Themed collection Frustrated Lewis Pairs

Front cover

Inside front cover

Contents list
Nothing frustrating about “Frustrated Lewis pairs”
In the six short years following our initial report, the areas impacted by the chemistry of “frustrated Lewis pairs” (FLPs) continue to grow most dramatically. The collection of papers presented in this issue of Dalton Transactions reflect the broadening impact and excitement of the new field of FLP chemistry.

Dalton Trans., 2012,41, 9015-9015
https://doi.org/10.1039/C2DT90093A
Hydrogen activation by 2-boryl-N,N-dialkylanilines: a revision of Piers’ ansa-aminoborane
Two 2-[bis(pentafluorophenyl)boryl]-N,N-dialkylanilines, exemplifying a new class of intramolecular frustrated B/N Lewis pairs, can activate hydrogen at near ambient conditions; besides, one of them can hydrogenate imines and enamines in a catalytic fashion.
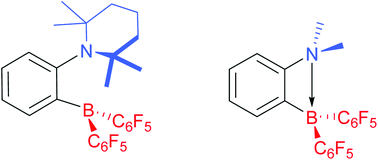
Dalton Trans., 2012,41, 9029-9032
https://doi.org/10.1039/C2DT30926B
Asymmetric hydrogenation of imines with a recyclable chiral frustrated Lewis pair catalyst
A camphor based chiral phosphonium hydrido borate zwitterion was synthesised and successfully applied in the enantioselective hydrogenation of imines with selectivities up to 76% ee. The high stability of the novel chiral FLP-system enables effective recycling of the metal-free catalyst.
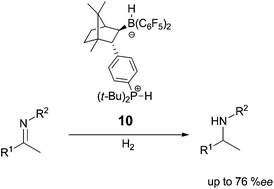
Dalton Trans., 2012,41, 9026-9028
https://doi.org/10.1039/C2DT30536D
Novel H2 activation by a tris[3,5-bis(trifluoromethyl)phenyl]borane frustrated Lewis pair
Tris[3,5-bis(trifluoromethyl)phenyl]borane (BArF18) forms a Frustrated Lewis pair with 2,2,6,6-tetramethylpiperidine, which cleaves H2 to provide the novel bridging borohydride [μ-H(BArF18)2]−.
![Graphical abstract: Novel H2 activation by a tris[3,5-bis(trifluoromethyl)phenyl]borane frustrated Lewis pair](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C2DT30384A&imageInfo.ImageIdentifier.Year=2012)
Dalton Trans., 2012,41, 9019-9022
https://doi.org/10.1039/C2DT30384A
Association of frustrated phosphine–borane pairs in toluene: molecular dynamics simulations
Explicit solvent molecular dynamics simulations of the (tBu)3P/B(C6F5)3 pair in toluene allowed the estimation of the degree of intermolecular association and the population of encounter complex states in solution phase.
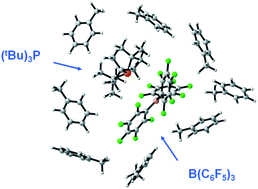
Dalton Trans., 2012,41, 9023-9025
https://doi.org/10.1039/C2DT30370A
Exchange chemistry of tBu3P(CO2)B(C6F5)2Cl
Exchange reactions of tBu3P(CO2)B(C6F5)2Cl afford tBu3P(CO2)B(C6F5)2(OSO2CF3), tBu3P(CO2)Al(C6F5)3 and [tBu3P(CO2)TiCp2Cl][B(C6F5)4].
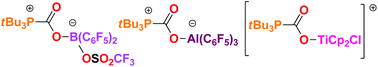
Dalton Trans., 2012,41, 9016-9018
https://doi.org/10.1039/C2DT30206C
Lewis pair polymerization by classical and frustrated Lewis pairs: acid, base and monomer scope and polymerization mechanism
Al(C6F5)3-based classical and frustrated Lewis pairs exhibit exceptional activity in Lewis pair polymerization of conjugated polar alkenes and renewable methylene butyrolactones.
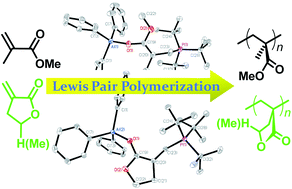
Dalton Trans., 2012,41, 9119-9134
https://doi.org/10.1039/C2DT30427A
Intramolecular Lewis acid–base pairs based on 4-ethynyl-2,6-lutidine
The reaction of 4-ethynyl-2,6-lutidine, (2,6-Me2)(4-HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)C5H2N, with the Lewis acid B(C6F5)3 leads to the zwitterion [(2,6-Me2)(4-(C6F5)3BC
C)C5H2N, with the Lewis acid B(C6F5)3 leads to the zwitterion [(2,6-Me2)(4-(C6F5)3BC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)-C5H2NH], whereas its reactions with MR3 (M = Al, Ga, In; R = Me, Et) lead exclusively to metallation of the ethynyl group and the products [(2,6-Me2)(R2MC
C)-C5H2NH], whereas its reactions with MR3 (M = Al, Ga, In; R = Me, Et) lead exclusively to metallation of the ethynyl group and the products [(2,6-Me2)(R2MC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)C5H2N].
C)C5H2N].
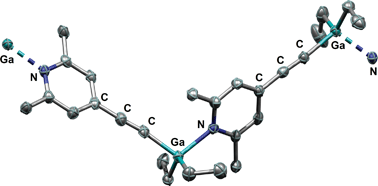
Dalton Trans., 2012,41, 9143-9150
https://doi.org/10.1039/C2DT30719G
Heterolytic addition of E–H bonds across Pt–P bonds in Pt N-heterocyclic phosphenium/phosphido complexes
The reactivity of E–H bonds (E = S, O, Cl) with Pt(II) complexes ligated by an N-heterocyclic phosphido-containing diphosphine ligand have been investigated.
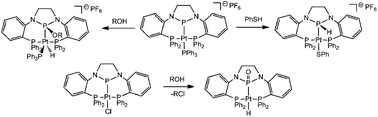
Dalton Trans., 2012,41, 9083-9090
https://doi.org/10.1039/C2DT30455D
Theoretical analysis of cooperative effects of small molecule activation by frustrated Lewis pairs
The cooperative nature of small molecule activation with FLPs is analysed in terms of many-body contributions to energy and deformation density.
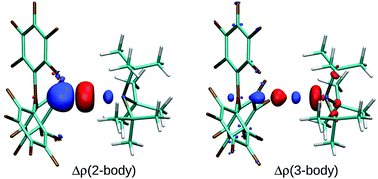
Dalton Trans., 2012,41, 9111-9118
https://doi.org/10.1039/C2DT30562C
Synthesis and reactivity of electron poor allenes: formation of completely organic frustrated Lewis pairs
The synthesis of several electron poor allenes is described and their application as Lewis acids for the formation of organic frustrated Lewis pairs evaluated.
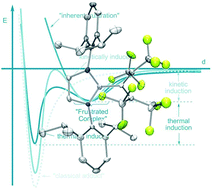
Dalton Trans., 2012,41, 9073-9082
https://doi.org/10.1039/C2DT30195D
Frustrated Lewis pair addition to conjugated diynes: Formation of zwitterionic 1,2,3-butatriene derivatives
The frustrated Lewis pair B(C6F5)3/PtBu3 reacts with 2,4-hexadiyne to give mixture of the 1,2- and 1,4-addition products.
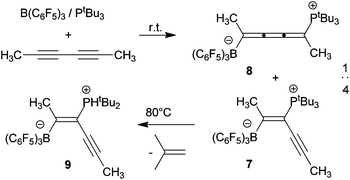
Dalton Trans., 2012,41, 9135-9142
https://doi.org/10.1039/C2DT30321C
Catalytic metal-free intramolecular hydroaminations of non-activated aminoalkenes: A computational exploration
Computations show the promise of using the previously designed catalysts to promote intramolecular hydroaminations of non-activated aminoalkenes affording nitrogen heterocycles.
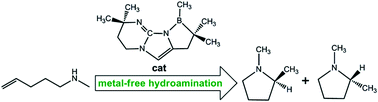
Dalton Trans., 2012,41, 9091-9100
https://doi.org/10.1039/C2DT30329A
Fixation of carbon dioxide and related small molecules by a bifunctional frustrated pyrazolylborane Lewis pair
The fixation of carbon dioxide and related small molecules is efficiently accomplished by a bifunctional frustrated pyrazolylborane Lewis pair; CO2 fixation presumably proceeds via a van der Waals complex with a low energy barrier of approximately 7.2 kcal mol−1.

Dalton Trans., 2012,41, 9101-9110
https://doi.org/10.1039/C2DT30448A
Computational studies of complexation of nitrous oxide by borane–phosphine frustrated Lewis pairs
Several interesting facets of FLP–N2O complexes were investigated computationally, including the preference for the BONNP core and the trans–trans structure, borane exchange reactions, and possibilities for N2O derivatization.
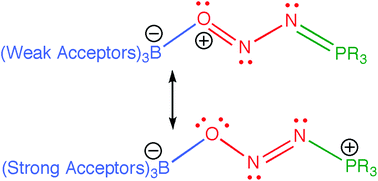
Dalton Trans., 2012,41, 9046-9055
https://doi.org/10.1039/C2DT30208J
Heterolytic activation of hydrogen using frustrated Lewis pairs containing tris(2,2′,2′′-perfluorobiphenyl)borane
The bulky borane tris(2,2,2-perfluorobiphenyl)borane is crystallographically characterised, and is used with nitrogen based Lewis bases to synthesize novel ‘frustrated Lewis pairs’ that activate dihydrogen.
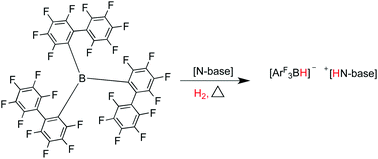
Dalton Trans., 2012,41, 9061-9066
https://doi.org/10.1039/C2DT30334E
[2.2]Paracyclophane derived bisphosphines for the activation of hydrogen by FLPs: application in domino hydrosilylation/hydrogenation of enones
The heterolytic splitting of hydrogen by two types of [2.2]paracyclophane derived bisphosphines (1, 2a and 2b) in combination with tris(pentafluorophenyl)borane (3) at room temperature is described.
![Graphical abstract: [2.2]Paracyclophane derived bisphosphines for the activation of hydrogen by FLPs: application in domino hydrosilylation/hydrogenation of enones](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C2DT30374D&imageInfo.ImageIdentifier.Year=2012)
Dalton Trans., 2012,41, 9056-9060
https://doi.org/10.1039/C2DT30374D
Cationic Ti(IV) and neutral Ti(III) titanocene–phosphinoaryloxide frustrated Lewis pairs: hydrogen activation and catalytic amine-borane dehydrogenation
Titanium–phosphorus frustrated Lewis pairs (FLPs) based on titanocene–phosphinoaryloxide complexes have been studied..
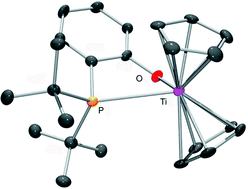
Dalton Trans., 2012,41, 9067-9072
https://doi.org/10.1039/C2DT30168G
Dimeric aluminum–phosphorus compounds as masked frustrated Lewis pairs for small molecule activation
Al–P-based dimers with coordinatively saturated Al and P atoms act as masked frustrated Lewis pairs by dipolar activation of CO2 and phenyl isocyanate.
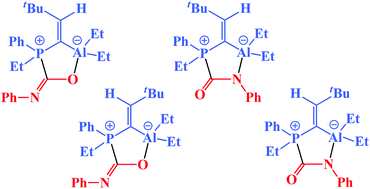
Dalton Trans., 2012,41, 9033-9045
https://doi.org/10.1039/C2DT30080J
About this collection
This themed issue reflects the broadening impact and excitement of the new field of FLP chemistry.