Themed collection Astrochemistry

Astrochemistry
This themed issue presents work from leading computational and experimental scientists, astronomers and chemists that address the processes important in the chemical evolution of the universe.

Phys. Chem. Chem. Phys., 2014,16, 3343-3343
https://doi.org/10.1039/C4CP90004A
Three milieux for interstellar chemistry: gas, dust, and ice
Chemistry occurs on bare dust particle cores and on the ice mantles surrounding them via a variety of mechanisms.
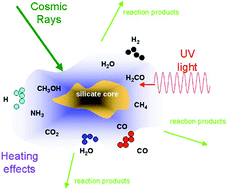
Phys. Chem. Chem. Phys., 2014,16, 3344-3359
https://doi.org/10.1039/C3CP54065K
Atomic oxygen diffusion on and desorption from amorphous silicate surfaces
Surface reactions involving atomic oxygen have attracted much attention in astrophysics and astrochemistry, but two of the most fundamental surface processes, desorption and diffusion, are not well understood.
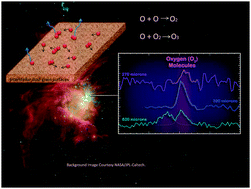
Phys. Chem. Chem. Phys., 2014,16, 3493-3500
https://doi.org/10.1039/C3CP54328E
A combined experimental and theoretical study of reactions between the hydroxyl radical and oxygenated hydrocarbons relevant to astrochemical environments
Rate coefficients for the reactions of the hydroxyl radical with acetone and dimethyl ether increase dramatically at very low temperatures.
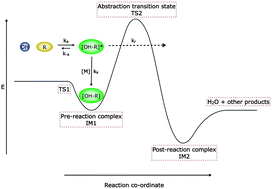
Phys. Chem. Chem. Phys., 2014,16, 3466-3478
https://doi.org/10.1039/C3CP54664K
The interaction of carbon monoxide with model astrophysical surfaces
The vibrational frequencies of CO adsorbed on astrophysically relevant ice films are measured.
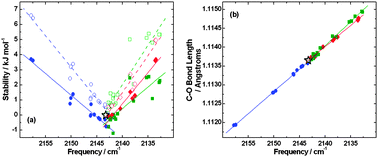
Phys. Chem. Chem. Phys., 2014,16, 3479-3492
https://doi.org/10.1039/C3CP54024C
The structure and dynamics of carbon dioxide and water containing ices investigated via THz and mid-IR spectroscopy
Using time-domain THz and FTIR spectroscopy, water and carbon dioxide containing ices are studied. THz spectroscopy is needed to understand the structure and dynamics of astrochemical ices.

Phys. Chem. Chem. Phys., 2014,16, 3442-3455
https://doi.org/10.1039/C3CP53767F
Porosity and thermal collapse measurements of H2O, CH3OH, CO2, and H2O:CO2 ices
Quantitative results for ice porosity and thermal collapse show that (i) H2O ice is not fully compacted after annealing, and (ii) ice segregation efficiency is determined by the initial ice porosity.

Phys. Chem. Chem. Phys., 2014,16, 3456-3465
https://doi.org/10.1039/C3CP54481H
Thermal and energetic processing of ammonia and carbon dioxide bearing solid mixtures
We present new experimental results on thermal and ion irradiation processing of frozen ammonia–carbon dioxide mixtures.
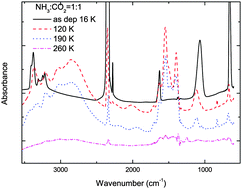
Phys. Chem. Chem. Phys., 2014,16, 3433-3441
https://doi.org/10.1039/C3CP54027H
Electron impact induced anion production in acetylene
Investigation of electron induced anion production (dissociative electron attachment and dipolar dissociation) in acetylene.
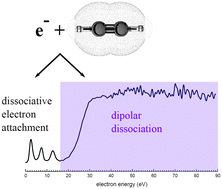
Phys. Chem. Chem. Phys., 2014,16, 3425-3432
https://doi.org/10.1039/C3CP54459A
Hydrogenation of PAH molecules through interaction with hydrogenated carbonaceous grains
Polycyclic aromatic hydrocarbons (PAHs) react with hydrogenated graphite forming superhydrogenated PAHs, potentially releasing H2.
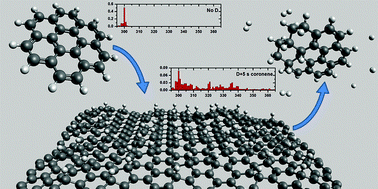
Phys. Chem. Chem. Phys., 2014,16, 3381-3387
https://doi.org/10.1039/C3CP54073A
Infrared and reflectron time-of-flight mass spectroscopic analysis of methane (CH4)–carbon monoxide (CO) ices exposed to ionization radiation – toward the formation of carbonyl-bearing molecules in extraterrestrial ices
Ice mixtures of methane and carbon monoxide were exposed to ionizing radiation in the form of energetic electrons at 5.5 K to investigate the formation of carbonyl bearing molecules in extraterrestrial ices.
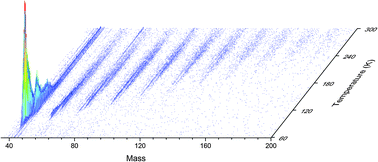
Phys. Chem. Chem. Phys., 2014,16, 3399-3424
https://doi.org/10.1039/C3CP54255F
Photochemistry of a 1 : 1 hydrogen-bonded CH3CN : HCOOH complex under astrochemically-relevant conditions
The interstellar medium is characterized by a complex chemistry, especially on the surface of ice grains in molecular clouds.
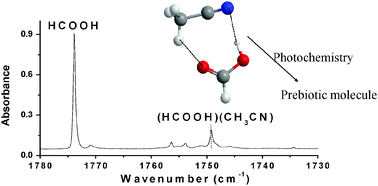
Phys. Chem. Chem. Phys., 2014,16, 3388-3398
https://doi.org/10.1039/C3CP54041C
Formation of hydroxyacetonitrile (HOCH2CN) and polyoxymethylene (POM)-derivatives in comets from formaldehyde (CH2O) and hydrogen cyanide (HCN) activated by water
Water as a catalyst for cometary ice reactivity: the case of CH2O and HCN.
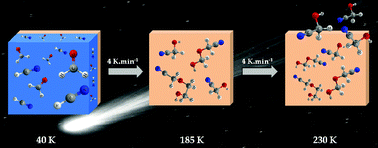
Phys. Chem. Chem. Phys., 2014,16, 3360-3370
https://doi.org/10.1039/C3CP54034K
The formation of carbamate ions in interstellar ice analogues
Solid phase reactions of NH3 and CO2 occurring in interstellar environments can induce the formation of carbamate ions.
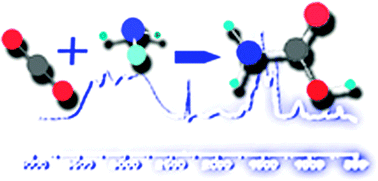
Phys. Chem. Chem. Phys., 2014,16, 3371-3380
https://doi.org/10.1039/C3CP53153H
About this collection
A collection of articles on the theme of astrochemistry, guest edited by Wendy Brown (University of Sussex).