9QO00382G
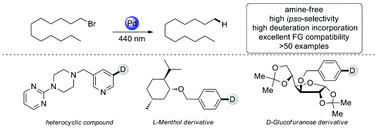
Visible-light-mediated hydrodehalogenation and Br/D exchange of inactivated aryl and alkyl halides with a palladium complex
Photo-induced radical reductive dehalogenation of inactivated aryl/alkyl bromides and chlorides with a palladium complex is described. Reductive cyclization, dehalogenative deuteration, and radical addition process can be achieved smoothly.
Org. Chem. Front., 2019,6, 1649-1654
https://doi.org/10.1039/C9QO00240E
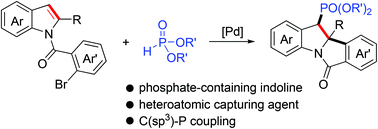
Palladium-catalyzed dearomative arylphosphorylation of indoles
A palladium-catalyzed dearomative arylphosphorylation reaction of indoles involving C–C/C–P bond formation is developed.
Org. Chem. Front., 2019,6, 1577-1580
https://doi.org/10.1039/C9QO00246D
Cooperation between an alcoholic proton and boryl species in the catalytic gem-hydrodiborylation of carboxylic esters to access 1,1-diborylalkanes
Cooperation between an alcoholic proton and boryl species in the selective gem-hydrodiborylation of carboxylic esters is described.

Org. Chem. Front., 2019,6, 900-907
https://doi.org/10.1039/C9QO00007K
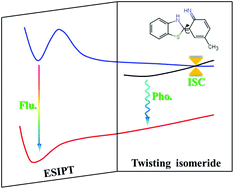
Fluorescence deactivation mechanism for a new probe detecting phosgene based on ESIPT and TICT
The ESIPT-fluorescence deactivation is caused by ISC and phosphorescence.
Org. Chem. Front., 2019,6, 597-602
https://doi.org/10.1039/C8QO01320A
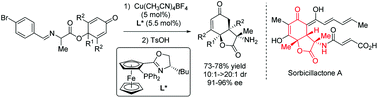
Enantioselective synthesis of 3-amino-hydrobenzofuran-2,5-diones via Cu(I)-catalyzed intramolecular conjugate addition of imino esters
The copper-catalyzed enantioselective intramolecular conjugate addition of imino esters for desymmetrization of cyclohexadienones was described, providing access to enantioenriched 3-amino-hydrobenzofuran-2,5-diones.
Org. Chem. Front., 2019,6, 579-583
https://doi.org/10.1039/C8QO01335G
About this collection
Read our on-going collection of the hottest research published as advanced article from Organic Chemistry Frontiers in 2019. These articles are recommended by reviewers as being of significant novelty and interest. Congratulations to all the authors whose articles are featured!