Themed collection Contemporary Research in Boron Chemistry

Contemporary Research in Boron Chemistry
Guest editors Shih-Yuan Liu and Douglas W. Stephan introduce the Boron Chemistry themed issue of Chemical Society Reviews.

Chem. Soc. Rev., 2019,48, 3434-3435
https://doi.org/10.1039/C9CS90053E
Solid-state hydrogen rich boron–nitrogen compounds for energy storage
Mechanistic studies of hydrogenation and dehydrogenation of boron and nitrogen containing compounds in the solid-state and its applications are reviewed.
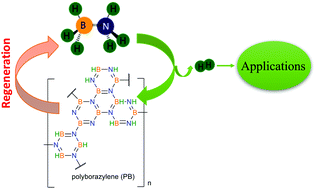
Chem. Soc. Rev., 2019,48, 5350-5380
https://doi.org/10.1039/C9CS00442D
Boronic acids as building blocks for the construction of therapeutically useful bioconjugates
This review summarizes boronic acid's contribution to the development of bioconjugates with a particular focus on the molecular mechanisms underlying its role in the construction and function of the bioconjugate, namely as a bioconjugation warhead, as a payload and as part of a bioconjugate linker.
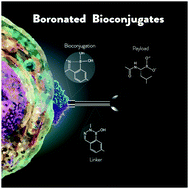
Chem. Soc. Rev., 2019,48, 3513-3536
https://doi.org/10.1039/C9CS00184K
Probing the structures and bonding of size-selected boron and doped-boron clusters
Photoelectron spectroscopy in conjunction with theoretical calculations has been used to investigate size-selected boron clusters, uncovering interesting structures and bonding.
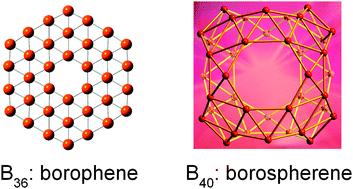
Chem. Soc. Rev., 2019,48, 3550-3591
https://doi.org/10.1039/C9CS00233B
Controlled functionalization of o-carborane via transition metal catalyzed B–H activation
This review summarizes recent advances in transition metal catalyzed vertex-specific BH functionalization of o-carborane for controlled synthesis of its derivatives.
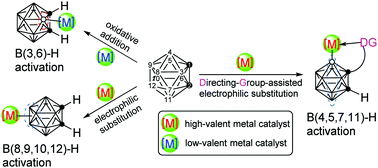
Chem. Soc. Rev., 2019,48, 3660-3673
https://doi.org/10.1039/C9CS00169G
Boron-based stimuli responsive materials
Representative types of boron-based molecular systems that respond to external stimuli such as temperature, pressure, light, or chemicals (oxygen, acid, base etc.) are described in this review article. The boron molecules are classified according to their operating mechanisms, with emphasis on systems, which are based on switchable boron-donor bonds and switchable excited states.
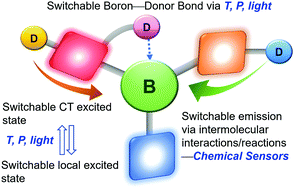
Chem. Soc. Rev., 2019,48, 3537-3549
https://doi.org/10.1039/C9CS00153K
Small molecule activation by boron-containing heterocycles
This review focuses on boron-containing heterocycles enabling the activation of σ- and π-bonds in small molecules.
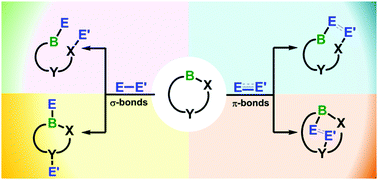
Chem. Soc. Rev., 2019,48, 3613-3659
https://doi.org/10.1039/C9CS00072K
FLP catalysis: main group hydrogenations of organic unsaturated substrates
This article is focused on recent developments in main group mediated hydrogenation chemistry and catalysis using “frustrated Lewis pairs” (FLPs).
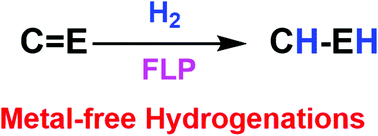
Chem. Soc. Rev., 2019,48, 3592-3612
https://doi.org/10.1039/C8CS00277K
New keys for old locks: carborane-containing drugs as platforms for mechanism-based therapies
Carborane derivatives as a new class of drugs (“new keys”) for important receptors (“old locks”) as targets for efficient therapeutic treatments.
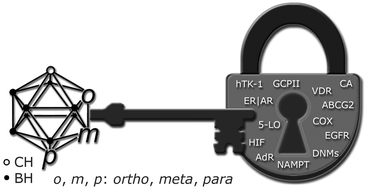
Chem. Soc. Rev., 2019,48, 3497-3512
https://doi.org/10.1039/C9CS00197B
Late-stage functionalization of BN-heterocycles
Late-stage functionalization techniques allow synthetic chemists to decorate an assembled BN-heterocycle for a variety of applications!
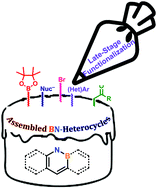
Chem. Soc. Rev., 2019,48, 3436-3453
https://doi.org/10.1039/C9CS00218A
Boronic acid catalysis
Although boronic acids are recognized primarily for their utility as reagents in transition metal-catalyzed transformations, other applications are emerging, including their use as reaction catalysts.
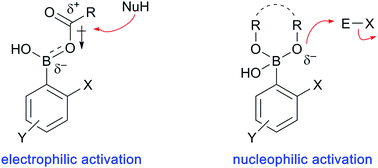
Chem. Soc. Rev., 2019,48, 3475-3496
https://doi.org/10.1039/C9CS00191C
Reactions of organoboron compounds enabled by catalyst-promoted metalate shifts
This tutorial review describes recent developments in catalytic reaction design that involve catalyst-promoted 1,2-metalate shifts as a critical part of the reaction mechanism.
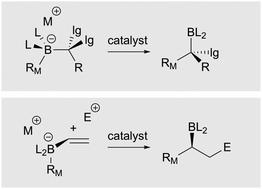
Chem. Soc. Rev., 2019,48, 3464-3474
https://doi.org/10.1039/C9CS00180H
Radicals derived from Lewis acid/base pairs
While conventional approaches to stabilizing main group radicals have involved the use of Lewis acids or bases, this tutorial review focuses on new avenues to main group radicals derived from combinations of donor and acceptor molecules.
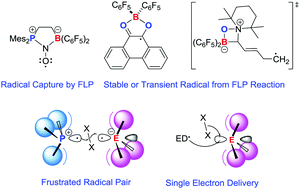
Chem. Soc. Rev., 2019,48, 3454-3463
https://doi.org/10.1039/C8CS00940F
About this collection
The 16th Boron Chemistry Meeting in the Americas (BORAM XVI), held last year at Boston College, collected more than 200 delegates from all over the world and from diverse backgrounds (academia, industry, and government) together to learn about the latest developments in boron chemistry. The scientific programmatic vision for this conference was to highlight the versatility of the element boron, in particular in four specific areas: medicinal chemistry, organic synthesis, materials chemistry, and fundamental chemistry.
The scope and aims of this themed issue on boron chemistry, Guest Edited by Doug Stephan (University of Toronto) and Shih-Yuan Liu (Boston College), follows the same spirit of the conference.