Themed collection Advances in Experimental and Regulatory Toxicology

Appreciation of Iain Purchase and Cliff Elcombe
Introducing this themed issue dedicated to the memory of Iain Purchase and Cliff Elcombe.

Toxicol. Res., 2018,7, 548-552
https://doi.org/10.1039/C8TX90008F
The association between chemical-induced porphyria and hepatic cancer
The haem biosynthetic pathway is of fundamental importance for cellular metabolism both for the erythroid and nonerythroid tissues.
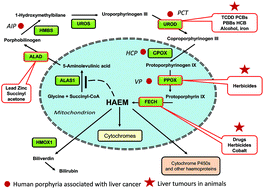
Toxicol. Res., 2018,7, 647-663
https://doi.org/10.1039/C8TX00019K
Role of xenobiotics in the induction and progression of fatty liver disease
Non-alcoholic fatty liver disease is a major cause of chronic liver pathology in humans.
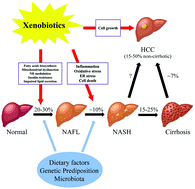
Toxicol. Res., 2018,7, 664-680
https://doi.org/10.1039/C7TX00326A
The safety evaluation of food flavouring substances: the role of metabolic studies
The safety assessment of a flavour substance examines several factors, including metabolic and physiological disposition data.
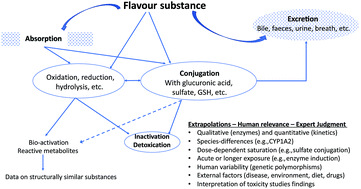
Toxicol. Res., 2018,7, 618-646
https://doi.org/10.1039/C7TX00254H
Human relevance of rodent liver tumour formation by constitutive androstane receptor (CAR) activators
A large number of nongenotoxic chemicals have been shown to increase the incidence of liver tumours in rats and/or mice by a mode of action (MOA) involving activation of the constitutive androstane receptor (CAR).
Toxicol. Res., 2018,7, 697-717
https://doi.org/10.1039/C8TX00008E
Chromatin dynamics underlying latent responses to xenobiotics
Xenobiotic-induced perturbations of somatic tissue or germline epigenomes may underlie delayed-onset and long-lasting adverse effects.
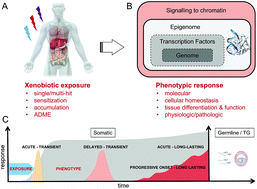
Toxicol. Res., 2018,7, 606-617
https://doi.org/10.1039/C7TX00317J
Goodbye to the bioassay
It is time to say goodbye to the standard two-year rodent bioassay.

Toxicol. Res., 2018,7, 558-564
https://doi.org/10.1039/C8TX00004B
Collaboration and competition: ethics in toxicology
Toxicology has frequently been a focus of scientific and societal concern. It's time to examine practices such as peer review, resourcing, transparency and data sharing to ensure the field stays current, relevant and ethical.

Toxicol. Res., 2018,7, 576-585
https://doi.org/10.1039/C7TX00306D
Skin and respiratory chemical allergy: confluence and divergence in a hybrid adverse outcome pathway
Sensitisation of the respiratory tract to chemicals resulting in respiratory allergy and allergic asthma is an important occupational health problem, and presents toxicologists with no shortage of challenges.
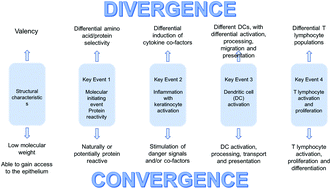
Toxicol. Res., 2018,7, 586-605
https://doi.org/10.1039/C7TX00272F
Case examples of an evaluation of the human relevance of the pyrethroids/pyrethrins-induced liver tumours in rodents based on the mode of action
The CAR-mediated MOA for liver tumorigenesis is of no carcinogenic risk for humans.
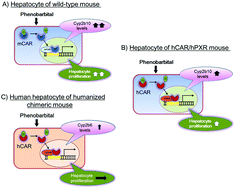
Toxicol. Res., 2018,7, 681-696
https://doi.org/10.1039/C7TX00288B
The failure of rodent carcinogenesis as a model for Man
Better understanding of carcinogenesis in Man makes clear that the detection of cancers in long-term rodent studies adds little to other predictive information, more reliably and reproducibly obtained.
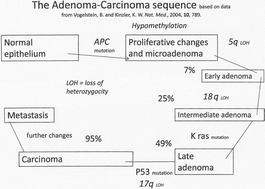
Toxicol. Res., 2018,7, 553-557
https://doi.org/10.1039/C7TX00283A
Screening for human urinary bladder carcinogens: two-year bioassay is unnecessary
Screening for carcinogens in general, and for the urinary bladder specifically, traditionally involves a two-year bioassay in rodents, the results of which often do not have direct relevance to humans with respect to mode of action (MOA) and/or dose response.
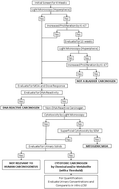
Toxicol. Res., 2018,7, 565-575
https://doi.org/10.1039/C7TX00294G
About this collection
This is a collection of papers consisting of invited contributions to honour the contributions made to toxicology by Professor Iain Purchase and Dr Cliff Elcombe. The British Toxicology Society (BTS) readily agreed with the guest editorial team that it would be appropriate to publish a special collection in their journal as a mark of respect and appreciation of the contribution of Iain Purchase and Cliff Elcombe made to the science of toxicology, primarily but not exclusively through their involvement with the Central Toxicology Laboratory (CTL). The contributors represent a range of scientific excellence, some having worked at CTL or had collaborated with scientists in CTL. Others are scientists whose success in solving toxicological problems represent the ambitions of Iain Purchase and Cliff Elcombe in their approach to addressing toxicological problems. Guest Edited by Brian Lake, Lewis Smith and Sam Cohen.