Ultrafast stimulated emission of nitrophenolates in organic and aqueous solutions†
Abstract
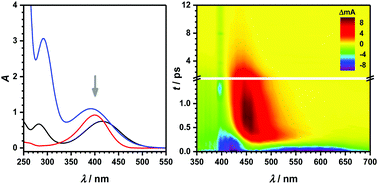
Early-time dynamics of nitroaromatics and its coressponding bases can give valuable insights into photo-induced reactions relevant to atmospheric and environmental processes. In this work, femtosecond broadband absorption spectroscopy between 350 and 700 nm has been applied to explore the ultrafast dynamics of o-, p- and m-nitrophenol anions (NP−) in basic organic and aqueous solution. Excitation at 400 nm promotes these compounds into the first bright electronic singlet state, which is a charge-transfer state. A surprising finding for all nitrophenolates was a characteristic, spectrally broad stimulated emission (SE) from the electronically excited state into the ground state. The corresponding lifetime was on the order of a few hundred femtoseconds for o- and p-NP− while it was roughly ten times larger for m-NP−. In line with earlier observations, the SE is governed by an out-of-plane torsional motion of the nitro group, leading to a close energetic approach of the relevant electronically excited singlet and ground states. Subsequent dynamics can be assigned to excited state absorption and ground state relaxation due to energy dissipation of the vibrational modes to the solvent that occur for up to several tens of picoseconds. No longer-lasting transient absorption (TA) was found; instead, a complete recovery of the ground state bleaching was observed indicating that triplet state relaxation is either not significantly involved in this spectral part or shifted to other regions. In the aqueous system, time constants for all processes are much smaller than in organic solution, a fact that can be explained by the larger dipole moment of the solvent and the correspondingly stronger intermolecular coupling between NP− and the aqueous solvent.
- This article is part of the themed collection: Bunsentagung 2018: Kinetics in the Real World


 Please wait while we load your content...
Please wait while we load your content...