Experimental and kinetic modeling investigation of rich premixed toluene flames doped with n-butanol†
Abstract
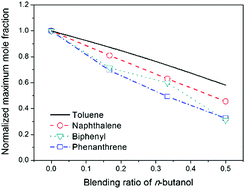
n-Butanol is a promising renewable biofuel and has a lot of advantages as a gasoline additive compared with ethanol. Though the combustion of pure n-butanol has been extensively investigated, the chemical structures of large hydrocarbons doped with n-butanol, especially for aromatic fuels, are still insufficiently understood. In this work, rich premixed toluene/n-butanol/oxygen/argon flames were investigated at 30 Torr with synchrotron vacuum ultraviolet photoionization mass spectrometry (SVUV-PIMS). The blending ratio of n-butanol was varied from 0 to 50%, while the equivalence ratio was maintained at a quite rich value (1.75) for the purpose of studying the influence of n-butanol on the aromatic growth process. Flame species including radicals, reactive molecules, isomers and polycyclic aromatic hydrocarbons (PAHs) were identified and their mole fraction profiles were measured. A kinetic model of toluene/n-butanol combustion was developed from our recently reported toluene and n-butanol models. It is observed that the production of most toluene decomposition products and larger aromatics was suppressed as the blending ratio of n-butanol increases. Meanwhile, the addition of n-butanol generally enhanced the formation of most observed C2–C4 hydrocarbons and C1–C4 oxygenated species. The rate of production (ROP) analysis and experimental observations both indicate that the interaction between toluene and n-butanol in their decomposition processes mainly occurs at the formation of small intermediates, e.g. acetylene and methyl. In particular, the interaction between toluene and n-butanol in methyl formation influences the formation of large monocyclic aromatics such as ethylbenzene, styrene and phenylacetylene, making their maximum mole fractions decay slowly upon increasing the blending ratio of n-butanol compared with toluene and benzyl. The increase of the blending ratio of n-butanol reduces the formation of key PAH precursors such as benzyl, fulvenallenyl, benzene, phenyl and propargyl, which leads to a remarkable reduction in the formation of PAHs.
- This article is part of the themed collection: Bunsentagung 2018: Kinetics in the Real World


 Please wait while we load your content...
Please wait while we load your content...