Dimeric aluminum–phosphorus compounds as masked frustrated Lewis pairs for small molecule activation†
Abstract
Hydroalumination of aryldialkynylphosphines RP(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–tBu)2 (R = Ph, Mes) with equimolar quantities of diethylaluminum hydride afforded mixed alkenyl–alkynyl cyclic dimers in which the dative aluminum–phosphorus bonds are geminal to the exocyclic alkenyl groups. Addition of triethylaluminum to isolated 1 (R = Ph) or to the in situ generated species (R = Mes) caused diethylaluminum ethynide elimination to yield the arylethylphosphorus dimers 2 and 3. These possess a chair-like Al2C2P2 heterocycle with intermolecular Al–P interactions. The boat conformation (4) was obtained by the reaction of tBu–P(C
C–tBu)2 (R = Ph, Mes) with equimolar quantities of diethylaluminum hydride afforded mixed alkenyl–alkynyl cyclic dimers in which the dative aluminum–phosphorus bonds are geminal to the exocyclic alkenyl groups. Addition of triethylaluminum to isolated 1 (R = Ph) or to the in situ generated species (R = Mes) caused diethylaluminum ethynide elimination to yield the arylethylphosphorus dimers 2 and 3. These possess a chair-like Al2C2P2 heterocycle with intermolecular Al–P interactions. The boat conformation (4) was obtained by the reaction of tBu–P(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–tBu)2 with di(tert-butyl)aluminum hydride. Despite being dimeric, 2 behaves as a frustrated Lewis pair and activates small molecules. The reaction with carbon dioxide gave cis/trans isomeric AlPC2O heterocycles that differ only by the configuration of the exocyclic alkenyl unit. Four isomers resulted from the reaction with phenyl isocyanate. This is caused by cis/trans isomerization of the initial C
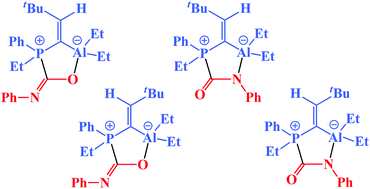
C–tBu)2 with di(tert-butyl)aluminum hydride. Despite being dimeric, 2 behaves as a frustrated Lewis pair and activates small molecules. The reaction with carbon dioxide gave cis/trans isomeric AlPC2O heterocycles that differ only by the configuration of the exocyclic alkenyl unit. Four isomers resulted from the reaction with phenyl isocyanate. This is caused by cis/trans isomerization of the initial C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O adduct and subsequent rearrangement to the AlPC2N heterocycle, being the C
O adduct and subsequent rearrangement to the AlPC2N heterocycle, being the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N adduct.
N adduct.
- This article is part of the themed collection: Frustrated Lewis Pairs

 Please wait while we load your content...
Please wait while we load your content...