Themed collection Bunsentagung 2018: Kinetics in the Real World

Kinetics in the real world: linking molecules, processes, and systems
This themed issue includes a collection of articles on Kinetics in the Real World. The image shows diagnostics of pattern-forming surface reactions using spatially resolved photoemission electron microscopy, for which the sample is illuminated with UV.

Phys. Chem. Chem. Phys., 2018,20, 10561-10568
https://doi.org/10.1039/C8CP90054J
Coupling free radical catalysis, climate change, and human health
Linking free radical catalytic loss of stratospheric ozone over the central United States to climate forcing by CO2 and CH4.
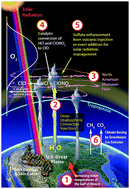
Phys. Chem. Chem. Phys., 2018,20, 10569-10587
https://doi.org/10.1039/C7CP08331A
Multi-fuel surrogate chemical kinetic mechanisms for real world applications
The most important driving force for development of detailed chemical kinetic reaction mechanisms in combustion is the desire by researchers to simulate practical systems.
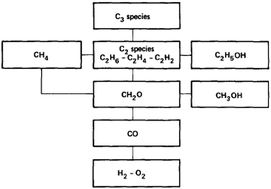
Phys. Chem. Chem. Phys., 2018,20, 10588-10606
https://doi.org/10.1039/C7CP07901J
Oxidation of substituted aromatic hydrocarbons in the tropospheric aqueous phase: kinetic mechanism development and modelling
An aqueous-phase chemistry mechanism for the oxidation of aromatic compounds in the atmosphere is developed based on available kinetic data. Detailed model studies successfully describe the oxidation and functionalization of monoaromatic compounds in the atmosphere.
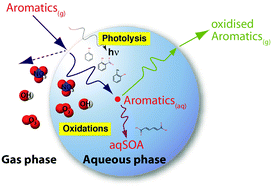
Phys. Chem. Chem. Phys., 2018,20, 10960-10977
https://doi.org/10.1039/C7CP08576A
Intramolecular vibrational energy redistribution in HCCCH2X (X = Cl, Br, I) measured by femtosecond pump–probe experiments in a hollow waveguide
Time resolved femtosecond probing of intramolecular energy flow after excitation of the two different infrared CH-chromophores in these bichromophoric molecules shows strong dependence on the chemical environment of the initial excitation.
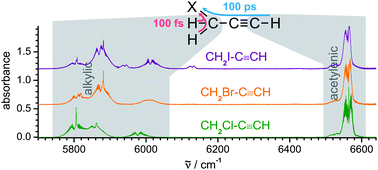
Phys. Chem. Chem. Phys., 2018,20, 10949-10959
https://doi.org/10.1039/C7CP08561C
Competition kinetics of OH radical reactions with oxygenated organic compounds in aqueous solution: rate constants and internal optical absorption effects
Atmospheric aqueous phase rate constants are determined and the effect of optical absorbers in competition kinetics is studied.
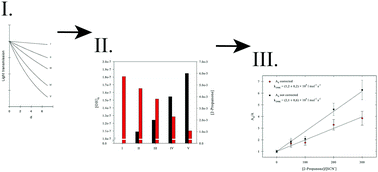
Phys. Chem. Chem. Phys., 2018,20, 10939-10948
https://doi.org/10.1039/C7CP08571K
A combined crossed molecular beams and computational study on the formation of distinct resonantly stabilized C5H3 radicals via chemically activated C5H4 and C6H6 intermediates
In a single-collision environment dicarbon reacts with 1-butyne to form resonantly stabilized C5H3 radical products.
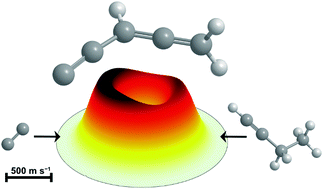
Phys. Chem. Chem. Phys., 2018,20, 10906-10925
https://doi.org/10.1039/C8CP00357B
Reactive polycyclic aromatic hydrocarbon dimerization drives soot nucleation
Nucleation is an important yet poorly understood step in soot formation. Strong chemical bonds between PAH monomers are required as physical dimerization cannot explain soot formation at high temperatures. Dimers can be considered as soot nuclei as larger oligomers have negligible contribution.
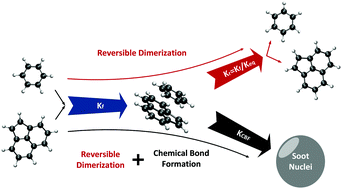
Phys. Chem. Chem. Phys., 2018,20, 10926-10938
https://doi.org/10.1039/C7CP07803J
CaRMeN: a tool for analysing and deriving kinetics in the real world
Open software that automates model vs. experiment comparisons to rapidly analyse and derive models, in particular chemical kinetics.
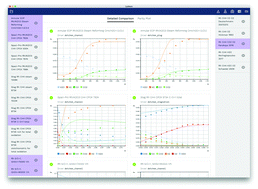
Phys. Chem. Chem. Phys., 2018,20, 10857-10876
https://doi.org/10.1039/C7CP07777G
Ab initio derived group additivity model for intramolecular hydrogen abstraction reactions
A systematic methodology to calculate reaction rate coefficients of intramolecular hydrogen abstraction reactions by group additivity has been developed.
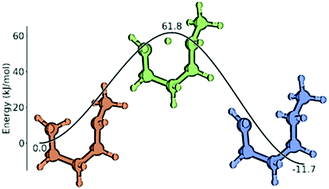
Phys. Chem. Chem. Phys., 2018,20, 10877-10894
https://doi.org/10.1039/C7CP07771H
Reaction kinetics of hydrogen atom abstraction from isopentanol by the H atom and HO2˙ radical
The formation of the transition state for the H-abstraction reaction from isopentanol by the HO2˙ radical.
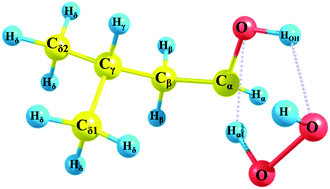
Phys. Chem. Chem. Phys., 2018,20, 10895-10905
https://doi.org/10.1039/C7CP08077H
Kinetics of the reaction of CO3˙−(H2O)n, n = 0, 1, 2, with nitric acid, a key reaction in tropospheric negative ion chemistry
One water molecule accelerates the reaction of CO3˙− with HNO3, while two water molecules quench the reactivity.
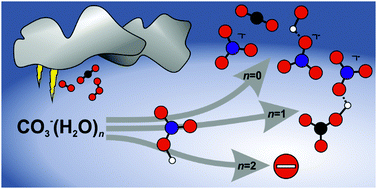
Phys. Chem. Chem. Phys., 2018,20, 10838-10845
https://doi.org/10.1039/C7CP07773D
Interfacial electrostatics of poly(vinylamine hydrochloride), poly(diallyldimethylammonium chloride), poly-L-lysine, and poly-L-arginine interacting with lipid bilayers
Charge densities of cationic polymers adsorbed to lipid bilayers are estimated from SHG spectroscopy and QCM-D measurements.
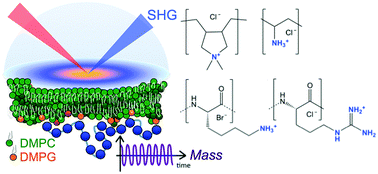
Phys. Chem. Chem. Phys., 2018,20, 10846-10856
https://doi.org/10.1039/C7CP07353D
High temperature pyrolysis of 2-methyl furan
Experiments and theory reveal the complex dissociation of 2-methylfuran and the surprising importance of H-atom loss.
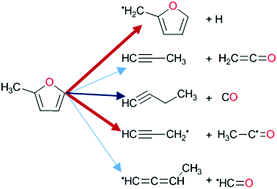
Phys. Chem. Chem. Phys., 2018,20, 10826-10837
https://doi.org/10.1039/C7CP07775K
Experimental and kinetic modeling investigation of rich premixed toluene flames doped with n-butanol
Blending of n-butanol for rich toluene combustion strongly suppresses the formation of PAHs.
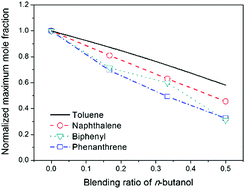
Phys. Chem. Chem. Phys., 2018,20, 10628-10636
https://doi.org/10.1039/C7CP08518D
Direct measurement of ˙OH and HO2˙ formation in ˙R + O2 reactions of cyclohexane and tetrahydropyran
A comparison of two fuels demonstrates how analysis of ˙OH and HO2˙ formation kinetics via the eigenvalues of a system of simplified kinetic equations can give mechanistic insights.

Phys. Chem. Chem. Phys., 2018,20, 10815-10825
https://doi.org/10.1039/C7CP08164B
Influences of the molecular fuel structure on combustion reactions towards soot precursors in selected alkane and alkene flames
Flame-sampling molecular beam mass spectrometry enables valuable insights into the fuel-structure-dependent formation kinetics of polycyclic aromatic hydrocarbons and their precursors.
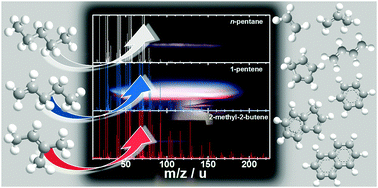
Phys. Chem. Chem. Phys., 2018,20, 10780-10795
https://doi.org/10.1039/C7CP07743B
Ab initio kinetic Monte Carlo simulation of seeded emulsion polymerizations of styrene
A kinetic Monte Carlo model for emulsion polymerizations based on elemental reactions and radical transfer into particles is introduced.
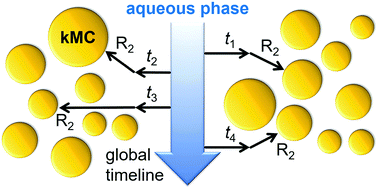
Phys. Chem. Chem. Phys., 2018,20, 10796-10805
https://doi.org/10.1039/C7CP07768H
Theoretical kinetic study of the formic acid catalyzed Criegee intermediate isomerization: multistructural anharmonicity and atmospheric implications
We performed a theoretical study on the double hydrogen shift isomerization reaction of a six carbon atom Criegee intermediate (C6-CI), catalyzed by formic acid (HCOOH), to produce vinylhydroperoxide (VHP), C6-CI + HCOOH → VHP + HCOOH.
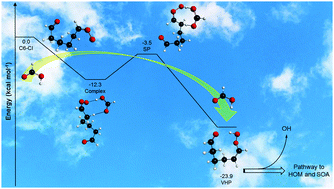
Phys. Chem. Chem. Phys., 2018,20, 10806-10814
https://doi.org/10.1039/C7CP08538A
H-Abstraction reactions by OH, HO2, O, O2 and benzyl radical addition to O2 and their implications for kinetic modelling of toluene oxidation
Theoretical study of relevant reactions in toluene oxidation.
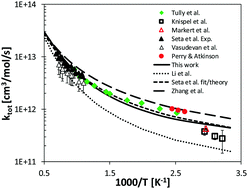
Phys. Chem. Chem. Phys., 2018,20, 10607-10627
https://doi.org/10.1039/C7CP07779C
In situ synchrotron XRD analysis of the kinetics of spodumene phase transitions
A kinetic model for the decomposition of α-spodumene and γ-spodumene is derived using in situ XRD to monitor the transitions.
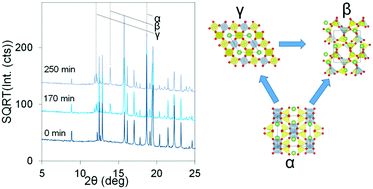
Phys. Chem. Chem. Phys., 2018,20, 10753-10761
https://doi.org/10.1039/C7CP07754H
Ab initio calculations and kinetic modeling of thermal conversion of methyl chloride: implications for gasification of biomass
Ab initio theory is used to obtain rate coefficients for key reactions in the CH3Cl subset; kinetic modeling is then used for analysis of practical application.
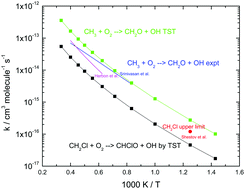
Phys. Chem. Chem. Phys., 2018,20, 10741-10752
https://doi.org/10.1039/C7CP07552A
Kinetics of autoignition: a simple intuitive interpretation and its relation to the Livengood–Wu integral
A simple intuitive interpretation of autoignition.
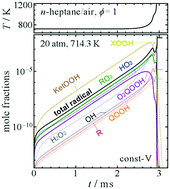
Phys. Chem. Chem. Phys., 2018,20, 10762-10769
https://doi.org/10.1039/C7CP07736J
Global quasi-linearization (GQL) versus QSSA for a hydrogen–air auto-ignition problem
A recently developed automatic reduction method for systems of chemical kinetics, the so-called Global Quasi-Linearization (GQL) method, has been implemented to study and reduce the dimensions of a homogeneous combustion system.
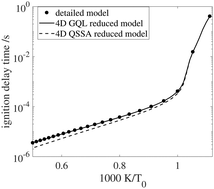
Phys. Chem. Chem. Phys., 2018,20, 10770-10779
https://doi.org/10.1039/C7CP07213A
Kinetics of the a-C3H5 + O2 reaction, investigated by photoionization using synchrotron radiation
The kinetics of the allyl + O2 reaction has been studied in a flow tube reactor, using VUV synchrotron radiation for product detection.
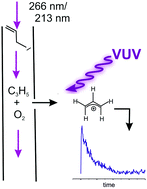
Phys. Chem. Chem. Phys., 2018,20, 10721-10731
https://doi.org/10.1039/C7CP07893E
Ultrafast stimulated emission of nitrophenolates in organic and aqueous solutions
Early-time dynamics of nitroaromatics and its corresponding bases can give valuable insights into photo-induced reactions relevant to atmospheric and environmental processes.
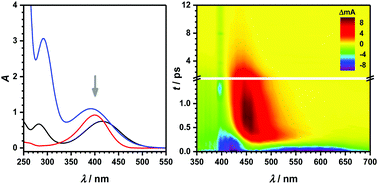
Phys. Chem. Chem. Phys., 2018,20, 10713-10720
https://doi.org/10.1039/C7CP07774B
Barriometry – an enhanced database of accurate barrier heights for gas-phase reactions
The kinetics of many reactions are critically dependent upon the barrier heights for which accurate determination can be difficult. More than 100 accurate barriers are obtained with the high-level W3X-L composite procedure.

Phys. Chem. Chem. Phys., 2018,20, 10732-10740
https://doi.org/10.1039/C7CP08045J
Shock-tube study of the decomposition of tetramethylsilane using gas chromatography and high-repetition-rate time-of-flight mass spectrometry
The decomposition of tetramethylsilane was studied in shock-tube experiments in a temperature range of 1270–1580 K and pressures ranging from 1.5 to 2.3 bar behind reflected shock waves combining GC/MS and HRR-TOF-MS.
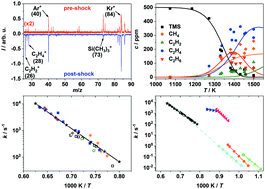
Phys. Chem. Chem. Phys., 2018,20, 10686-10696
https://doi.org/10.1039/C7CP06827A
Modeling study of the anti-knock tendency of substituted phenols as additives: an application of the reaction mechanism generator (RMG)
This work presents kinetic modeling efforts to evaluate the anti-knock tendency of several substituted phenols if used as gasoline additives.
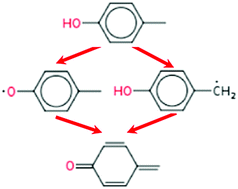
Phys. Chem. Chem. Phys., 2018,20, 10637-10649
https://doi.org/10.1039/C7CP07058F
Pronounced exciton and coherent phonon dynamics in BiI3
Long-lived coherent oscillations in transient absorption signals of BiI3 suggest strong electron–phonon coupling in this material for photovoltaics.
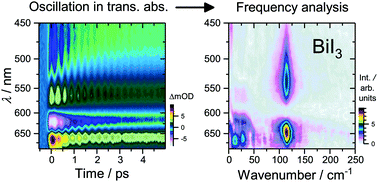
Phys. Chem. Chem. Phys., 2018,20, 10677-10685
https://doi.org/10.1039/C7CP07729G
Gas phase 1H NMR studies and kinetic modeling of dihydrogen isotope equilibration catalyzed by Ru-nanoparticles under normal conditions: dissociative vs. associative exchange
Exposure of surface H-containing Ru-nanoparticles to D2 gas produces HD via associative adsorption, surface H-transfer and associative desorption.
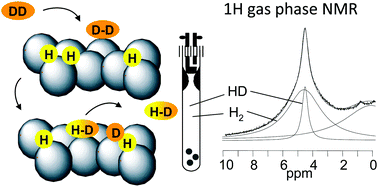
Phys. Chem. Chem. Phys., 2018,20, 10697-10712
https://doi.org/10.1039/C7CP07770J
Can electrostatic catalysis of Diels–Alder reactions be harnessed with pH-switchable charged functional groups?
Quantum-chemical calculations demonstrate that acid and base groups can be used to pH-switch regio- and diastereoselectivity in Diels–Alder reactions and provide a practical means of harnessing electrostatic catalysis.
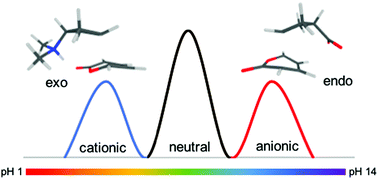
Phys. Chem. Chem. Phys., 2018,20, 10671-10676
https://doi.org/10.1039/C7CP07562F
The reaction of fluorine atoms with methanol: yield of CH3O/CH2OH and rate constant of the reactions CH3O + CH3O and CH3O + HO2
Measurement and theory of CH3O + HO2 and CH3O + CH3O reactions, product yields for F + CH3OH.
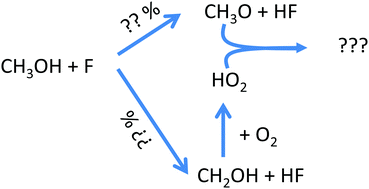
Phys. Chem. Chem. Phys., 2018,20, 10660-10670
https://doi.org/10.1039/C7CP05770A
Effect of a single water molecule on the HO2 + ClO reaction
The presence of a single water molecule retards the reaction of HO2 with ClO.
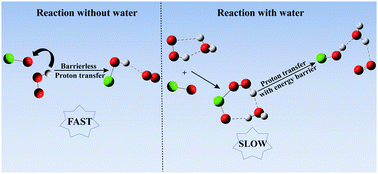
Phys. Chem. Chem. Phys., 2018,20, 10650-10659
https://doi.org/10.1039/C7CP05008A
About this collection
This is the official themed issue of the international Bunsentagung 2018 meeting organised by the Deutsche Bunsen-Gesellschaft (DBG) in Hannover, Germany on 10-12th May. The issue features high quality and original research on the theme of " Kinetics in the Real World". The issue is guest-edited by Professor Jürgen Troe (University of Göttingen), Professor Katharina Kohse-Höinghaus (University of Bielefeld), Professor Jens-Uwe Grabow (Hannover University), Professor Gernot Friedrichs (Kiel University), Professor Matthias Olzmann (Karlsruhe Institute of Technology) and
Professor Ruth Signorell (ETH Zürich and PCCP Associate Editor).