Activated clay/Opuntia microdasys incorporated polyvinyl alcohol membranes for fouling mitigation in wastewater filtration
Elikplim
Nyabi
a,
Andrews Ayim
Oduro
a,
Roland Tsoeke
Agbetsi
a,
Gideon
Addai
a,
Sampson Kofi
Kyei
 b,
Francis
Opoku
b,
Francis
Opoku
 a,
Ray Bright
Voegborlo
a and
Eric Selorm
Agorku
a,
Ray Bright
Voegborlo
a and
Eric Selorm
Agorku
 *a
*a
aDepartment of Chemistry, Kwame Nkrumah University of Science and Technology, PMB, Kumasi, Ghana. E-mail: seaky2k@gmail.com; Tel: +233 549394366
bDepartment of Chemical Engineering, Kumasi Technical University, Kumasi, Ghana
First published on 8th January 2026
Abstract
Membrane technology in water treatment is evolving rapidly, yet persistent challenges such as low selectivity, poor antifouling and inadequate contaminant recovery still limit its broader application. In this study, a sustainable membrane fabrication strategy integrating polyvinyl alcohol (PVA) with activated clay (AC) and Opuntia microdasys (OM) was developed to enhance ultrafiltration performance for dye–laden wastewater. Composite membranes were prepared via solution casting process and crosslinked with urea–formaldehyde to improve stability. Structural and surface properties were characterised using SEM-EDS, FTIR, 1H NMR, XRD, and ZETA-sizer. The incorporation of OM in addition to AC introduced surface porosity and hydrophilicity, enabling a dual-action mechanism of size exclusion and surface interaction for fouling control. The optimized composite membrane, MAC1:OM1, achieved 68.0% porosity and 67.5 L m−2 h−1 pure water flux (PWF) significantly outperforming the neat PVA membrane (52.1% porosity, 55.4 L m−2 h−1 PWF). A 29.7% increase in dye removal efficiency (RE) from 54.6% to 84.3% and a 56.4.% reduction in fouling (from 29.6% to 12.9%) were recorded. The flux recovery ratio (FRR) improved from 70.4% to 87.1%, indicating superior antifouling properties. This work highlights the synergistic role of natural fillers in engineering next-generation ultrafiltration membranes for efficient and eco-friendly wastewater remediation.
1. Introduction
Water pollution remains one of the gravest environmental threats globally, affecting both ecosystem health and human well-being. An alarming 80% of wastewater is discharged into the environment untreated, carrying with it a toxic mix of dyes, oils, heavy metals and organic compounds from industrial, agricultural and domestic sources.1–6 Among these, synthetic dyes are particularly problematic due to their recalcitrance, visibility and potential carcinogenicity.7–9 Methylene blue (MB), a cationic dye commonly used in textile, pharmaceutical and dyeing processes, poses a major environmental threat due to its high chemical stability and toxicity. Even at trace concentrations, MB can inhibit aquatic photosynthesis, disrupt aquatic ecosystems and cause adverse health effects in humans, including nausea, skin irritation and respiratory complications.10With freshwater scarcity intensifying, impacting over one billion people globally, the demand for robust, efficient and sustainable water treatment technologies has soared.11–13 Among the various technologies employed, membrane-based filtration stands out due to its operational simplicity, energy efficiency and minimal chemical requirements.14–16 However, persistent challenges such as membrane fouling, structural instability and short operational lifespan hinder its full-scale applicability.17 Fouling, in particular, reduces water flux, increasing energy demand and shortens membrane life, necessitating innovative solutions and membrane design.
Polyvinyl alcohol (PVA) has emerged as a popular matrix for membrane fabrication due to its biodegradability, hydrophilicity, chemical tunability and film-forming ability.18–20 However, native PVA membranes suffer from mechanical instability, swelling and fouling under prolonged exposure to aqueous environment.21 Recent approaches to address these shortcomings involve crosslinking and blending with natural or organic additives to enhance their mechanical strength, porosity and antifouling properties.22,23
Activated clay (AC), known for its large surface area, high cation exchange capacity, adsorptive power and abundance of negatively charged active sites that facilitate interactions with cationic pollutants. Additionally, AC contributes to antifouling properties by reducing organic deposition on several membrane and adsorbent systems with remarkable outcomes.24–26 Simultaneously, Opuntia microdasys (OM), bio-based cactus species rich in mucilage, lignin and carboxylic functionalities, has demonstrated coagulation and biosorption capabilities, yet remains underutilized in composite membrane applications.27–31 The integration of these two materials offers a unique synergy. AC impacts surface charge modulation and porosity, while OM introduces biopolymeric functionalities that promote hydrophilicity and fouling resistance.
Although various studies have explored clay-, polymer- or biosorbent-based membrane materials individually, few have combined both organic OM and inorganic AC filers in biodegradable PVA matrix for dye removal and antifouling enhancement. Table 1 presents a comparative summary of recent literature on membrane-based composite membranes highlighting the novelty of this work. Specifically, this study investigates the synergistic incorporation of AC and OM into crosslinked PVA membranes, evaluating their structural, physicochemical and antifouling properties for methylene blue (MB) dye removal under dead-end filtration. Our results demonstrate a significant increase in porosity, water flux, removal efficiency and flux recovery, confirming the dual-function mechanism of Donnan exclusion and size-sieving as the core drivers of membrane performance.
| Membrane | Material removed | Flux (L m−2 h−1) | Removal efficiency (%) | Ref. |
|---|---|---|---|---|
| GO-WO2.89/PPSU | Serum albumin | 636.01 | 82.86% | 32 |
| PES/SiO2-g-PEI | Mebeverine hydrochloride | 140 ± 1.28 | 99.99% | 33 |
| PVA/AC/HA | Methylene blue | 70.3 | 95.5 | |
| CA/PVA | Crystal violet dye | 17 | 99.9 | 34 |
| MAC3.0/CNS3.0 | Methylene blue | 71 | 95.7 | 35 |
| PVDF-ZnO/Ag2CO3/Ag2O | IBF | 60 | 24 | 36 |
| Mt/PSSMA + PVA/PSf | Ibuprofen | 14.4l | 83.37 | 37 |
| PSF/PVP/SiO2 | Amoxicillin | 42.28 | 89.81 | 38 |
| TiO2/PVA coated PVDF | Reactive Blue | 16.30 | 44.4 | 39 |
| TiO2/PVA coated PVDF | Rhodamine b | 16.30 | 45.8 | 39 |
| MAC1:OM1 | Methylene blue | 67.5 | 84.3 | This work |
This study therefore, provides a sustainable and locally accessible strategy for enhancing membrane performance using natural abundant fillers. The successful integration of AC and OM into PVA membranes addresses two present global concerns, environmental remediation and circular use of agro-residues. By providing performance data and mechanistic insights, this work contributes to the development of scalable, low cost of ultrafiltration membranes tailored for wastewater treatment applications in resource-limited settings.
2. Experimental
2.1 Materials and methods
A variety of chemicals were utilized, such as polyvinyl alcohol (PVA) from Breckland Scientific Supplies (United Kingdom) of molecular weight 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–124
000–124![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Da. Sulfuric acid (H2SO4, >98%) and formaldehyde (CH2O >98%) from Breckland Scientific Supplies (United Kingdom). Methylene blue dye, ethanol (CH3CH2OH > 98%) and urea (CH4N2O > 98%) were purchased from Sigma-Aldrich (Germany). Raw clay was obtained from Teleku Bukazo in the Western Region of Ghana. Opuntia microdasys was also sourced from Wudome-Peki, in the Volta Regio of Ghana.
000 Da. Sulfuric acid (H2SO4, >98%) and formaldehyde (CH2O >98%) from Breckland Scientific Supplies (United Kingdom). Methylene blue dye, ethanol (CH3CH2OH > 98%) and urea (CH4N2O > 98%) were purchased from Sigma-Aldrich (Germany). Raw clay was obtained from Teleku Bukazo in the Western Region of Ghana. Opuntia microdasys was also sourced from Wudome-Peki, in the Volta Regio of Ghana.
2.2 Activated clay preparation procedure
A raw clay sample of 25 g was placed in a 400 mL beaker, after which 100 mL of distilled water and 100 mL of 4 M H2SO4 were added in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. The mixture was stirred using a magnetic stirrer for 30 minutes. The activation process continued for 24 hours, after which the residue was filtered and washed several times with distilled water to remove the remaining acid. The clay was dried at 110 °C in an oven. The activated clay (AC) was finally transferred to a desiccator to cool.
1 ratio. The mixture was stirred using a magnetic stirrer for 30 minutes. The activation process continued for 24 hours, after which the residue was filtered and washed several times with distilled water to remove the remaining acid. The clay was dried at 110 °C in an oven. The activated clay (AC) was finally transferred to a desiccator to cool.
2.3 Mucilage extraction from Opuntia microdasys (OM)
Fresh, healthy and mature pads (cladodes) of OM (Fig. 1(a)) were collected from Wudome-Peki, in the Volta Regio of Ghana. A sharp knife was used to remove the spines, glochids and the green outer pads. To eliminate debris, the mucilaginous inner tissue was divided into smaller pieces and rinsed with distilled water. The strained mucilage was spread evenly on a tray and oven-dried at 50 °C for several hours until it became brittle, and a constant weight was obtained. The mucilage was ground into a fine powder (Fig. 1(b)) using a spice grinder. The powdered form was stored in an airtight container and stored in a cool, dry place.2.4 Synthesis of PVA/AC:OM composite membranes
Prior to final formulation, a range of PVA/AC:OM blends were prepared with total filler content spanning 10–50 wt%. Visual inspection, mechanical testing (folding endurance and film integrity), and initial filtration tests (MB rejection and water flux) were performed. Membranes with less than 15 wt% showed weak performance gains, while those with greater than 45 wt% exhibited phase separation, casting difficulties and physical instability. As such, three compositions (Table 2) were selected as representative and practical for full characterization in this study.| Membrane | Composition | PVA (wt%) | Composition of filers (wt%) | |
|---|---|---|---|---|
| AC | OM | |||
| MPVA | PVA only | 100.0 | — | — |
| MAC1:OM1 | PVA + AC + OM | 45.5 | 27.3 | 27.3 |
| MAC2:OM1 | PVA + AC + OM | 50.0 | 20.0 | 30.0 |
| MAC1:OM2 | PVA + AC + OM | 50.0 | 30.0 | 20.0 |
Membranes composed of pure PVA (MPVA) and PVA reinforced with AC and OM fillers (PVA/AC:OM) were fabricated via the classical solution casting method, utilizing distilled water as the solvent medium. Initially, 5 g of PVA was gradually dissolved in 40 mL of distilled water at 90 °C under continuous magnetic stirring of 500 rpm in a round-bottom flask for four hours. This ensured complete dissolution and homogenization of the polymer chains, yielding a clear, viscous aqueous PVA solution.
In a parallel step, predetermined amounts of AC and OM were individually dispersed in 10 mL of distilled water for 45 minutes to initiate swelling and surface activation. The swollen fillers were than sonicated using a probe sonicator at 40 kHz and 300 W for one hour to ensure particle dispersion and deagglomeration. The swelling behaviour of AC arises from its inherent layered silicate structure, wherein water molecules intercalate into the interlayer galleries, expanding the clay matrix.40 This swelling is critical, as the negatively charged surface of AC facilitates electrostatic interactions with cationic contaminants, particularly dyes, enhancing its performance as a functional filler. Meanwhile, natural macromolecules such as proteins, lipids, fibres and polyphenols in OM can interact physically and chemically with water through van der Waals interactions and hydrogen bonding thereby augmenting the hydrophilicity and adsorption potential of the composite matrix.28 The PVA solution that had already been refluxed was then mixed with the bulk solutions of AC and OM under continuous stirring at room temperature for one hour and again subjected to 30 minutes of sonication to enhance homogeneity. The resulting polymer-filler solution was carefully cast onto a previously clean, dust-free glass substrate and allowed to air-dry at ambient temperature (25–28 °C) for 72 hours to form a uniform membrane thickness. After drying, the membranes were peeled off and stored in a desiccator for further characterisation and performance evaluation. A schematic overview of the fabrication process is illustrated in Fig. 2. Table 2 gives a summary of the concentration of PVA, AC and OM.
 | ||
| Fig. 2 Schematic illustration of the synthesis and fabrication process of PVA/AC:OM composite membranes via the solution casting method. | ||
2.5 Crosslinking of membranes
The inherent hydrophilic nature of PVA due to the presence of an abundance of hydroxyl functional groups (–OH), which facilitate strong hydrogen bonding interactions with water molecules offers a great advantage for the hydrophilicity-driven filtration process. It also renders PVA highly soluble leading to excessive swelling and eventual dissolution in aqueous environments. Furthermore, the incorporation of AC and OM fillers, which also exhibit hydrophilic tendencies, exacerbates the structural instability of the membrane over prolonged exposure to water.To mitigate this limitation, crosslinking is employed as a strategy to introduce covalent interchain linkages within the PVA matrix thereby restricting polymer chain mobility and reducing water-induced swelling. Crosslinking enhances the mechanical integrity, thermal stability and water resistance of the membrane ensuring its long-term performance. Additionally, it minimizes the leaching of embedded fillers, thereby maintaining the membrane's functional properties and prolonging its operational lifespan To crosslink these membranes, urea, formaldehyde and sulphuric acid were used.41 The precise composition of the crosslinking mixture consisted of 1.4 w/w% sulphuric acid, 2.0 wt% formaldehyde, 2.5 w/w% urea and a 64 w/w% aqueous ethanol solution. The dried membranes were immersed in the crosslinking solution. The membranes were checked to make sure they were completely submerged and equally covered with the solution. Periodically, the mixture was gently shaken to make sure the membranes were evenly exposed to the crosslinking solution for 5 hours. The crosslinked PVA composite membranes were washed with distilled water to remove excess reagents. Finally, the membranes were dried to obtain the solid crosslinked PVA membranes.
Fig. 3 depicts the reaction pathway for the urea–formaldehyde production and PVA crosslinking. Formaldehyde and water react in the first stage to create dihydroxymethane. Water attacks the carbonyl carbon in formaldehyde as a nucleophile because it is highly electrophilic, which leads to the creation of dihydroxymethane. Acidic conditions are necessary for the urea–formaldehyde synthesis to occur, as the acid acts as a dehydrating agent. Crosslinked PVA membranes are created in this technique when the hydroxyl groups positioned axially in the PVA react with the nitrogen atoms in the NH2 groups of the urea–formaldehyde crosslinker.42
 | ||
| Fig. 3 Mechanism of urea–formaldehyde cross-linker synthesis (a) and its reaction with PVA backbone (b). | ||
2.6 Preparation of working solutions and calibration curve of methylene blue (MB) dye
A stock solution of MB dye (Fig. 4) was prepared by dissolving 0.0125 g of the dye in distilled water to obtain a concentration of 50 mg L−1. This was then serially diluted to give several working solutions, with MB dye concentrations ranging from 5 to 50 mg L−1.2.7 Characterization
FTIR analysis was performed utilizing a Bruker Alpha II Platinum ATR type FTIR spectrometer (Germany) to identify the functional groups in MPVA and MPVA/AC:OM composites membranes before and after filtration of MB dye solution. Using a Shimadzu UV-1800 spectrophotometer (Japan), the concentration of MB dye was measured at a wavelength of 664 nm both before and after filtering. To find the crystallinity and mineralogical compositions of the AC and raw clay components, an X-ray diffraction examination was carried out using a Bruker D2 Phaser X-ray powder diffractometer (Germany) fitted with Cu-Kα X-ray radiation (λ = 1.5418 Å). A Malvern Zetasizer (UK) was used to evaluate the particle size and zeta potential (ZP) of the particles in the MPVA/AC:OM composite membranes using dynamic light scattering (DLS) and electrophoretic light scattering (ELS) techniques. This analysis provided into the membrane's polydispersity index (PDI), colloidal dispersion stability, and particle size distribution. The Zeiss EVO MA 15 SEM-EDX (Germany) was used to analyse the membranes' surface morphology and elemental composition. Structural analysis of the composites was carried out using D2O on a Bruker 400 1H NMR spectrometer (Germany). To aid in the filtration process, a Stuart RE3022C vacuum pump (UK) was also used.2.8 Membrane performance
Membrane performance is a vital factor in wastewater filtration, separation and treatment processes. The selective passage of substances is a key variable influencing various characteristics such as porosity, morphology, hydrophilicity, pure water flux, and resistance to fouling.43 The overall efficiency and practicality of membranes depend on a blend of their selectivity and the rate at which materials are processed. This study assessed the suitability of 256 µm composite membranes for various applications.2.9 Particle size calculation
The average crystallite sizes and minerals present in both raw and acid-activated clay were calculated using the Scherrer equation (eqn (1)).44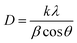 | (1) |
2.10 Porosity measurement
The porosity of the membranes was determined by calculating the mass loss of the membranes after drying. The membranes were initially soaked in distilled water, removed then blotted dry. They were afterwards dried in the oven at 35 °C to a constant weight, which was recorded. The weight difference between the wet and dry membrane was used to calculate the porosity, as indicated in eqn (2).45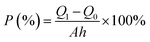 | (2) |
2.11 Pure water flux and dye removal efficiency of membranes
Composite membranes made from PVA, activated clay and Opuntia microdasys were tested with 2000 mL of pure water and a 50 mg L−1 MB dye solution. The filtration tests on PVA composite membranes were carried out in a dead-end filtration set-up with a membrane filtration area of 78.5 cm2. A pressure of 0.1 MPa was used to measure the pure water flux of the membrane (PWF, L m−2 h−1). The initial water flux (Q0), pollutant flux (MB dye solution) (Q1) and final water flux (Q2) were all monitored at 10-minute intervals during the 120 minutes of constant pressure at room temperature. To remove any fouling from the membranes caused by dye adsorption, the membranes were cleaned with acetone followed by DI water before Q2 evaluation. The permeate flux (Jp) was calculated using eqn (3).46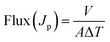 | (3) |
MB dye solution was used to test the performance of the membrane to examine the impact of dye concentration of feed solution on separation performance.
Using eqn (4), the dye removal efficiency (RE) was determined.
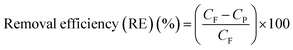 | (4) |
The flux recovery rate (FRR, %) was determined using eqn (5).46
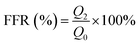 | (5) |
2.12 Anti-fouling performance experiments
There is an inverse relationship between flux recovery ratio (FRR) and membrane fouling. Membrane fouling occurs when molecules adsorb onto the surface of a membrane or block its pores. The fouling extent was calculated using eqn (6).45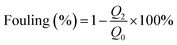 | (6) |
2.13 Membrane reusability
The best-performing membrane (MAC1:OM1) was subjected to cycles of dye filtration to evaluate its reusability. This was followed by cleaning the membrane's surface using acetone and water after every cycle and repeating the filtration process. The amount of dye removal for the MB dye was calculated using eqn (4).3. Results and discussion
Characterization comprises one of the fundamental processes in material science or chemistry by which the structural, morphological, size and chemical composition properties of materials are analysed. Techniques such as Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), scanning electron microscopy (SEM), nuclear magnetic resonance (NMR), Zetasizer/potential (Zp), etc. were predominantly used to study material composition, morphology, and stability, which are vital in enhancing material performance in diverse applications.On the other hand, membranes are some of the most utilized materials in separation processes, allowing, selective permeation, while blocking others. They are involved in several processes like the purification of water, gas separation, biomedical-related applications and industrial filtration. Membrane efficacy is affected by pore size, surface charge, hydrophilicity and mechanical strength, all of which can often justifiably be controlled through material characterization.43
3.1 FTIR analysis
The FTIR spectra of both raw and activated clay (AC) are illustrated in Fig. 5(a). The prominent bands at 3691 cm−1 and 3670 cm−1 in raw and AC respectively, are associated with O–H stretching vibrations. In contrast to the AC, the raw clay's peak intensity was lower, suggesting that moisture was lost during activation and drying.47 Si–O bonds in raw clay showed vibrations located at 1001 cm−1 which slightly shifted to 1000 cm−1 in activated clay reflecting subtle modifications in the silica framework. The Si–OH stretching vibration characteristics of kaolinite in the raw clay found at 909 cm−1 were not found in AC. The peak at 750 cm−1 shows the existence of Al–O vibration suggesting the presence of aluminium–oxygen interactions in raw clay structure was shifted to 751 cm−1 in AC indicating potential restructuring of aluminium sites.48 The band at 691 cm−1 corresponds to Si–O, indicating that the sample may contain free silica or unmodified quartz. Al–O and Al–O–Si deformations show minor shifts, from 528.77 cm−1 and 460.02 cm−1 in raw clay to 529.35 cm−1 and 460.85 cm−1 in AC respectively.49 Corundum, associated with Al2O3 was found only confirming its formation due to acid treatment. Additionally, albite was identified by the peaks at 749.71 cm−1 in both samples with slight peak shifts indicating structural changes in response to activation.50 These observations showed how acid caused structural and chemical changes in the clay.
Fig. 5b compares the FTIR spectra of urea–formaldehyde crosslinked PVA membranes and pure PVA membranes. Upon crosslinking PVA membranes with urea–formaldehyde, the OH stretching decreased slightly from 3265.68 cm−1 to 3263.06 cm−1. This is explained by the chemical reaction between the hydroxyl group of PVA and the formaldehyde group.42 The C–H stretching vibration of methyl (CH3) groups is responsible for the peak around 2939.18 cm−1. The cross-linking procedure of PVA resulted in a new absorption peak in the spectrum that was assigned to the stretching of an amide (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) group at 1626.90 cm−1. This indicated the existence of urea in the crosslinked material.51 This peak's existence indicates that MPVA and the urea–formaldehyde crosslinker may have established amide bonds.51 The stretching vibration of C–O bonds in ether groups is indicated by the peak which was observed at 1082 cm−1. This peak observed in the MPVA shifted to 1087 cm−1 in the crosslinked PVA indicating the presence of ether functionalities.52
O) group at 1626.90 cm−1. This indicated the existence of urea in the crosslinked material.51 This peak's existence indicates that MPVA and the urea–formaldehyde crosslinker may have established amide bonds.51 The stretching vibration of C–O bonds in ether groups is indicated by the peak which was observed at 1082 cm−1. This peak observed in the MPVA shifted to 1087 cm−1 in the crosslinked PVA indicating the presence of ether functionalities.52
The FTIR spectra of PVA, AC, OM and their composite membranes (PVA/AC:OM) are shown in Fig. 6. The chemical interactions and structural characteristics of AC, OM and PVA/AC:OM composite can be thoroughly understood using FTIR spectroscopy investigation. Notably, the chemical compositions of each constituent and their composite are clarified by the FTIR spectra, which show characteristic peaks for each.
MPVA has a broad peak at 3265 cm−1 that is attributable to hydroxyl (OH) groups, whereas AC shows a notable band at 3619 cm−1 that is linked to stretching vibrations of OH.53 The large absorption band in the OM spectrum at 3357 cm−1 also indicates the stretching frequency of the OH group.54 The combined OH contributions from PVA, AC and OM are shown by a peak on the composite membrane (PVA/AC:OM) at 3265.59 cm−1.55 The peaks at 2910 cm−1 (MPVA), 2918 cm−1 (OM) and 2908 cm−1 (MPVA/AC:OM) clearly show aliphatic vibrations, which are absent in AC. There is no discernible peak for AC in this region, indicating that aliphatic vibrations are not present. At 1603 cm−1, OM shows a prominent stretching band linked to double bond stretching in carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), C
O), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, or C–N groups. The composite combines the contributions of PVA and OM peaks at 1655 cm−1 for C
C, or C–N groups. The composite combines the contributions of PVA and OM peaks at 1655 cm−1 for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups.56 Moreover, the OM peak at 1316 cm−3 indicates C–N stretching vibrations, which are suggestive of functional groups such as amines or amides. PVA and OM interactions are shown by the composite's peak at 1325.67 cm−1, which is linked to C–N vibrations.
O groups.56 Moreover, the OM peak at 1316 cm−3 indicates C–N stretching vibrations, which are suggestive of functional groups such as amines or amides. PVA and OM interactions are shown by the composite's peak at 1325.67 cm−1, which is linked to C–N vibrations.
Importantly, notable spectra overlap occurs in the 1000–1100 cm−1 region, where both C–O stretching vibrations from PVA/OM ether linkages and Si–O–Si stretching modes from the AC component contribute. The OM peak at 1029 cm−1 (C–O stretching) and the AC peak at 1001 cm−1 (Si–O–Si stretching) appear merged in the composite, producing a broadened absorption centred at 1085–1080 cm−1.57 This broad intensified band signifies their coexistence and interaction of organic (C–O) and inorganic (Si–O–Si) functionalities within the polymer–clay matrix.58
The peak at 909 cm−1 for AC which shifted to 914 cm−1 in the PVA/AC:OM is ascribed to Al2O3 bending vibrations.58 Furthermore, the Si–O (749 cm−1) bending vibrations in muscovite59 in the AC shifted to 691 cm−1 in the composite membrane indicating Si–O interactions with the PVA. The Al–O stretch in phengite, observed at 528 cm−1 in AC shifted towards 532 cm−1 in the composite, indicating the inorganic contribution of AC to the composite membrane.59 Again Al–O–Si bending vibrations in albite were observed in the AC at a peak shown at 460 cm−1 and in the composite membrane, the value shifted to 437 cm−1 indicating chemical interactions in the composite.60
3.2 XRD analysis
The crystalline components of both raw and activated clay were determined using X-ray diffraction analysis. The diffractogram with diffraction lines corresponding to the following minerals are displayed in Fig. 7. Both activated and raw forms of phengite (P), magnetite (Ma), muscovite (M), quartz (Q), corundum (C), kaolinite (K), and albite (A) were observed. While corundum showed distinct peaks at 2θ values of 25.6°, 35.2°, 37.8°, 41.7° and 43.6°, albite showed peaks at 21.4°, 27.7° and 15.5°. Intense peaks were observed in quartz with 2θ values at 20.6°, 26.4°, 36.1°, 39.1° and 39.9°. These findings are similar to the results published by Fuente et al.61 At 2θ values of approximately 18.7°, 30.8°, 36.3°, 37.9°, 44.0° and 48.3°, magnetite was observed to exhibit peaks. Phengite revealed XRD peaks at 2θ values of 36.4°, 36.9° and 37.3°, which is in agreement with findings made by Kangmennaa et al.62 while muscovite showed peaks at 8.9°, 17.8°, 19.9° and 20.2°.63 Albite and kaolinite were not observed in the activated clay after acid activation. Acid attacks on the aluminium–silicon bonds in kaolinite are responsible for the dissolving of these minerals because they cause the solubilization of silica and alumina components as well as a disruption of the crystalline structure. Comparably, acid activation dissolves albite, a sodium and aluminium-rich for the activated clayfeldspar mineral, making it undetectable in the XRD analysis.26The average grain sizes calculated using eqn (1) for raw clay and activated clay were 82.14 nm and 66.72 nm respectively.
3.3 1H NMR of MPVA and PVA/AC:OM composite membrane
The 1H NMR spectra of the MPVA/AC:OM composite membrane and the MPVA membrane are shown in Fig. 8. The aliphatic (CH3) chemical group of PVA protons is responsible for the chemical shift at 1.7 ppm (Fig. 8a). The vinyl alcohol units' –OH groups are shown by the peaks at 4.91 ppm.64 Furthermore, the signal at 3.80 ppm represents the protons of methylene (CH2) in the polymer chain.65 The composite membrane spectrum (b) shows a notable signal at 1.6 ppm, which denotes chemical shifts from the aliphatic protons of both PVA and OM inside the membrane. The protons in the methylene groups of PVA and OM in the composite material are linked to the chemical shift at 4.0 ppm. The peak at 5.2 ppm influences the OH peaks in both PVA and OM.66 These studies emphasize the unique chemical shifts connected to the different components of the PVA/AC:OM composite membrane and offer insightful information about the molecular structure and interactions within it.3.4 SEM analysis of MPVA/AC:OM
The surface morphology of the PVA/AC:OM composite membrane was examined using a scanning electron microscope (SEM) at low and high magnifications at 539×, 1.88k× (Fig. 9a and b). Top-view images of PVA/AC:OM before filtration are displayed in Fig. 9a. The membrane has a unique and complex surface morphology when examined under a scanning electron microscope. The surface exhibits a non-linearity, suggesting a multifaceted interaction between the PVA, AC and OM constituents. The membrane's core is made of a PVA matrix. The surface appears to be made up of a network of connected PVA strands at lower magnifications, indicating a well-integrated matrix. The PVA matrix's general homogeneity offers a solid basis for the inclusion of other elements. Across the membrane surface, AC particles are visible. According to the SEM images, the AC is uniformly distributed and forms aggregations and clusters inside the MPVA matrix. The presence of AC contributes to modifications in surface topography enhancing specific properties such as mechanical strength or adsorption capacity.67 Within the matrix, OM particles are visible and frequently cohabit with the AC clusters. The inclusion of OM creates more surface characteristics that give the surface a textured look. It is clear that OM and PVA interact, affecting the membrane's overall surface properties. The membrane surface is covered in pores of various sizes and shapes. The addition of OM and AC aided in the creation of pores, which could affect the permeability of the membrane. At greater magnifications, surface roughness becomes visible, exposing details and imperfections. The SEM study shows several pores and holes distributed across the membrane. In membrane science, pores refer to interconnected voids or channels that traverse or partially penetrate the membrane matrix, allowing the selective passage of molecules critical for filtration performance. Holes, on the other hand, are typically larger, more irregular surface openings that may or not be interconnected. These are often by-products of structural heterogeneity or filler agglomeration. The interaction of PVA, AC and OM results in the formation of surface characteristics, which increases the membrane's overall roughness.Changes in the PVA/AC:OM membrane's surface morphology after filtration are visible by SEM imaging Fig. 10. The original complex network shown in the untreated membrane changed, and the total topography is affected by the filtration of methylene blue (MB) dye. The successful filtration of the dye through the membrane is indicated by the distinct MB dye particles that are visible across the membrane surface. Clusters of MB dye may be seen, indicating differences in adsorption or contact with the membrane components, and the dispersion of MB dye particles is not uniform. Changes in the pore structure can be seen by comparing the SEM images taken before and after filtration. Changes in pore size and shape might be caused by the presence of MB dye particles inside the membrane pores. Areas of significant adsorption or possible concentration gradients during filtration are indicated by portions of the membrane surface that are extensively covered in dye particles, as seen in SEM pictures. The SEM examination evaluates the general integrity of the PVA/AC:OM membrane post-filtration, even if the distribution of MB dye is clearly visible. The membrane structure seems to be preserved, and the dye particles are mostly confined to the surface instead of profoundly entering the matrix. MB dye particle aggregation (a phenomenon in which several dye particles group together) were observed. The interaction of the dye with the components of the membrane, which affects the adsorption kinetics, may have an impact on this phenomenon.
3.5 EDX analysis of PVA/AC:OM
To determine the elements in the membrane both before and after filtration with MB dye, an energy-dispersive X-ray (EDX) analysis of the composite membrane was performed. Before MB dye filtering, the presence of various components and their related weight percentages were shown in Fig. 11. Carbon, which makes up the largest percentage (58.08%), comes from the PVA component as well as the organic compounds that are in the OM extract. Next in importance at 38.59% is oxygen, which is present in PVA, OM, and AC minerals like quartz, muscovite, and albite. This indicates that the composite membrane is hydrophilic. The trace amounts of silicon (1.25%), calcium (0.55%), and aluminium (0.52%) indicate that clay minerals like quartz and albite may have contributed to the composition of the membrane. The trace amounts of sulphur (0.87%) and potassium (0.13%) arise from organic compounds present in the OM. Overall, the elemental composition gives information on the composite membrane's chemical composition prior to the filtration processes by reflecting the mix of PVA, AC and OM. | ||
| Fig. 11 SEM image, elemental mapping analysis and EDX analysis of MAC1:OM1 composite membrane (a) before and (b) after filtration. | ||
3.6 ZETA-sizer of clay and MAC1:OM1 composite membrane
Fig. 12 presents key characterisation data of the membrane system: (a) the zeta potential of AC, (b) the size distribution by volume, (c) the size distribution by intensity and the zeta potential for the MPVA/AC:OM composite membrane. The zeta potential of AC was measured at −8.20 mV, with a corresponding particle size distribution of 82.06 nm. This negative surface charge indicates good colloidal stability, minimizing flocculation or aggregation during suspension.68 | ||
| Fig. 12 (a) Zeta potential of activated clay, (b) size distribution by volume, (c) size distribution by intensity and (d) zeta potential of MAC1:OM1. | ||
When AC and OM were incorporated into the PVA matrix, a noticeable increase in particle size was observed. The composite membrane exhibited a zeta average diameter of 84.01 nm, slightly larger than that of AC, suggesting that the fillers contributed to increased particle aggregation or the formation of more complex hydrated structures within the matrix.69 This increase in particle may influence membrane porosity and subsequently affect its filtration behaviour, as larger particles could lead more open structures, potentially enhancing permeability while still allowing effective separation.70
A comparison of particle size estimated from two characterisation techniques, XRD and dynamic light scattering (DLS) via the Zetasizer further revealed key insights. XRD-derived crystalline sizes were 82.14 nm for raw clay and 66.72 nm for acid-activated clay, both smaller than the hydrodynamic size obtained from DLS (82.06 nm). This discrepancy is expected, given the different measurement principles. XRD captures only the size of coherent crystalline domains, while DLS measures the hydrodynamic diameter, which includes solvation layers, surface water, and possible inter-particle interactions in suspension.71,72 Thus, while XRD reveals the intrinsic crystallinity of the clay, DLS reflects its dynamic behaviour in an aqueous environment.
The zeta potential of the MAC1:OM1 composite membrane was recorded at −1.36 mV, a shift towards neutrality compared to the AC. This decrease in surface charge can be attributed to the integration of OM and AC into the MPVA matrix, which may alter the membrane's surface chemistry.73 Although less negative, the membrane still maintains electrostatic stability, aided by its surface functional groups. A zeta deviation of 35.2 mV suggests a degree of surface heterogeneity within the composite, potentially resulting in variable charge distribution across the membrane surface. This heterogeneity could positively influence membrane performance enhancing porosity, promoting fouling resistance, and facilitating better dye rejection.55
Together, the zeta potential and particle size distribution provide critical insights into the physicochemical nature of the composite membrane. They highlight how AC and OM influence membrane morphology, electrostatic behaviour, and stability. These factors that are directly linked to performance characteristics such as permeability, fouling resistance and separation efficiency.
3.7 Membrane performance
In the field of water filtration, separation and treatment, membrane performance is a pivotal factor in determining both efficiency and long-term operational stability. The selective transport of substances across a membrane is shaped by interrelated properties such as porosity, morphology, hydrophilicity, pure water flux and antifouling behaviour. To enhance these characteristics, the incorporation of functional additives into polymeric membranes has become a proven approach.74 In this study, AC and OM were introduced as natural, and functional fillers to improve membrane structure, fouling and performance. Their inclusion is known to increase hydrophilicity, modulate pore architecture and reduce fouling tendencies, thereby improving overall permeability. The experimental investigation focused on three composite membranes compared to a MPVA membrane of 256 µm thickness, assessing key parameters such as porosity, permeability and resistance to fouling. The membranes are visually presented in Fig. 1 illustrating their distinct physical appearances and setting the stage for performance evaluation.3.8 Porosity and pure water flux (PWF) of synthesized membranes
The experimental data, presented in Table 3 and visualised in Fig. 13 and 14 demonstrates a clear correlation between membrane composition and performance particularly in relation to porosity and PWF. The unmodified MPVA exhibited a baseline porosity of 52.1% and a corresponding PWF of 55.4 L m−2 h−1. Upon incorporation of AC and OM, all composite membranes showed significant improvement in these parameters. Notably, the MAC1:OM1 (27.3 wt% AC:27.3 wt% OM) recorded the highest porosity (68.0%) and PWF (67.5 L m−2 h−1), indicating a synergistic enhancement in structural and transport properties.| Membrane | Porosity (%) | PWF (L m−2 h−1) |
|---|---|---|
| MPVA | 52.1 | 55.4 |
| MAC1:OM1 | 68.0 | 67.5 |
| MAC2:OM1 | 62.7 | 60.4 |
| MAC1:OM2 | 58.5 | 58.2 |
 | ||
| Fig. 14 Comparison of porosity and PWF for pristine MPVA and composite PVA/AC:OM synthesized membranes. | ||
The observed enhancement in porosity can be attributed to the unique physicochemical characteristics of AC. AC primarily composed of aluminosilicates, exhibits a layered structure with high surface area and interstitial spacing.75 When dispersed within the PVA matrix, these clay platelets disrupt the dense polymer network, creating microvoids and interfacial channels that enhance porosity and facilitate water permeation. The presence of hydroxyl and silanol groups on AC surfaces also enhances hydrogen bonding interactions with water, increasing membrane hydrophilicity and promoting flux.76
OM, on the other hand, contributes to membrane enhancement through its rich composition of hydrophilic polysaccharides, such as pectin and cellulose.77 These macromolecules contain multiple hydroxyl groups, which not only increase the hydrophilic nature of the membrane but also improve water adsorption and wettability.77 Furthermore, the fibrous and irregular morphology of OM introduces additional pore networks and interconnectivity, which synergistically boost the porosity and flux characteristics of the composition membranes.78
The trend in PWF aligns with porosity values, supporting the Hagen–Poiseuille principle, which states that water flux through a porous medium is directly proportional to the pore radius and number of pores.79 The MAC1:OM1 membrane's superior performance can be ascribed to the optimal distribution and interaction of both additives within the polymeric matrix. Conversely, MAC2:OM1 (20 wt% AC, 30 wt% OM) and MAC1:OM2 (30 wt% AC, 20 wt% OM) exhibited slightly reduced porosity and flux values, emphasizing the critical role of additive ratios. The performance drops in MAC2:OM1 suggest that a lower AC concentration limits the creation of effective transport channels, while the reduced OM content in MAC1:OM2 slightly hampers the hydrophilic enhancement and structural support provided by OM.
3.9 PWF and MB dye removal efficiency of membranes
This study sought to unravel the interplay between membrane composition, water permeability (PWF) and removal efficiency, specifically in relation to MB dye removal. By systematically varying the proportions of AC and OM within the polymeric matrix, the study aimed to understand the underlying mechanisms influencing filtration performance and to identify optimal additive ratios for enhanced functionality. The results, summarized in Table 4 and Fig. 15 reveal a clear trend. Increasing the concentrations of AC and OM leads to significant improvements in both PWF and RE. Conversely, a reduction in additive content results in a measurable decline in membrane performance.| Membrane | PWF (L m−2 h−1) | RE (%) |
|---|---|---|
| MPVA | 55.4 | 54.6 |
| MAC1:OM1 | 67.5 | 84.3 |
| MAC2:OM1 | 60.4 | 81.7 |
| MAC1:OM2 | 58.2 | 73.6 |
The MPVA served as a baseline, exhibiting a PWF of 55.4 L m−2 h−1 and a relatively low RE of 54.6%. In contrast, the composite membranes demonstrate substantial improvements in both parameters. Notably, the MAC1:OM1 (27.3 wt% AC:27.3 wt% OM) membrane achieves the highest RE of 84.3% alongside a PWF of 67.5 L m−2 h−1.
The significant enhancement may be attributed to the complementary roles of AC and OM in modulating the physicochemical properties of the membrane. AC, with its high surface area and layered aluminosilicate structure, introduces a dense network of interlayer spaces and adsorption sites.80 These features facilitate effective dye entrapment via electrostatic interactions between the negatively charged AC surface and the cationic MB dye molecules.62 Moreover, the porosity induced by the dispersed AC layers enhances water permeability while simultaneously enabling deeper dye adsorption within the membrane matrix.43
OM contributes through a distinct but equally vital mechanism. Rich in natural polysaccharides such as cellulose and pectin, OM introduces abundant hydroxyl and carboxyl groups which can form hydrogen bonds and electrostatic attractions with MB dye molecules. Moreover, the fibrous, spongy-like structure of OM also creates a porous network that can trap dye molecules.78
The observed trend in RE correlates with the trends in PWF. The MAC1:OM1 membrane, exhibiting the highest PWF, also demonstrates the highest RE. This correlation suggests that the increased porosity and surface area of the composite membranes facilitate both water transport and dye molecule adsorption.27
The variations in RE among the composite membranes (MAC2:OM1 and MAC1:OM2) highlight the importance of additive ratios. The MAC2:OM1 membrane exhibits a lower RE (81.7%) compared to MAC1:OM2, indicating that a higher concentration of AC is crucial for optimal dye removal. This observation underscores the dominant role of AC in dye adsorption.81 Similarly, the MAC1:OM2 membrane shows reduced RE (73.6%), suggesting that while OM contributes to dye removal, its impact is secondary to that of AC.
The data indicates that the dominant mechanism driving the dye removal is adsorption, which is directly related to the high surface area provided by the AC additive.82 The OM additive provides additional adsorption sites and contributes to the increased porosity of the membrane, thus indirectly increasing the adsorption capacity.83
3.10 Permeate flux of pure water and MB dye
Membrane performance is fundamentally evaluated by its ability to maintain high permeability while effectively rejecting contaminants. Table 5 and Fig. 16 present a comparative analysis of PWF and MB dye flux across the membranes studied. A clear reduction in permeate flux is observed for all membranes when switching from pure water to MB dye solution, an expected outcome attributed to the larger molecular size, high viscosity and interactive behaviour of dye molecules, which tend to adsorb onto the membrane surface and partially obstruct the pores.84 This interaction results in increased resistance to flow, thereby lowering the permeate flux during dye filtration.| Membrane | Flux of water (L m−2 h−1) | Flux of MB dye (L m−2 h−1) |
|---|---|---|
| MPVA | 55.4 | 47.5 |
| MAC1:OM1 | 67.5 | 64.8 |
| MAC2:OM1 | 65.3 | 61.8 |
| MAC1:OM2 | 64.1 | 60.2 |
Among the membranes tested, the MPVA membrane exhibited the most pronounced flux drop, decreasing from 55.4 L m−2 h−1 (pure water) to 47.5 L m−2 h−1 (MB dye), reflecting a 14.3% reduction. This significant decline indicates a high susceptibility to fouling and pore blockage by the MB dye molecules, likely due to the absence of surface functional groups capable of resisting dye adsorption.
In contrast, the composite membranes incorporating AC and OM showed notable improved flux retention. For instance, MAC1:OM1 recorded a minimal flux drop of only 4.0% from 67.5 to 64.8 L m−2 h−1 demonstrating the membrane's enhanced resistance to fouling. Similarly, MAC2:OM1 and MAC1:OM2 showed relatively lower flux losses of 5.4% and 6.1%, respectively. These improvements suggest that the combination of negatively charged AC and hydrophilic OM creates less prone to dye adsorption and blockage.
The observed flux decline in MB dye filtration can be attributed to several mechanisms. MB dye molecules, being larger and more complex than water, tend to interact with the membrane surface, leading to partial pore blockage, adsorption onto active sites, and concentration polarization effects. This phenomenon restricts water transport and contributes to the observed permeability drop.43,84
However, the presence of AC and OM in the composite membranes appears to mitigate these effects. The layered structure and high surface area of AC, coupled with the hydrophilic and porous nature of OM, enhance water transport while reducing dye accumulation on the membrane surface. This synergy not only improves fouling resistance but also contributes to maintaining a high permeate flux under dye filtration conditions.
3.11 Flux decline in MPVA and composite membranes
The phenomenon of flux drop in membrane filtration refers to the reduction in water permeability after exposure to MB dye, and it serves as a critical indicator of a membrane's resistance to fouling.85 To assess the flux drop and recovery capacity of the membrane after MB dye filtration, a two-hour post-filtration step using pure water was conducted. This step allows for the evaluation of the membrane's fouling resistance and its ability to recover permeability after exposure to dye contaminants. The pure water flux measured after this cleaning phase is compared to the initial pure water flux measured before dye filtration and the percentage reduction is known as the flux drop (reversible fouling).86Fig. 17 depicts the percentage drop in flux for the membranes. The MPVA membrane exhibited a significant flux drop of 48%, with its initial pure water flux decreasing from 55.4 L m−2 h−1 to 39.0 L m−2 h−1 post-MB dye filtration and rinsing, indicating that a substantial amount of dye fouling was not removed during the acetone rinse. Conversely, the best performing composite membrane MAC1:OM1 demonstrated a much smaller drop of 13.4% from 67.5 to 58.8 L m−2 h−1 suggesting that the presence of hydrophilic and negatively charged functional groups from AC and OM effectively prevented strong dye adhesion and facilitated easier cleaning. This test confirms that the composite membranes not only perform better during filtration but also exhibit superior fouling resistance and recovery behaviour compared to the unmodified MPVA membrane.3.12 Fouling behaviour of the prepared membranes
Fouling remains one of the most critical challenges in polymer-based water treatment, as it directly affects long-term filtration performance, membrane lifespan, and operational costs.87 In this study, the fouling behaviour of the fabricated membranes was evaluated using the flux recovery ratio (FRR) and the corresponding fouling percentage, as detailed in Table 6 and Fig. 18. These values offer insight into each membrane's resilience against irreversible fouling by MB dye and its potential for reuse.| Membrane | PWF (L m−2 h−1) | FRR (%) | Fouling (%) |
|---|---|---|---|
| MPVA | 55.4 | 70.4 | 29.6 |
| MAC1:OM1 | 67.5 | 87.1 | 12.9 |
| MAC2:OM1 | 65.3 | 83.2 | 16.8 |
| MAC1:OM2 | 64.1 | 82.4 | 17.6 |
 | ||
| Fig. 18 Antifouling performance of MPVA and composite PVA/AC:OM membranes based on PWF, FRR fouling percentages. | ||
The MPVA exhibited the lowest FRR of 70.4%, indicating a relatively high fouling tendency (29.6% fouling). This result aligns with expectations, as MPVA lacks surface-modifying agents or fillers that could improve antifouling resistance. The absence of active sites and hydrophilic reinforcements likely makes it more prone to poor dye adsorption and pore-clogging.
In contrast, the composite membranes showed remarkable improvements. The MAC1:OM1 membrane, achieved the highest FRR at 87.1%, corresponding to a minimal fouling percentage of 12.9%. This reflects a more hydrophilic and smoother surface, resulting from the combined effects of AC's layered structure and OM's mucilage content, both of which are known to hinder dye attachment and promote surface hydration.43,83
MAC2:OM1 and MAC1:OM2 also performed well, with FRRs of 83.2% and 82.4%, respectively. The fouling percentages (16.8% and 17.6%) were significantly lower than the pure MPVA membrane, further demonstrating the effectiveness of these bio-derived additives in mitigating dye-induced fouling. The relatively higher fouling in MAC2:OM1 may be attributed to reduced AC content, which could compromise the membrane's rigidity and resistance to structural deformation during dye exposure. Similarly, MAC1:OM2, with lower OM content, may offer fewer hydrophilic sites, slightly diminishing its antifouling effect.
These findings clearly establish that the incorporation of AC and OM not only enhances water flux but also confers strong antifouling characteristics. The improved FRR values and reduced fouling percentages confirm that the synergistic chemistry of these additives plays a crucial role in surface hydration, dye rejection, and fouling mitigation. Ultimately, optimizing the ratio of AC and OM is vital to achieving a balance between permeability and long-term operational performance.
3.13 A comparative performance discussion of MAC1:OM1 with literature-reported membranes
To contextualize the performance of the MAC1:OM1 composite membrane within the evolving landscape of polymer-based filtration technologies, Table 7 provides summary of benchmark data from some recent membrane studies. This comparative synthesis highlights key parameters including water flux, dye rejection, porosity, and antifouling efficiency across systems utilizing both synthetic and bio-based materials.| Membrane | Flux (L m−2 h−1) | Dye removed | Removal efficiency (%) | Ref. |
|---|---|---|---|---|
| TiO2/PVA coated PVDF | 16.30 | Reactive Blue | 44.4 | 39 |
| TiO2/PVA coated PVDF | 16.30 | Rhodamine b | 45.8 | 39 |
| TiO2/PVA coated PVDF | 16.30 | Methyl orange | 47.8 | 39 |
| PES/PVA | 21.4 | Disperse dye | 97.7 | 89 |
| PES/BiFeO3 | 63.5 | Methylene blue | 97.974 | 88 |
| PVA/AC/HA | 70.3 | Methylene blue | 95.5 | 43 |
| CA/PVA | 17 | Crystal violet dye | 99.9 | 34 |
| MAC1:OM1 | 67.5 | Methylene blue | 84.3 | This research |
For example, a high-performing PES membrane integrated with 0.98 wt% PEI-grafted silica (SiO2-g-PEI) reported exceptional MB rejection of 99.99% and water flux of 140 ± 1.28 L m−2 h−1 and a FRR of 95.4%, alongside a porosity nearing 90%.33 However, this impressive performance relied on surface-grafted nanostructures and the use of a hydrophobic PES matrix, approaches that are both cost-intensive and less environmentally benign than the PVA-based system developed in this study.
Similarly, PES/BiFeO3 membranes optimised via response surface methodology (RSM) achieved 97.97% dye removal with a flux of 63.5 L m−2 h−1 under optimized operational parameters.88 Although comparable in rejection, the MAC1:OM1 membrane exceeds this system's flux under similar pressure conditions, leveraging biowaste-derived fillets and avoiding the use of metal oxide nanoparticles or chemical optimisation routes.
In another study, a PPSU membrane loaded with 0.1 wt% GO-WO2.89 composite delivered a notable high flux 636.01 L m−2 h−1 and 82.86% BSA rejection.32 While this flux value far exceeds those observed in our study, it was achieved under ultrafiltration conditions, picturing larger pore sizes and lower dye concentration than those in our tests. Additionally, our membrane achieved a superior MB dye rejection of 84.3% and a more robust FRR of 87.1% compared to 83–92% range in the PPSU-GO-WO2.89 system.
Closer analogues to our work include PVA-based membranes enhanced with clay and biopolymers. For instance, AC/HA–PVA membranes achieved a flux of 70.3 L m−2 h−1, 95.5% MB rejection, and an FRR of 83.9%,48 while a CNS-reinforced PVA membrane reported 71 L m−2 h−1, 95.7% rejection and an FRR of 89.8%.35 In comparison, our MAC1:OM1 membrane delivered a flux of 67.5 L m−2 h−1, porosity of 68% dye rejection of 84.3% and FRF of 87.1. While the rejection values are slightly lower, our system achieves this performance using only green, low-cost fillers and a simpler fabrication process without synthetic crosslinkers or polymer blends.
Taken all together, this comparative evaluation underscores that although the MAC1:OM1 membrane does not top individual performance metrics, it occupies a uniquely balanced position, delivering solid flux, consistent rejection and strong antifouling behaviour within an eco-friendly and scalable framework. The synergistic interplay between AC's, layered structure and the gelling capacity of OM enables a structural network that balances permeability with selective adsorption. This makes the membrane not merely competitive, but practical and future-forward for sustainable wastewater remediation.
3.14 Long-time performance and durability properties
Five consecutive cycles of durability and reusability testing were conducted on the best-performing membrane MAC1:OM1. The results are shown in Fig. 19 and Table 8. During the first cycle, the PWF before filtration was 67.5 L m−2 h−1 which decreased to 64.8 L m−2 h−1 during MB dye filtration and recovered to 58.8 L m−2 h−1. The membrane's flow rate in the second filtration cycle using MB dye solution was 64.8 L m−2 h−1 after a 45-minute acetone cleaning protocol involving rinsing with acetone, followed by DI–water. Subsequent cycles showed a gradual decline in flux (from 65.8 → 61.7 L m−2 h−1 before filtration), primarily due to minor pore compaction and the partial accumulation of dye residues within the membrane matrix. Nevertheless, flux recovery remained consistently above 90%, indication excellent antifouling and regeneration performance.| Cycle | PWF (L m−2 h−1) before filtration | PWF (L m−2 h−1) of MB dye | PWF (L m−2 h−1) after filtration |
|---|---|---|---|
| 1 | 67.5 | 64.8 | 58.8 |
| 2 | 65.8 | 63.2 | 57.1 |
| 3 | 64.4 | 61.3 | 56.0 |
| 4 | 63.1 | 60.3 | 55.2 |
| 5 | 61.7 | 59.1 | 54.2 |
The MAC1:OM1 membrane consistently exhibited strong durability and reusability across all five filtration cycles, demonstrating minimal structural degradation and strong self-cleaning capability. This enhanced long-term performance confirms the mechanical stability and reusability of the AC/OM-integrated PVA membrane for continuous water purification operations.
3.10 Mechanism of MB dye adsorption on composite MPVA/AC:OM membranes
The adsorption mechanism of adsorbent surfaces towards MB dye as a pollutant as shown (Fig. 20) in this study is influenced by many interactions such as electrostatic interactions, hydrogen bonding, physical adsorption and interlayer expansion/intercalation.90 These interactions facilitate the effective removal of MB dye by the MPVA/AC:OM polymer membrane composite. van der Waals forces such as π–π interactions, London dispersion forces and hydrophobic contacts between the MB dye molecules and the AC particles promote physical adsorption. Effective physical adsorption removal is made possible by the large surface area and porous structure of AC, which offer the ideal surface and numerous adsorption sites for MB molecules to momentarily adhere to ref. 91. The anionic character of the AC is partly due to the presence of SiO2 and Al2O3. The cationic dye's adsorption indicated that the positively charged dye interacted with the negatively charged Si–O– and Al–O– groups in AC, giving AC two functions: (i) it took on a negative charge and (ii) it served as a binder for coating the substrate.90PVA introduces hydroxyl groups (–OH) to the membrane structure, which can form hydrogen bonds with the (–NH2) functional groups of methylene blue.43 When hydrogen bonds are formed, the PVA matrix and the dye molecules have a stronger affinity.92 Activated clay expands between layers when it is included in the composite membrane because of its layered nature.93 This expansion allows the MB molecules to intercalate between the layers of clay. The interlayer gap increases the accessibility of dye molecules to adsorption sites, hence improving the overall effectiveness of the dye removal process. Electrostatic interactions play a key role in the removal of charged dye. The polar components of OM and the charged spots on the surface of activated clay form electrostatic interactions with the dye molecules.94 The aromatic structures of OM and MB dyes can interact with one another π–π. These interactions, which include the stacking of aromatic rings, help the dye adsorb onto the membrane. Additionally, polyphenols' hydroxyl groups enable them to engage with the aromatic rings and positive charge of MB dye.95
4. Conclusion
This study successfully engineered and evaluated green composite ultrafiltration membranes based on PVA, reinforced with AC and OM as sustainable fillers for dye–laden wastewater treatment. By employing a solution casting technique followed by urea–formaldehyde crosslinking, membranes were fabricated in varying compositions and thoroughly characterized via zeta-sizer, 1H NMR, FTIR, SEM-EDS and XRD. These techniques provide crucial insights into the structural, morphological and surface charge properties of the membranes. The incorporation of AC and OM induced synergistic effects. AC greatly enhanced the membrane porosity, mechanical stability and hydrophilicity due to its layered silicate structure and high surface area, which facilitated water transport and contaminant adsorption. OM, rich in polysaccharide and phenolic compounds, introduces functional groups such as hydroxyl and carboxyl, enhancing the membrane's antifouling capacity through increased hydrophilicity and electrostatic interactions. Performance evaluation revealed that the pristine PVA membrane (MPVA), showed moderate efficiency, with a pure PWF of 55.4 L m−2 h−1, dye removal efficiency of 52.1%, porosity was 54.6%, and antifouling property was 70.4%. In contrast, the MAC1:OM1 composite membrane exhibited the highest performance metrics, achieving a porosity of 68.0%, PWF of 67.5 L m−2 h−1, MB dye removal efficiency of 84.3% and antifouling property of 87.1%. These enhancements are attributed to the synergistic interactions between AC and OM within the PVA matrix, which not only improved surface roughness and hydrophilicity but also contributed to a stronger negative surface charge, favouring dye molecule adsorption. These findings demonstrate that biowaste-derived fillers can effectively balance flux, rejection and antifouling performance, offering a scalable and environmentally benign pathway for water purification membranes. The outcomes not only provide proof of concept for green PVA composites but also position AC–OM systems as low-cost candidates for dye wastewater remediation where fouling resistance is critical.Ethical statement
Ethical considerations with analysis are not addressed in this paper.Author contributions
The manuscript was written through the contributions of all authors. All authors have approved the final version of the manuscript.Conflicts of interest
The authors have no conflicting interests in this publication.Data availability
All relevant data supporting the findings of this study are included in the manuscript. Additional raw/processed data can be made available upon reasonable request from the corresponding author.Acknowledgements
Funding was not obtained for this study. The authors are grateful to the Central Laboratory, Kwame Nkrumah University of Science and Technology (KNUST), Kumasi-Ghana, and Regional Water and Environmental Sanitation Centre, KNUST, Kumasi-Ghana for the technical support in characterizing the materials.References
- R. P. Schwarzenbach, T. Egli, T. B. Hofstetter, U. von Gunten and B. Wehrli, Annu. Rev. Environ. Resour., 2010, 35, 109–136 Search PubMed.
- O. V. Vysokomornaya, E. Yu Kurilenko and A. A. Shcherbinina, MATEC Web. Conf., 2015, 23, 01041 Search PubMed.
- P. R. Rout, T. C. Zhang, P. Bhunia and R. Y. Surampalli, Sci. Total Environ., 2021, 753, 141990 Search PubMed.
- D. Fytili and A. Zabaniotou, Renewable Sustainable Energy Rev., 2008, 12, 116–140 CrossRef CAS.
- S. Khalid, M. Shahid, N. Natasha, I. Bibi, T. Sarwar, A. H. Shah and N. K. Niazi, Int. J. Environ. Res. Public Health, 2018, 15, 895 CrossRef PubMed.
- M. A. Hanjra, J. Blackwell, G. Carr, F. Zhang and T. M. Jackson, Int. J. Hydrogen Environ. Health, 2012, 215, 255–269 CrossRef PubMed.
- E. S. Agorku, A. Kangmennaa, B. Y. Danu, F. K. Ampong and R. B. Voegborlo, Next Sustainability, 2025, 5, 100069 CrossRef.
- A. Azanaw, B. Birlie, B. Teshome and M. Jemberie, Case Stud. Chem. Environ. Eng., 2022, 6, 100230 CrossRef CAS.
- L. P. Amenorfe, E. S. Agorku, F. Sarpong and R. B. Voegborlo, Sci. Afr., 2022, 17, e01359 Search PubMed.
- G. J. Nohynek, R. Fautz, F. Benech-Kieffer and H. Toutain, Food Chem. Toxicol., 2004, 42, 517–543 CrossRef CAS PubMed.
- A. Baruah, V. Chaudhary, R. Malik and V. K. Tomer, Nanotechnology in Water and Wastewater Treatment, Elsevier, 2019, pp. 337–368 Search PubMed.
- V. G. Gude, Rev. Environ. Sci. Biotechnol., 2017, 16, 591–609 CrossRef.
- E. Santoso, R. Ediati, Y. Kusumawati, H. Bahruji, D. O. Sulistiono and D. Prasetyoko, Mater. Today Chem., 2020, 16, 100233 CrossRef CAS.
- H. Peng and J. Guo, Environ. Chem. Lett., 2020, 18, 2055–2068 CrossRef CAS.
- N. Voutchkov, Desalination, 2010, 261, 354–364 CrossRef CAS.
- K. Castro and R. Abejón, Membranes, 2024, 14, 180 CrossRef CAS PubMed.
- S. Abd Hamid, M. Shahadat, B. Ballinger, S. Farhan Azha, S. Ismail, S. Wazed Ali and S. Ziauddin Ahammad, J. Saudi Chem. Soc., 2020, 24, 785–798 CrossRef CAS.
- A. Saravanan, P. Thamarai, P. S. Kumar and G. Rangasamy, Chemosphere, 2022, 308, 136368 CrossRef CAS PubMed.
- A. A. Sapalidis, Symmetry, 2020, 12, 960 CrossRef CAS.
- H. Zheng, Y. Du, J. Yu, R. Huang and L. Zhang, J. Appl. Polym. Sci., 2001, 80, 2558–2565 CrossRef CAS.
- B. Ye, Y. Li, Z. Chen, Q.-Y. Wu, W.-L. Wang, T. Wang and H.-Y. Hu, Water Res., 2017, 124, 381–387 CrossRef CAS PubMed.
- G. S. A. Suleiman, X. Zeng, R. Chakma, I. Y. Wakai and Y. Feng, Polym. Adv. Technol., 2024, 35 DOI:10.1002/pat.6327.
- S. Abd Hamid, M. Shahadat, B. Ballinger, S. Farhan Azha, S. Ismail, S. Wazed Ali and S. Ziauddin Ahammad, J. Saudi Chem. Soc., 2020, 24, 785–798 CrossRef CAS.
- H. Ouaddari, A. Karim, B. Achiou, S. Saja, A. Aaddane, J. Bennazha, I. El Amrani El Hassani, M. Ouammou and A. Albizane, J. Environ. Chem. Eng., 2019, 7, 103268 CrossRef CAS.
- T. S. Anirudhan, F. Shainy and J. R. Deepa, Chem. Ecol., 2019, 35, 235–255 CrossRef CAS.
- A. Kangmennaa, S. Acquah, R. B. Forkuo, J. K. Adusei, G. A. Atongo, F. A. Amarh, F. Opoku and E. S. Agorku, J. Dispersion Sci. Technol., 2025, 46, 807–820 CrossRef CAS.
- C. Lavado-Meza, M. C. Fernandez-Pezua, F. Gamarra-Gómez, E. Sacari-Sacari, J. Angeles-Suazo and J. Z. Dávalos-Prado, Molecules, 2023, 28, 4451 CrossRef CAS PubMed.
- H. Chahdoura, P. Morales, J. C. M. Barreira, L. Barros, V. Fernández-Ruiz, I. C. F. R. Ferreira and L. Achour, LWT – Food Sci. Technol., 2015, 64, 446–451 Search PubMed.
- M. Choudhary, M. B. Ray and S. Neogi, Sep. Purif. Technol., 2019, 209, 714–724 CrossRef CAS.
- N. Adjeroud-Abdellatif, Y. Hammoui, A. Boudria, S. Agab, F. Choulak, J.-P. Leclerc, B. Merzouk and K. Madani, Int. J. Environ. Anal. Chem., 2022, 102, 5822–5846 CrossRef CAS.
- J. A. Figueirôa, G. U. Menezes Novaes, H. de Souza Gomes, V. L. M. de Morais Silva, D. de Moraes Lucena, L. M. R. Lima, S. A. de Souza, L. G. F. C. Viana, L. A. Rolim, J. R. G. da Silva Almeida, A. P. de Oliveira and J. P. Gomes, Heliyon, 2021, 7, e07292 CrossRef PubMed.
- R. M. Al-Maliki, Q. F. Alsalhy, S. Al-Jubouri, A. A. AbdulRazak, M. A. Shehab, Z. Németh, K. Hernadi and H. Sh. Majdi, Membranes, 2023, 13, 269 Search PubMed.
- D. A. Hussein Al-Timimi, Q. F. Alsalhy and A. A. AbdulRazak, Alexandria Eng. J., 2023, 66, 167–190 CrossRef.
- E. S. Mansor, H. Abdallah and A. M. Shaban, J. Environ. Chem. Eng., 2020, 8, 103706 Search PubMed.
- E. Nyabi, R. T. Agbetsi, G. Addai, F. Opoku, M. Toku, R. B. Voegborlo and E. S. Agorku, Next Sustainability, 2025, 5, 100136 CrossRef.
- N. Rosman, W. N. Wan Salleh, J. Jaafar, Z. Harun, F. Aziz and A. F. Ismail, Catalysts, 2022, 12, 209 Search PubMed.
- F. Medhat Bojnourd and M. Pakizeh, Appl. Clay Sci., 2018, 162, 326–338 Search PubMed.
- M. Shakak, R. Rezaee, A. Maleki, A. Jafari, M. Safari, B. Shahmoradi, H. Daraei and S.-M. Lee, Environ. Technol. Innov., 2020, 17, 100529 Search PubMed.
- S. Sakarkar, S. Muthukumaran and V. Jegatheesan, Chemosphere, 2020, 257, 127144 CrossRef CAS PubMed.
- E. Pavón and M. D. Alba, Prog. Nucl. Magn. Reson. Spectrosc., 2021, 124–125, 99–128 Search PubMed.
- D. S. Kim, I. C. Park, H. Il Cho, D. H. Kim, G. Y. Moon, H. K. Lee and J. W. Rhim, J. Ind. Eng. Chem., 2009, 15, 265–269 Search PubMed.
- A. Mittal, S. Garg, D. Kohli, M. Maiti, A. K. Jana and S. Bajpai, Carbohydr. Polym., 2016, 151, 926–938 CrossRef CAS PubMed.
- A. A. Oduro, S. A. B. Biney, E. S. Nortey, A. Kangmennaa, R. B. Forkuo and E. S. Agorku, J. Appl. Polym. Sci., 2025, 142, e56701 Search PubMed.
- M. Cao and H. Zhang, J. Sustainable Cem.-Based Mater., 2023, 12, 1322–1333 CrossRef CAS.
- S. F. Azha, L. Sellaoui, M. S. Shamsudin, S. Ismail, A. Bonilla-Petriciolet, A. Ben Lamine and A. Erto, Chem. Eng. J., 2018, 351, 221–229 Search PubMed.
- E. S. Mansor, E. A. Ali and A. M. Shaban, Chem. Eng. J., 2021, 407, 127175 CrossRef CAS.
- A. Kangmennaa, S. Acquah, R. B. Forkuo, J. K. Adusei, G. A. Atongo, F. A. Amarh, F. Opoku and E. S. Agorku, J. Dispers Sci. Technol., 2024, 1–14 Search PubMed.
- A. A. Oduro, S. A. B. Biney, E. S. Nortey, A. Kangmennaa, R. B. Forkuo and E. S. Agorku, J. Appl. Polym. Sci., 2025, e56701 Search PubMed.
- K. Ravikumar and J. Udayakumar, Chem. Ecol., 2020, 36, 270–291 Search PubMed.
- P. Tarte, Spectrochim. Acta, Part A, 1967, 23, 2127–2143 CrossRef CAS.
- C.-A. Teacă, R. Bodîrlău and I. Spiridon, Carbohydr. Polym., 2013, 93, 307–315 CrossRef PubMed.
- A. Mittal, S. Garg, D. Kohli, M. Maiti, A. K. Jana and S. Bajpai, Carbohydr. Polym., 2016, 151, 926–938 CrossRef CAS PubMed.
- A. Mittal, S. Garg, D. Kohli, M. Maiti, A. K. Jana and S. Bajpai, Carbohydr. Polym., 2016, 151, 926–938 CrossRef CAS PubMed.
- M. Cortez-Valadez, L. P. Ramírez-Rodríguez, J.-G. Bocarando-Chacon, M. Flores-Acosta, S. Velumani and R. Ramírez-Bon, NANO, 2015, 10, 1550100 CrossRef CAS.
- Q. Wang, W. Lin, S. Chou, P. Dai and X. Huang, Water Res., 2023, 236, 119943 CrossRef CAS PubMed.
- J. J. Sahlin and N. A. Peppas, J. Appl. Polym. Sci., 1997, 63, 103–110 CrossRef CAS.
- M. Ghanipour and D. Dorranian, J. Nanomater., 2013, 897043 CrossRef.
- H. G. Dill, Earth Sci. Rev., 2016, 161, 16–129 CrossRef CAS.
- G. Jovanovski and P. Makreski, Maced. J. Chem. Chem. Eng., 2016, 35, 125 CrossRef CAS.
- M. Ghanipour and D. Dorranian, J. Nanomater., 2013, 897043 CrossRef.
- D. de la Fuente, J. Alcántara, B. Chico, I. Díaz, J. A. Jiménez and M. Morcillo, Corros. Sci., 2016, 110, 253–264 CrossRef CAS.
- A. Kangmennaa, S. Acquah, R. B. Forkuo, J. K. Adusei, G. A. Atongo, F. A. Amarh, F. Opoku and E. S. Agorku, J. Dispers Sci. Technol., 2024, 1–14 Search PubMed.
- D. de la Fuente, J. Alcántara, B. Chico, I. Díaz, J. A. Jiménez and M. Morcillo, Corros. Sci., 2016, 110, 253–264 CrossRef CAS.
- O. S. Lezova, D. V. Myasnikov, O. A. Shilova, A. G. Ivanova and S. I. Selivanov, Int. J. Hydrogen Energy, 2022, 47, 4846–4853 CrossRef CAS.
- Y. Zhang, P. C. Zhu and D. Edgren, J. Polym. Res., 2010, 17, 725–730 CrossRef CAS.
- J. Zhang, F. Zhang, Y. Luo and H. Yang, Process Biochem., 2006, 41, 730–733 CrossRef CAS.
- K. Buruga, H. Song, J. Shang, N. Bolan, T. K. Jagannathan and K.-H. Kim, J. Hazard. Mater., 2019, 379, 120584 CrossRef CAS PubMed.
- C. Rattanakawin and R. Hogg, Colloids Surf., A, 2001, 177, 87–98 CrossRef CAS.
- S. Samal, Powder Technol., 2020, 366, 43–51 CrossRef CAS.
- H. B. Park, J. Kamcev, L. M. Robeson, M. Elimelech and B. D. Freeman, Science, 2017, 356 DOI:10.1126/science.aab0530.
- C. Giannini, M. Ladisa, D. Altamura, D. Siliqi, T. Sibillano and L. De Caro, Crystals, 2016, 6, 87 CrossRef.
- J. Rodriguez-Loya, M. Lerma and J. L. Gardea-Torresdey, Micromachines, 2023, 15, 24 CrossRef PubMed.
- X. Liu and M. Bai, Adv. Powder Technol., 2017, 28, 836–841 CrossRef CAS.
- N. Nasrollahi, L. Ghalamchi, V. Vatanpour, A. Khataee and M. Yousefpoor, J. Ind. Eng. Chem., 2022, 109, 100–124 Search PubMed.
- A. Z. Khalifa, Ö. Cizer, Y. Pontikes, A. Heath, P. Patureau, S. A. Bernal and A. T. M. Marsh, Cem. Concr. Res., 2020, 132, 106050 Search PubMed.
- M. Swaminathan and G. Swaminathan, Sep. Purif. Rev., 2024, 1–21 Search PubMed.
- M. C. Fernández-Martínez, C. Jiménez-Martínez, M. R. Jaime-Fonseca and L. Alamilla-Beltrán, Polymers, 2024, 16, 3383 CrossRef PubMed.
- F. Mannai, H. Elhleli, M. Ammar, R. Passas, E. Elaloui and Y. Moussaoui, Ind. Crops Prod., 2018, 126, 347–356 CrossRef CAS.
- B. Ghanbarian, A. G. Hunt and H. Daigle, Water Resour. Res., 2016, 52, 2045–2058 Search PubMed.
- A. Amari, H. Gannouni, M. I. Khan, M. K. Almesfer, A. M. Elkhaleefa and A. Gannouni, Appl. Sci., 2018, 8, 2302 Search PubMed.
- G. George, A. M. Ealias and M. P. Saravanakumar, Environ. Sci. Pollut. Res., 2024, 31, 12748–12779 CrossRef CAS PubMed.
- Y. Shi, Q. Chang, T. Zhang, G. Song, Y. Sun and G. Ding, J. Environ. Chem. Eng., 2022, 10, 108639 Search PubMed.
- N. Barka, K. Ouzaouit, M. Abdennouri and M. El Makhfouk, J. Taiwan Inst. Chem. Eng., 2013, 44, 52–60 CrossRef CAS.
- L. Zhou, G. Xiao, Y. He, J. Wu, H. Shi, F. Zhong, X. Yin, Z. Li and J. Chen, J. Hazard. Mater., 2021, 413, 125379 CrossRef CAS PubMed.
- C. Thamaraiselvan and M. Noel, Crit. Rev. Environ. Sci. Technol., 2015, 45, 1007–1040 CrossRef CAS.
- K. H. Chu, S. S. Yoo, Y. Yoon and K. B. Ko, Sep. Purif. Technol., 2015, 151, 147–154 CrossRef CAS.
- N. M. A. Omar, M. H. D. Othman, Z. S. Tai, T. A. Kurniawan, M. H. Puteh, J. Jaafar, M. A. Rahman, A. F. Ismail, N. Rajamohan, H. Abdullah and K. Y. Wong, J. Water Process Eng., 2024, 62, 105399 CrossRef.
- S. K. Jawad, K. T. Rashid, M. A. Toma, A. A. Abdul Razak, M. A. Shehab, M. Y. Ghadhban, M. Al-lami, A. Mahmood, H. H. Mohammed, A. A. Hmood, M. F. A. Al-Ogaili and S. Alsarayefi, Results Eng., 2025, 26, 105246 CrossRef CAS.
- J. Babu and Z. V. P. Murthy, Sep. Purif. Technol., 2017, 183, 66–72 CrossRef CAS.
- S. Yu, X. Wang, Z. Chen, J. Wang, S. Wang, T. Hayat and X. Wang, J. Hazard. Mater., 2017, 321, 111–120 CrossRef CAS PubMed.
- E. Santoso, R. Ediati, Y. Kusumawati, H. Bahruji, D. O. Sulistiono and D. Prasetyoko, Mater. Today Chem., 2020, 16, 100233 CrossRef CAS.
- Y. Gao, P. Cai, L. Zhong, R. Zhang, X. Hou, X. Ren, J. Wang, X. Chu, Y. Lu and Z. Zhou, Int. J. Biol. Macromol., 2024, 254, 127886 CrossRef CAS PubMed.
- M. C. Almandoz, C. L. Pagliero, N. A. Ochoa and J. Marchese, Ceram. Int., 2015, 41, 5621–5633 CrossRef CAS.
- D. Lan, H. Zhu, J. Zhang, S. Li, Q. Chen, C. Wang, T. Wu and M. Xu, Chemosphere, 2022, 293, 133464 Search PubMed.
- Q. Zhou, Y. Hu, R. Zhao, Y. Xu, R. Rao and N. Li, Colloids Surf., A, 2024, 698, 134591 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |














