Themed collection Peptide-membrane interactions

List of participants
Faraday Discuss., 2021,232, 497-499
https://doi.org/10.1039/D1FD90070F
Poster list
Faraday Discuss., 2021,232, 494-496
https://doi.org/10.1039/D1FD90069B
Concluding remarks: peptide–membrane interactions
This article is based on the concluding remarks lecture given at the Faraday Discussion meeting on peptide–membrane interactions, held online, 8–10th September 2021. (Figure reproduced from Kratochvil et al., DOI: 10.1039/D1FD00061F).
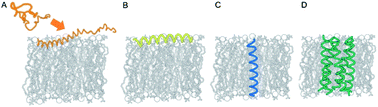
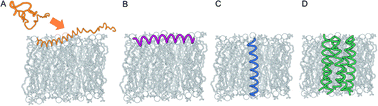
Faraday Discuss., 2021,232, 482-493
https://doi.org/10.1039/D1FD00077B
Spiers Memorial Lecture: Analysis and de novo design of membrane-interactive peptides
Membrane–peptide interactions play critical roles in many cellular and organismal functions, including protection from infection, remodeling of membranes, signaling, and ion transport.
Faraday Discuss., 2021,232, 9-48
https://doi.org/10.1039/D1FD00061F
Heterogeneity of cell membrane structure studied by single molecule tracking
We use total internal reflection fluorescence microscopy combined with super-resolution tracking of multiple individual molecules, in order to create high-resolution maps of local membrane viscosity.

Faraday Discuss., 2021,232, 358-374
https://doi.org/10.1039/D1FD00035G
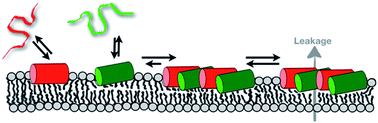
Antimicrobial peptides: mechanism of action and lipid-mediated synergistic interactions within membranes
Biophysical and structural studies of peptide–lipid interactions, peptide topology and dynamics have changed our view of how antimicrobial peptides insert and interact with membranes.
Faraday Discuss., 2021,232, 419-434
https://doi.org/10.1039/D0FD00041H
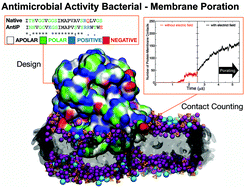
Virus-inspired designs of antimicrobial nanocapsules
We propose a novel approach to antimicrobial drug design that exploits the assembly of antimicrobial peptidic units in nanocapsules that can penetrate and rupture the bacterial membrane.
Faraday Discuss., 2021,232, 448-462
https://doi.org/10.1039/D1FD00041A
Peptide lipidation in lysophospholipid micelles and lysophospholipid-enriched membranes
Acyl transfer to a membrane-associated peptide from both lysolipids and lipids occurs readily, leading to the generation of a lipidated peptide that adopts secondary structure in the absence of lipids.
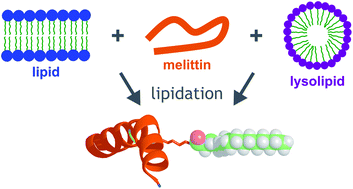
Faraday Discuss., 2021,232, 282-294
https://doi.org/10.1039/D1FD00030F
Interactions of polymyxin B with lipopolysaccharide-containing membranes
Polymyxin B uses bacterial LPS as docking receptor to cross the outer membrane.
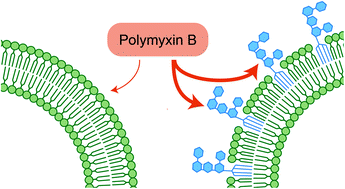
Faraday Discuss., 2021,232, 317-329
https://doi.org/10.1039/D1FD00036E
Antimicrobial peptide activity in asymmetric bacterial membrane mimics
We report on the response of asymmetric lipid membranes composed of palmitoyl oleoyl phosphatidylethanolamine and palmitoyl oleoyl phosphatidylglycerol, to interactions with the frog peptides L18W-PGLa and magainin 2 (MG2a), as well as the lactoferricin derivative LF11-215.

Faraday Discuss., 2021,232, 435-447
https://doi.org/10.1039/D1FD00039J
Common principles of surface deformation in biology
Living organisms are separated from their environment by an interface/surface. Herein I discuss principles by which living surfaces deform.
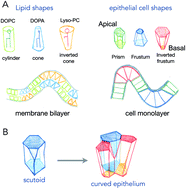
Faraday Discuss., 2021,232, 347-357
https://doi.org/10.1039/D1FD00040C
Molecular interactions of the M and E integral membrane proteins of SARS-CoV-2
Microsecond molecular dynamics simulations provide valuable insights into the aggregation patterns and membrane response around the M and E proteins of SARS-CoV-2. This work highlights the complex lipid–protein interactions during early viral assembly.
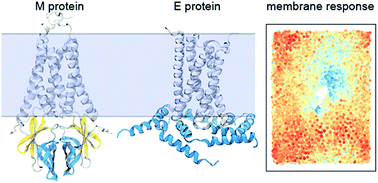
Faraday Discuss., 2021,232, 49-67
https://doi.org/10.1039/D1FD00031D
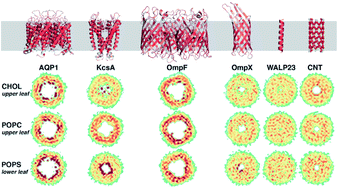
Lipid distributions and transleaflet cholesterol migration near heterogeneous surfaces in asymmetric bilayers
Varied lipid localization and cholesterol flipping dynamics were observed around different membrane-embedded entities, suggesting that unique lipid environments may be recruited by specific proteins.
Faraday Discuss., 2021,232, 103-113
https://doi.org/10.1039/D1FD00003A
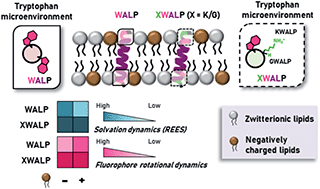
Membrane electrostatics sensed by tryptophan anchors in hydrophobic model peptides depends on non-aromatic interfacial amino acids: implications in hydrophobic mismatch
We investigate the contribution of membrane interfacial electrostatics in tryptophan-mediated responses of membrane proteins to hydrophobic mismatch.
Faraday Discuss., 2021,232, 330-346
https://doi.org/10.1039/D0FD00065E
Impact of antimicrobial peptides on E. coli-mimicking lipid model membranes: correlating structural and dynamic effects using scattering methods
Using X-rays and neutrons we address the effect of AMPs on structure and dynamics of lipids in bacterial model membranes.
Faraday Discuss., 2021,232, 203-217
https://doi.org/10.1039/D0FD00046A
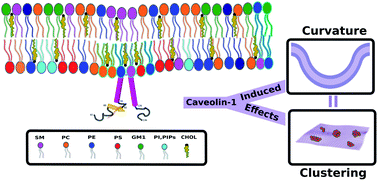
Caveolin induced membrane curvature and lipid clustering: two sides of the same coin?
Interplay between lipid clustering and curvature in plasma membrane mediated by caveolin-1: the direct and indirect effects.
Faraday Discuss., 2021,232, 218-235
https://doi.org/10.1039/D0FD00062K
Order-disorder transitions of cytoplasmic N-termini in the mechanisms of P-type ATPases
Circular dichroism of an H+, K+-ATPase N-terminal peptide at varying trifluoroethanol concentrations is investigated, indicating that its secondary structure is environmentally sensitive.
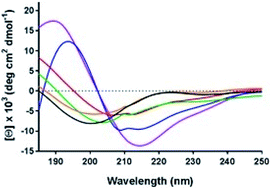
Faraday Discuss., 2021,232, 172-187
https://doi.org/10.1039/D0FD00040J
Estimating the accuracy of the MARTINI model towards the investigation of peripheral protein–membrane interactions
In this article, we investigate the ability of the MARTINI CG force field, specifically the 3 open-beta version, to reproduce known experimental observations regarding the membrane binding behavior of 12 peripheral membrane proteins and peptides.
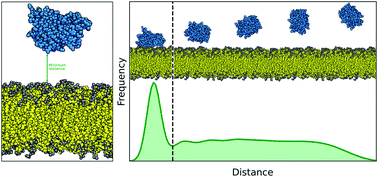
Faraday Discuss., 2021,232, 131-148
https://doi.org/10.1039/D0FD00058B
Controllable membrane remodeling by a modified fragment of the apoptotic protein Bax
We strategically introduced glutamic acid residues into a short sequence of the Bax protein that constitutively creates membrane pores. The resulting BaxE5 achieves acidity-triggered membrane remodeling.
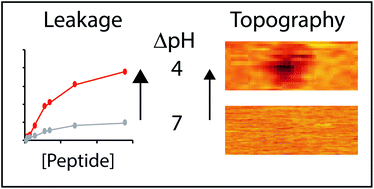
Faraday Discuss., 2021,232, 114-130
https://doi.org/10.1039/D0FD00070A
Lipid specificity of the immune effector perforin
Physical membrane properties play a determining role in defining the sensitivity of membranes to the immune effector perforin.
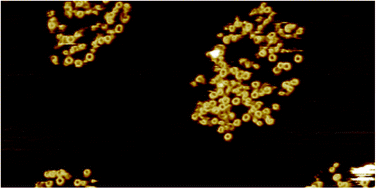
Faraday Discuss., 2021,232, 236-255
https://doi.org/10.1039/D0FD00043D
The influence of phosphatidylserine localisation and lipid phase on membrane remodelling by the ESCRT-II/ESCRT-III complex
ESCRT-II/III machinery generates intraluminal vesicles with liquid ordered membranes but requires liquid disordered membrane in the bud neck for efficiency.
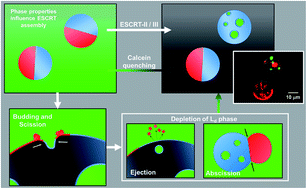
Faraday Discuss., 2021,232, 188-202
https://doi.org/10.1039/D0FD00042F
Bcl-xL inhibits tBid and Bax via distinct mechanisms
Quantification of interactions and localization dynamics of Bcl-xL with tBid and Bax reveals differences in the mechanism of inhibition.

Faraday Discuss., 2021,232, 86-102
https://doi.org/10.1039/D0FD00045K
Modulation of a host’s cell membrane nano-environment by mycobacterial glycolipids: involvement of PI(4,5)P2 signaling lipid?
Structurally diverse mycobacterial lipids distinctly alter a host’s PI(4,5)P2 membrane organization and co-localization with actin, impacting the plasma membrane–cytoskeletal interactions.
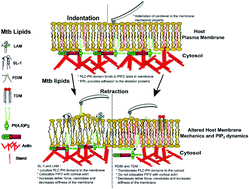
Faraday Discuss., 2021,232, 295-316
https://doi.org/10.1039/D0FD00051E
The impact of antibacterial peptides on bacterial lipid membranes depends on stage of growth
Impact of maculatin 1.1 on supported lipid bilayers (SLBs) derived from early growth phase (EGP) or stationary growth phase (SGP) E. coli lipid extracts, monitored by atomic force microscopy which images bilayer morphology in real time.

Faraday Discuss., 2021,232, 399-418
https://doi.org/10.1039/D0FD00052C
Structural changes in the model of the outer cell membrane of Gram-negative bacteria interacting with melittin: an in situ spectroelectrochemical study
Molecular scale changes in Gram-negative bacteria model membranes exposed to physiological electric fields and interacting with melittin antimicrobial peptide are discussed.
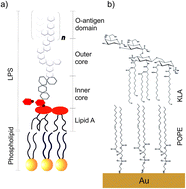
Faraday Discuss., 2021,232, 68-85
https://doi.org/10.1039/D0FD00039F
Peptide–membrane interactions and biotechnology; enabling next-generation synthetic biology: general discussion
Faraday Discuss., 2021,232, 463-481
https://doi.org/10.1039/D1FD90068D
Behaviour and interactions of proteins and peptides with and within membranes; from simple models to cellular membranes: general discussion
Faraday Discuss., 2021,232, 375-398
https://doi.org/10.1039/D1FD90067F
Theoretical and experimental studies of complex peptide–membrane systems: general discussion
Faraday Discuss., 2021,232, 256-281
https://doi.org/10.1039/D1FD90066H
Theoretical and experimental comparisons of simple peptide–membrane systems; towards defining the reaction space: general discussion
Faraday Discuss., 2021,232, 149-171
https://doi.org/10.1039/D1FD90065J
About this collection
We are delighted to share with you a selection of the papers associated with a Faraday Discussion on Peptide-membrane interactions. More information about the event may be found here: http://rsc.li/peptide-fd2020. Additional articles will be added to the collection as they are published. The final versions of all the articles presented and a record of the live discussions will be published after the event.
It is difficult to overstate the importance of improving our understanding of how macromolecules such as peptides interact with membranes as this is such a fundamental aspect of how living systems operate. These processes are involved in protein folding, cell signalling, biogenesis, morphogenesis, disease and medical therapy. Next-generation synthetic biology goals will require a clearer understanding of how to control reticulated membrane structures in order to fabricate the supramolecular structures necessary for advanced synthesis and behaviour.
The molecular questions that require clarification and their solutions bridge the many divides between the fundamental physical chemistry of macromolecular polyelectrolytes and the supramolecular chemistry of membrane assemblies related to cell biology as well as drug development and synthetic biology. This Discussion meeting will seek to establish a sound platform for further developments in this important field by addressing several related aspects of peptide interactions with membranes. It will consider model theoretical and experimental systems in order to define the ‘reaction space’ that is possible and where appropriate with relevance to fundamental questions in cell biology including how peptides and proteins behave within biological membranes