Amelioration of diet-induced hyperglycemia by wheat peptides in a multi-target manner: integrated modulation of inflammation, lipid metabolism, gut microbiota, and DPP-IV inhibition
Fei
Shen
acf,
Qian
Deng
a,
Yishan
Song
bc,
Guanghua
He
a,
Xiaojun
Chu
f,
Minjie
Zhao
 c,
Juan
Du
e,
Fengqin
Feng
c,
Juan
Du
e,
Fengqin
Feng
 c,
Xi
Zhang
*b and
Hao
Zhong
c,
Xi
Zhang
*b and
Hao
Zhong
 *d
*d
aSchool of Biological and Chemical Engineering, Zhejiang University of Science & Technology, Hangzhou 310023, China
bCollege of Traditional Chinese Medicine, Yunnan University of Chinese Medicine, Kunming, 650500, China. E-mail: zhonghao@zjut.edu.cn; zhangxi1030@hotmail.com; Fax: +011 86 18888915279; Fax: +011 86-571-88982163; Tel: +011 86 18888915279 Tel: +011 86-571-88982163
cCollege of Biosystems Engineering and Food Science, Zhejiang University, Hangzhou, 310058, China
dCollege of Food Science and Technology, Zhejiang University of Technology, Hangzhou 310014, China
eHangzhou Kangyuan Food Science & Technology Co., Ltd, Hangzhou 310012, China
fBeingmate (Hangzhou) Food Research Institute Co., Ltd, Hangzhou, 310000, China
First published on 15th November 2025
Abstract
Wheat peptides (WP) have been claimed to have the potential to regulate metabolism and effectively prevent/mitigate gut microbiota dysbiosis. However, many studies into the effects of WP on hyperglycemia have provided conflicting findings, and the underlying mechanism has been elusive. In this study, WP intervention (50–1000 mg kg−1) dose-dependently attenuated high fat diet (HFD)-induced weight gain, fasting hyperglycemia, glucose intolerance and insulin resistance. WP suppressed systemic inflammation by normalizing serum levels of lipopolysaccharide (LPS), tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β), while concurrently reducing adipocyte hypertrophy and hepatic steatosis. Serum lipid profiles were improved, with significant reductions in total cholesterol (TC) and triglycerides (TG), though low-density lipoprotein (LDL) and high-density lipoprotein (HDL) levels remained unaltered. Although gut microbiota α-diversity was unaffected, WP modulated microbial composition by decreasing the Firmicutes/Bacteroidota ratio and enriching beneficial genera, including Bifidobacterium and Lactobacillus. Metabolomic analyses further revealed that WP-restored metabolic homeostasis is associated with upregulating functional lipids [PE(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1/20
1/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), PG(18
3), PG(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0/20
0/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), and PS(22
4), and PS(22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6/22
6/22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)] and the tryptophan metabolite 5-HIAA, all of which exhibited inverse correlations with indices of metabolic dysfunction. Critically, the WP-derived peptides LPQ and LPQF, characterized by a proline residue at the second position (Pro2), exhibited potent dipeptidyl peptidase-IV (DPP-IV) inhibitory activity, mediated by high-affinity binding involving van der Waals forces, hydrogen bonding, and electrostatic interactions. Furthermore, in hyperglycemic zebrafish models, LPQ/LPQF (0.1–5 μg mL−1) normalized glycemic levels. Collectively, WP exerts hypoglycemic effects through potentially synergistic mechanisms: (i) suppression of inflammation, (ii) restoration of functional lipid and tryptophan metabolic pathways, (iii) modulation of the gut microbiota toward a beneficial profile, and (iv) DPP-IV inhibition by structurally optimized peptides. These findings highlight WP's therapeutic potential for metabolic syndrome, underscoring its utility as a multifaceted intervention for metabolic dysregulation.
1)] and the tryptophan metabolite 5-HIAA, all of which exhibited inverse correlations with indices of metabolic dysfunction. Critically, the WP-derived peptides LPQ and LPQF, characterized by a proline residue at the second position (Pro2), exhibited potent dipeptidyl peptidase-IV (DPP-IV) inhibitory activity, mediated by high-affinity binding involving van der Waals forces, hydrogen bonding, and electrostatic interactions. Furthermore, in hyperglycemic zebrafish models, LPQ/LPQF (0.1–5 μg mL−1) normalized glycemic levels. Collectively, WP exerts hypoglycemic effects through potentially synergistic mechanisms: (i) suppression of inflammation, (ii) restoration of functional lipid and tryptophan metabolic pathways, (iii) modulation of the gut microbiota toward a beneficial profile, and (iv) DPP-IV inhibition by structurally optimized peptides. These findings highlight WP's therapeutic potential for metabolic syndrome, underscoring its utility as a multifaceted intervention for metabolic dysregulation.
1 Introduction
Diabetes mellitus has emerged as one of the most severe public health crises of the 21st century, with a global prevalence exceeding 537 million individuals. Type 2 diabetes mellitus (T2DM) accounts for over 90% of cases, and its incidence continues to rise exponentially.1 Obesity acts as a central driver, disrupting glucose homeostasis and inducing chronic hyperglycemia by promoting insulin resistance (IR) and pancreatic β-cell dysfunction.2 This disease imposes substantial burdens on national healthcare resources and socioeconomic development, while also driving the high incidence of complications such as cardiovascular disease, nephropathy, and retinopathy.3 Current T2DM management relies on pharmacological interventions combined with lifestyle modifications. Although traditional agents like metformin (MET) remain first-line therapy, their efficacy as monotherapy is often limited. While DPP-4 inhibitors (e.g., saxagliptin) enhance incretin hormone (GLP-1/GIP) activity, they carry risks of headache and infection.4,5 Although newer dual receptor agonists (e.g., tirzepatide) offer synergistic glycemic and weight control, their high cost and requirement for injectable administration present significant barriers. Consequently, the development of effective, safe, and orally administered alternative therapies represents an urgent unmet need.Bioactive peptides, naturally occurring in food or derived from food proteins through partial hydrolysis during digestion or processing, typically contain a free carboxyl group and a free amino group, although exceptions such as prolyl and pyroglutamyl peptides lacking a free amino group have been identified following enzymatic hydrolysis.6 These peptides exhibit a wide range of biological functions, including antimicrobial, anticancer, anti-obesity, and antidiabetic effects, positioning them as promising dietary supplements or nutraceuticals for managing chronic diseases.7 Their potential antidiabetic mechanisms include inhibiting key enzymes like α-glucosidase and α-amylase (as demonstrated by peptides from germinated soybean and Spirulina platensis, thereby reducing carbohydrate hydrolysis and subsequent glucose absorption) and suppressing dipeptidyl peptidase-IV (DPP-IV) activity (as shown by milk protein- and Atlantic salmon-derived peptides, which preserve incretin hormones to exert antihyperglycemic effects).8 Additional mechanisms involve stimulating insulin synthesis in pancreatic β-cells, enhancing insulin sensitivity in peripheral tissues, reducing adiposity, modulating fatty acid metabolism, alleviating inflammation, and modulating gut microbiota.8 While wheat germ-derived bioactive peptides are recognized for their antibacterial, antitumor, immunomodulatory, antioxidant, and anti-inflammatory properties,9 the potential role of wheat peptides (WP) in mitigating hyperglycemia and diabetes remains largely unexplored.
Among the numerous targets for glycemic control, dipeptidyl peptidase-IV (DPP-IV) represents an ideal target for peptide-based therapeutics due to its direct role in regulating incretin hormone degradation. Compared to synthetic inhibitors (e.g., sitagliptin), DPP-IV inhibitory peptides derived from plant proteins have garnered increasing research interest owing to their efficacy and safety profiles. Specific peptides demonstrating DPP-IV inhibitory activity include EPAAV from soybean, NPLL from lupin, and APFTVV from quinoa.10 Furthermore, enzymatic hydrolysis of defatted rice bran using Umamizyme G successfully yielded a DPP-IV inhibitory hydrolysate (IC50 = 2.3 ± 0.1 mg mL−1), with IP and LP identified as contributing peptides.11
This study investigates the identification and mechanisms of action of wheat peptides (WP) in hyperglycemia. We demonstrate their efficacy in ameliorating insulin resistance and organ damage within HFD mouse models. Gut microbiome analysis and serum metabolomics were employed to elucidate the underlying mechanisms. Furthermore, we aim to identify the primary functional peptide sequences responsible for the observed hypoglycemic effects.
2 Materials and methods
2.1 Materials
Wheat peptides (WP) were provided by Hangzhou Kangyuan Biotechnology Co., Ltd (Hangzhou, China). The WP were prepared by enzymatic hydrolysis of wheat gluten. Hydrolysis was performed in a stepwise manner using alkaline, neutral, and flavor proteases. The resulting hydrolysate was then centrifuged, and the supernatant was purified through cation resin adsorption, followed by nanofiltration, concentration, and lyophilization. Its primary composition is detailed in SI Table S1, the molecular weight distribution in Table S2, and the amino acid profile in Table S3. Standard rodent chow was purchased from Shenzhen Maohua Biotechnology Co., Ltd (Shenzhen, China). A purified 45% high-fat diet (MD12032) was supplied by Jiangsu Medisen Biotechnology Co., Ltd (Jiangsu, China). Peptides LPQ and LPQF (purity ≥ 98%, confirmed by HPLC) were synthesized by GL Biochem (Shanghai) Ltd (Shanghai, China). Glucose was obtained from Yunnan Kunming Yirun Food Co., Ltd (Kunming, China). Assay kits for triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C) were acquired from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). Enzyme-linked immunosorbent assay (ELISA) kits for lipopolysaccharide (LPS), tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), interleukin-6 (IL-6), ghrelin, and dipeptidyl peptidase-IV (DPP-IV) were purchased from Wuhan GeneMei Biotechnology Co., Ltd (Wuhan, China).2.2 Investigating the antihyperglycemic effects of WP in an HFD-induced hyperglycemic mouse model
2.3 Gut microbiota analysis
The gut microbiota analysis was performed according to our previously described methods.14,16,17 Fresh fecal samples were collected from mice at week 8 of intervention into sterile tubes and preserved at −80 °C. Microbial genomic DNA was extracted. The V3–V4 hypervariable region of the bacterial 16S rRNA gene was amplified using primers 341F (5′-ACTCCTACGGGRSGCAGCAG-3′) and 806R (5′-GGACTACVVGGGTATCTAATC-3′). Amplicons were purified and sequenced on the Illumina MiSeq platform (performed by Majorbio Bio-Pharm Technology Co., Ltd). Raw sequences were processed into operational taxonomic units (OTUs) at a 97% similarity threshold using UPARSE. α-Diversity indices (Shannon, Simpson, ACE, and Chao1) were calculated in QIIME, while β-diversity was assessed using unweighted/weighted UniFrac distances. Taxonomic classification and visualization (including heatmaps of dominant genera) were conducted using the Majorbio Cloud Platform.2.4 Serum non-targeted metabolomics analysis
Serum samples from the NC, MC, and WP-M groups underwent metabolomic profiling following established protocols.18 Metabolite separation was achieved using an Agilent 1290 UHPLC system equipped with an ACQUITY UPLC BEH Amide column (1.7 μm, 2.1 × 100 mm), coupled to a TripleTOF 6600 mass spectrometer (AB Sciex). Metabolites were identified by matching exact masses and MS/MS spectra against a commercial database (Shanghai Applied Protein Technology). Raw data were normalized to the total peak intensity and subjected to multivariate analysis using SIMCA-P + (v14.1). Partial least squares discriminant analysis (PLS-DA) models were constructed to distinguish metabolic profiles. Key metabolic pathways mediating the antihyperglycemic effects of wheat peptides were identified through KEGG pathway analysis. All data processing utilized the Majorbio Cloud Platform.2.5 Peptide analysis
In vitro simulated gastrointestinal digestion of wheat peptides was performed, followed by LC-MS/MS identification of digestion-resistant peptides. Molecular docking was employed to investigate their interaction mechanisms with DPP-IV and screen high-potential inhibitory peptides.(1) In vitro digestion: pre-warmed (37 °C) WP solution (10 mL) was mixed with isotonic saline (9 mg mL−1 NaCl, 37 °C). After adjusting the pH to 2.0 with 1 M HCl, gastric digestion was initiated by adding 4 mL of simulated gastric fluid (0.4 g pepsin in 0.01 M HCl). The mixture was incubated at 37 °C with agitation (120 rpm) for 2 h. The pH was adjusted to 6.5, and 2 mL of aliquots were transferred to centrifuge tubes. Intestinal digestion was performed by adding 4 mL of simulated intestinal fluid (a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 2 mg mL−1 of trypsin and 12 mg mL−1 of bile salts in 0.1 M NaHCO3), adjusting the pH to 7.0, and incubating (37 °C, 120 rpm, 2 h). All procedures were conducted under dark and anaerobic conditions. Digested samples were lyophilized and stored at −80 °C.
1 mixture of 2 mg mL−1 of trypsin and 12 mg mL−1 of bile salts in 0.1 M NaHCO3), adjusting the pH to 7.0, and incubating (37 °C, 120 rpm, 2 h). All procedures were conducted under dark and anaerobic conditions. Digested samples were lyophilized and stored at −80 °C.
(2) LC-MS/MS identification: pre- and post-digestion samples were dissolved in NH4HCO3 buffer, reduced with dithiothreitol (56 °C, 1 h), and alkylated with iodoacetamide (40 min, dark). After desalting and solvent evaporation, peptides were reconstituted in mobile phase A (0.1% formic acid).
The chromatographic separation employed an Acclaim PepMap RPLC C18 analytical column (150 × 0.15 mm, 3 μm particle size) maintained at 37 °C. Mobile phases consisted of 0.1% (v/v) aqueous formic acid (A) and 80% (v/v) acetonitrile containing 0.1% (v/v) formic acid (B), delivered at 600 nL min−1. A multi-step gradient profile was executed: initial conditions at 4% B were linearly increased to 8% B over 2 minutes, followed by a ramp to 40% B from 2–45 minutes. Subsequent gradients elevated B to 60% at 55 minutes and 95% at 56 minutes, with final conditions maintained until 66 minutes.
Mass spectrometric analysis was performed on an Orbitrap platform using data-dependent acquisition. Full MS scans (m/z 100–1500) were acquired at 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 resolution (FWHM at m/z 200) with a 100 ms maximum injection time and a 3e6 AGC target. For MS/MS acquisition, the top 20 most intense precursors underwent HCD fragmentation at 28% normalized collision energy, with product ions analyzed at 17
000 resolution (FWHM at m/z 200) with a 100 ms maximum injection time and a 3e6 AGC target. For MS/MS acquisition, the top 20 most intense precursors underwent HCD fragmentation at 28% normalized collision energy, with product ions analyzed at 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 resolution using a 50 ms maximum injection time and a 1e5 AGC target. Precursors were isolated with a 1.6 m/z window, and dynamic exclusion was set to 30 s. All detected peptides were sequenced through de novo interpretation using PEAKS Studio algorithms.
500 resolution using a 50 ms maximum injection time and a 1e5 AGC target. Precursors were isolated with a 1.6 m/z window, and dynamic exclusion was set to 30 s. All detected peptides were sequenced through de novo interpretation using PEAKS Studio algorithms.
2.6 In vitro DPP-IV inhibition assay
The DPP-IV inhibitory activity of peptides was quantified using a commercial DPP-IV inhibitor screening kit according to the established protocols.19 Reaction mixtures containing 30 μL of assay buffer, 10 μL of DPP-IV enzyme, and 10 μL of peptide sample were incubated with 50 μL of substrate solution at 37 °C for 30 min in 96-well plates. Fluorescence intensity was measured at 355 ± 5 nm excitation and 460 ± 5 nm emission. Control readings included blanks (buffer instead of enzyme) and negative controls (buffer instead of sample). Percent inhibition was calculated asAnti-DPP-IV effects were determined for the peptides LPQ, LPQF, LP, FP, VP, and LPT at 500 μM and 1000 μM concentrations (in triplicate).
2.7 Antihyperglycemic efficacy in a hyperglycemic zebrafish model
The glucose-lowering effects of peptides were evaluated using a streptozotocin (STZ)/glucose-induced hyperglycemic zebrafish model. The hyperglycemic zebrafish model was established based on the method described by Kim et al., with some modifications.20 Wild-type AB strain zebrafish (4 days post-fertilization) were distributed into 6-well plates (10 fish per well, three wells per group) and treated as follows: (1) NC: system water, (2) MC: 333 μM STZ + 2.67% (w/v) glucose in system water, (3) Met: 333 μM STZ + 2.67% glucose + 5 μg mL−1 metformin, and (4) peptide groups: 333 μM STZ + 2.67% glucose + 1 μg mL−1 peptide (LPQ, LPQF, LP, FP, VP, or LPT). Following 24 h of exposure, zebrafish were rinsed three times with PBS to remove surface glucose. The fish were transferred to 1.5 mL microtubes using disposable pipettes, and excess fluid was aspirated. The samples were dehydrated with 100 μL of absolute ethanol (15 min, dark), dried at 60 °C for 120 min, and then homogenized in 5 μL of ultrapure water. After vortex mixing (10 min), glucose levels in 2 μL supernatants were quantified using a glucometer.2.8 Data processing and statistical analysis
Statistical comparisons of body weight, food intake, tissue weights, serum biochemical parameters, and inflammatory cytokine levels were performed using GraphPad Prism version 9.0. Data are presented as mean ± SD or mean ± SEM, as specified in the corresponding figure legends. Statistical significance was evaluated using one-way analysis of variance (ANOVA) with Duncan's test. Differences were considered statistically significant at p < 0.05. Graphical representations were generated using the same software platform.3 Results
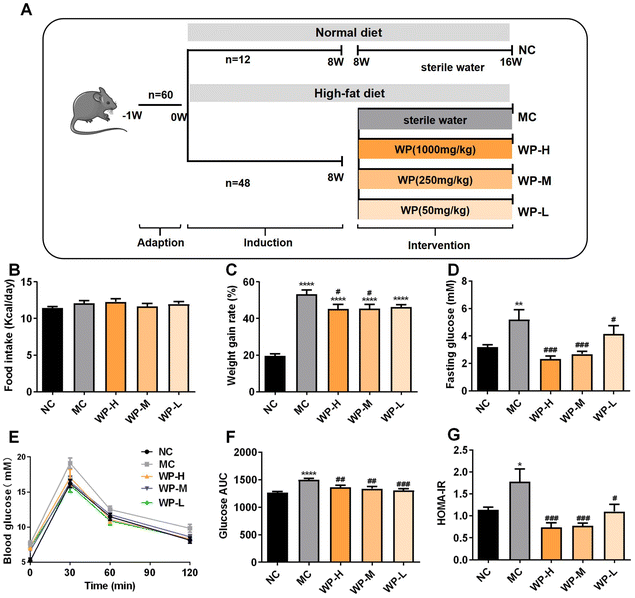
3.1 WP attenuate HFD-induced weight gain and hyperglycemia
To investigate the antihyperglycemic effects of WP, mice were subjected to 8 weeks of high-fat diet (HFD) induction followed by low- (50 mg kg−1), medium- (250 mg kg−1), or high-dose (1000 mg kg−1) WP intervention (experimental design: Fig. 1A). Analysis of energy intake and body weight gain revealed no significant differences in cumulative caloric consumption among the groups (Fig. 1B). However, HFD-fed model control (MC) mice exhibited significantly higher weight gain versus normal chow controls (NCs) (Fig. 1C, p < 0.0001). Medium- and high-dose WP intervention significantly attenuated this weight gain (p < 0.05) without altering energy intake.Fasting blood glucose levels were markedly elevated in HFD-fed mice (Fig. 1D, p < 0.01). All WP doses significantly reduced fasting hyperglycemia compared to MC (p < 0.05). Oral glucose tolerance tests (OGTTs) demonstrated impaired glucose clearance in MC mice, with significantly increased AUC compared to NC (Fig. 1E and F, p < 0.0001). All WP doses substantially improved glucose tolerance, significantly lowering AUC values (p < 0.01). Consistent with these findings, HOMA-IR analysis confirmed pronounced insulin resistance in MC mice compared to NC (Fig. 1G, p < 0.05). WP intervention at all doses significantly reduced HFD-induced insulin resistance (p < 0.05), demonstrating WP's efficacy in counteracting diet-induced metabolic dysfunction.
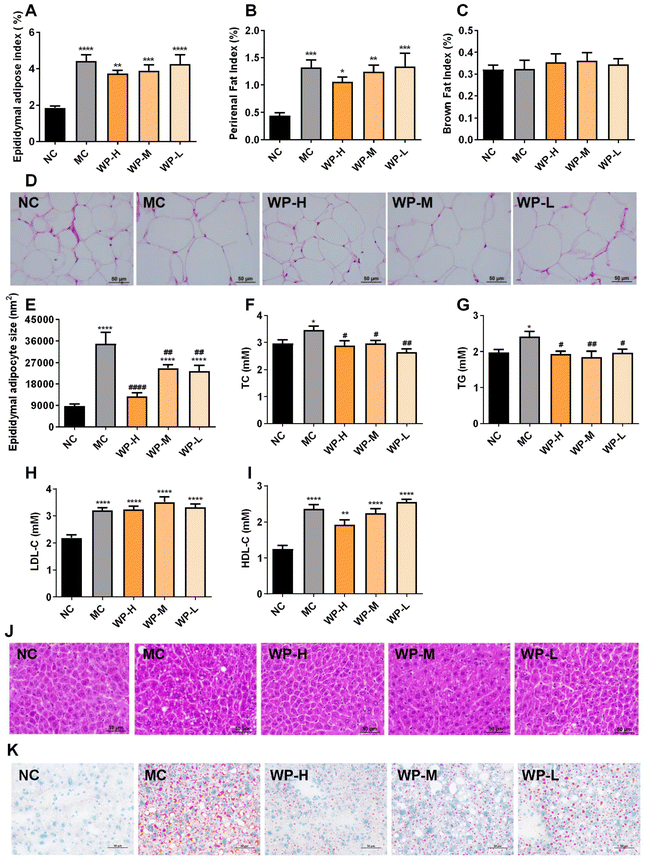
3.2 WP modulate adipose composition and lipid metabolism in HFD-fed mice
Adipose tissue indices revealed no significant differences in epididymal or perirenal fat mass between high-dose WP (1000 mg kg−1) and MC groups (Fig. 2A and B, p > 0.05). Interscapular brown adipose mass remained comparable across all groups (Fig. 2C). However, WP intervention significantly reduced epididymal adipocyte size relative to MC (Fig. 2D and E, p < 0.01), which exhibited marked hypertrophy compared to NC controls.Serum lipid profiling demonstrated that all WP doses significantly lowered TC and TG levels in HFD-fed mice (Fig. 2F and G, p < 0.05), while LDL-C and HDL-C levels remained unaffected (Fig. 2H and I, p > 0.05). Histological analysis of liver sections showed pronounced lipid droplet accumulation in MC mice, with WP treatment attenuating hepatic steatosis severity (Fig. 2J and K). These findings indicate favorable modulation of lipid metabolism by WP in diet-induced metabolic dysfunction.
3.3 WP attenuate HFD-induced low-grade inflammation
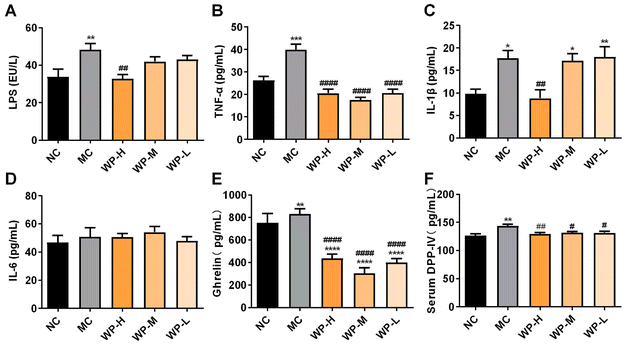
Obesity and insulin resistance are commonly associated with low-grade systemic inflammation.21 Low-grade inflammation is a chronic and systemic state of mild inflammation characterized by an increase in circulating pro-inflammatory cytokines (such as TNF-α and LPS), without the classic signs of acute inflammation.21 LPS, a component of Gram-negative bacterial cell walls, triggers inflammation via TLR4 activation upon binding to LPS-binding protein.22 Serum LPS levels were significantly elevated in MC mice compared to NC (Fig. 3A, p < 0.01). WP intervention restored LPS to the baseline (p > 0.05 vs. NC), with high-dose WP yielding a significant reduction compared to MC (p < 0.01). Proinflammatory cytokines TNF-α and IL-1β were markedly increased in MC mice (Fig. 3B and C, p < 0.05). All WP doses significantly suppressed TNF-α levels (p < 0.0001 vs. MC), while high-dose WP additionally reduced IL-1β (p < 0.01). No significant differences in IL-6 were observed across groups (Fig. 3D, p > 0.05). These findings demonstrate WP's efficacy in mitigating diet-induced systemic inflammation.3.4 WP regulate hormonal and DPP-IV activity in HFD-fed mice
Serum ghrelin, an orexigenic gut hormone that promotes hyperglycemia through growth hormone secretagogue receptor (GHSR) signaling, was significantly elevated in MC mice compared to NC (Fig. 3E, p < 0.05). All WP doses potently suppressed this HFD-induced ghrelin upregulation (p < 0.0001), suggesting appetite-modulatory effects. Concurrently, DPP-IV activity, critically involved in incretin degradation, showed marked elevation in MC mice (p < 0.01 vs. NC). WP intervention at all doses significantly reduced serum DPP-IV levels compared to MC (Fig. 3F, p < 0.05).3.5 The effect of WP on the intestinal microbiota in HFD-fed mice
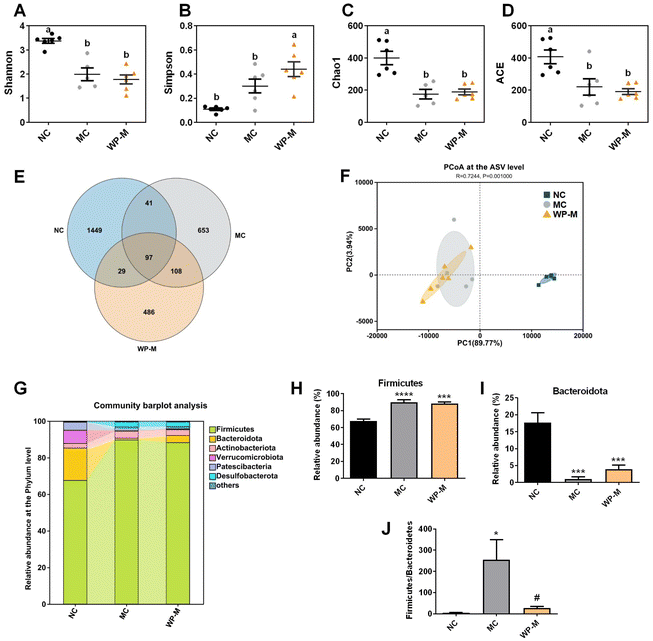
Chronic HFD exposure reduced gut microbial diversity. α-Diversity analysis (Shannon, Simpson, Chao1, and ACE indices) revealed a significantly lower Shannon index in MC compared to NC (Fig. 4A, p < 0.05), indicating reduced community evenness. Medium-dose WP (WP-M) did not restore Shannon values (p > 0.05). The Simpson index increased significantly in WP-M compared to both NC and MC (Fig. 4B, p < 0.05). Reduced Chao1 and ACE indices in MC mice (Fig. 4C and D, p < 0.05) confirmed diminished species richness, unaffected by WP-M intervention. Venn analysis of amplicon sequence variants (ASVs) showed decreased OTU counts: NC (1616) > MC (899) > WP-M (720) (Fig. 4E). β-Diversity demonstrated complete separation between NC and MC groups, while WP-M clustered closely with MC (Fig. 4F), suggesting limited structural reorganization of the gut microbiota.Phylum-level analysis identified six dominant taxa, including Firmicutes, Bacteroidota, Actinobacteriota, Verrucomicrobiota, Patescibacteria and Desulfobacterota (Fig. 4G). HFD significantly increased Firmicutes (Fig. 4H, p < 0.0001) and decreased Bacteroidota abundance (Fig. 4I, p < 0.001) compared to NC. Although WP-M did not significantly alter individual phylum abundances, it significantly reduced the Firmicutes/Bacteroidota (F/B) ratio (Fig. 4J, p < 0.05), which is a key biomarker positively correlated with adiposity.23 These results indicate that although WP does not significantly alter the microbiota structurally, it promotes the positive development of Firmicutes/Bacteroidota.
LEfSe analysis identified discriminant taxa across groups (Fig. 5A and B). NC mice exhibited significantly higher abundances of Bifidobacterium, Lactobacillus, and Akkermansia compared to MC (Fig. 5C–F). WP-M intervention significantly increased Bifidobacterium and Lactobacillus (p < 0.05), with a non-significant upward trend in Akkermansia. Additionally, WP-M enhanced Dubosiella abundance. Correlation analyses revealed significant negative correlations of Akkermansia with weight gain, HDL-C, and IL-1β, and of Lactobacillus with weight gain, AUC, HDL-C, and LPS.
3.6 WP alter serum metabolites in HFD-fed mice
Serum metabolites reflect not only endogenous compounds derived from cellular metabolism but also those originating from dietary components, thereby providing a functional readout of food–host metabolic interactions. PLS-DA analysis of serum metabolites (both positive and negative ion modes) revealed distinct clustering patterns among the NC, MC, and WP-M groups (Fig. 6A). Partial separation between the WP-M and MC cohorts indicated partial metabolic recovery following medium-dose WP intervention. Volcano plot analysis identified 130 upregulated and 231 downregulated metabolites in MC compared to NC (FC ≥ 1, p < 0.05), while WP-M treatment reversed this profile with 94 upregulated and 34 downregulated metabolites compared to MC (Fig. 6B and C).KEGG pathway enrichment analysis demonstrated significant alterations in lipid metabolism and amino acid metabolism (Fig. 6D). WP-M specifically modulated five key pathways: ovarian steroidogenesis, neuroactive ligand–receptor interaction, glycerophospholipid metabolism, arachidonic acid metabolism, and tryptophan metabolism (Fig. 6E).
Correlation analysis integrated significantly altered metabolites with metabolic parameters (Fig. 6F and G). Key metabolites including PE(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1/20
1/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), PE(14
3), PE(14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0/18
0/18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), PG(18
0), PG(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0/20
0/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), PS(22
4), PS(22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6/22
6/22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and HIAA showed significant inverse correlations (p < 0.05) with weight gain and glucose AUC. These molecules functionally connect the observed metabolic shifts to WP's antihyperglycemic effects.
1) and HIAA showed significant inverse correlations (p < 0.05) with weight gain and glucose AUC. These molecules functionally connect the observed metabolic shifts to WP's antihyperglycemic effects.
3.7 LPQ/LPQF-containing peptides mediate WP's antihyperglycemic effects
LC-MS/MS analysis identified 525 and 231 peptide sequences in pre- and post-digestion wheat peptide (WP) samples, respectively, with average local confidence scores exceeding 80%. Notably, multiple LPQ/LPQF-containing peptides (LPQFEELLNR, LPQFEELR, LPQFAELR, LPQFEALR, LPQFEELNR, LPQFEF, and LPQQL) were detected in undigested WP. Digestion-resistant fragments including LPQF, LPQFE, and LPQQL persisted post-hydrolysis (Table 1; MS/MS spectra: SI Fig. S1–S9). The structural presence of second proline residues in LPQ motifs aligns with established DPP-IV inhibitory mechanisms,24 suggesting potential target engagement.| WP | Peptide | Scan | Score | Length | m/z | z | RT | Area | Mass | ppm |
|---|---|---|---|---|---|---|---|---|---|---|
| Before digestion | LPQFEELLNR | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 502 502 |
97 | 10 | 629.848 | 2 | 41.84 | 1.74 × 108 | 1257.6716 | 7.8 |
| LPQFEELR | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 963 963 |
99 | 8 | 516.2833 | 2 | 32.43 | 4.65 × 107 | 1030.5447 | 7.2 | |
| LPQFAELR | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 769 769 |
99 | 8 | 487.2795 | 2 | 33.75 | 1.74 × 107 | 972.5392 | 5.4 | |
| LPQFEALR | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 450 450 |
91 | 8 | 487.2795 | 2 | 33.75 | 1.74 × 107 | 972.5392 | 5.4 | |
| LPQFEELNR | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 475 475 |
89 | 9 | 573.3058 | 2 | 30.3 | 8.83 × 106 | 1144.5876 | 8.2 | |
| LPQFEF | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 508 508 |
84 | 6 | 780.3959 | 1 | 47.86 | 2.86 × 106 | 779.3854 | 4.1 | |
| LPQQL | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 242 242 |
94 | 5 | 598.3623 | 1 | 23.03 | 5.99 × 106 | 597.3486 | 10.8 | |
| After digestion | LPQF | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 812 812 |
97 | 4 | 504.2824 | 1 | 38.74 | 1.65 × 106 | 503.2744 | 1.5 |
| LPQFE | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 929 929 |
97 | 5 | 633.3247 | 1 | 35.44 | 1.18 × 106 | 632.3170 | 0.8 | |
| LPQQL | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 218 218 |
94 | 5 | 598.3560 | 1 | 26.5 | 2.04 × 106 | 597.3486 | 0.2 |
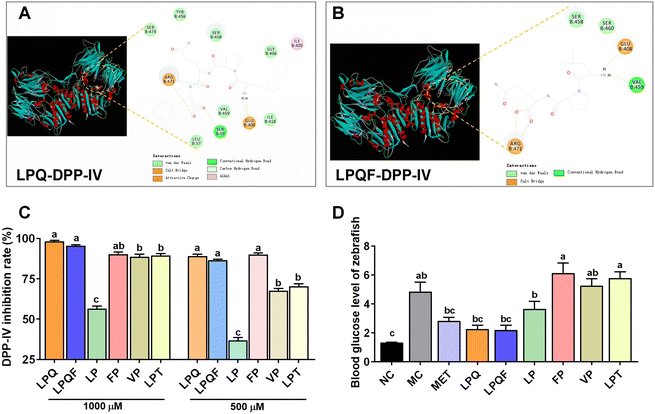
Molecular docking simulations against DPP-IV (PDB:1WCY) revealed high-affinity binding for both LPQ (docking energy: −32.50 kcal mol−1) and LPQF (−52.58 kcal mol−1). LPQ formed critical interactions through van der Waals forces (Ser458, Ser460), hydrogen bonds (Arg471, Val459), and electrostatic interactions (Glu408, Arg471). LPQF exhibited enhanced binding via additional hydrophobic interactions (Ile405, Tyr456) and hydrogen bonding (Ser59) (Fig. 7A and B).
In vitro validation demonstrated potent DPP-IV inhibition (>85% at 500–1000 μM) for both peptides (Fig. 7C). In hyperglycemic zebrafish (333 μM streptozotocin + 2.67% glucose), LPQ and LPQF (0.1–5 μg mL−1) significantly normalized blood glucose (p < 0.001 vs. MC; p > 0.05 vs. NC), outperforming 5 μg mL−1 metformin (p < 0.01 vs. MC) at equivalent concentrations (Fig. 7D).
Furthermore, structure–activity relationship analysis revealed that the dipeptide LP showed negligible activity (p > 0.05 vs. MC). Alanine-substituted LPT exhibited reduced efficacy (p < 0.001 vs. LPQ/LPQF). Control dipeptides (FP/VP) demonstrated minimal bioactivity. These findings establish the tripeptide LPQ and tetrapeptide LPQF as principal mediators of WP's antihyperglycemic effects through potent DPP-IV inhibition.
4 Discussion
This study employed a high-fat diet (HFD) model to establish obesity-associated hyperglycemia in mice, consistent with established metabolic research.25 The linear relationship between adiposity and type 2 diabetes pathogenesis manifests through adiposity-driven β-cell dysfunction and multi-organ insulin resistance.2 Our HFD cohort exhibited significantly elevated weight gain, fasting glucose, glucose AUC, and HOMA-IR, confirming successful metabolic dysregulation. Critically, oral administration of WP (50–1000 mg kg−1) counteracted these abnormalities by attenuating weight gain progression, fasting hyperglycemia, glucose intolerance, and insulin resistance, collectively demonstrating its therapeutic potential for metabolic homeostasis restoration in diet-induced diabetes. It is noteworthy that bioactive peptides can sometimes exhibit non-monotonic or plateaued dose–response effects, where benefits do not linearly increase with dosage and may even reach a maximum effect at a certain concentration.26,27 Therefore, we intentionally selected a broad dose range to identify the effective therapeutic window for WP. The results demonstrate that WP administration within this range (50–1000 mg kg−1) effectively counteracted key metabolic abnormalities. We also found the absence of a strict linear dose dependence across all parameters. It may be attributed to a confluence of factors, including receptor saturation, ceiling effects in certain responses, and the distinct pathophysiological thresholds of different metabolic organs. This non-uniform response pattern underscores the multi-faceted and complex nature of WP's action within the diet-induced metabolic dysregulation.HFD-induced dyslipidemia was mitigated by WP intervention, as evidenced by histological analyses. Epididymal adipose H&E staining revealed that WP significantly suppressed adipocyte hypertrophy, thereby attenuating ectopic lipid accumulation. Hepatic sections demonstrated pronounced macrovesicular steatosis in HFD controls, whereas WP-treated mice exhibited markedly reduced lipid droplet deposition, indicating improved hepatic lipid metabolism. Biochemically, HFD elevated serum TC, TG, LDL-C, and HDL-C levels. WP administration specifically normalized TC and TG levels, confirming its regulatory effects on systemic lipid homeostasis. Mechanistically, these lipid-modulating actions may contribute to improved glucose metabolism through reduced lipotoxicity and enhanced insulin sensitivity.28 However, interestingly, in terms of HDL-C, while the WP-treated groups showed no statistically significant difference compared to the MC group, a downward trend with increasing dosage was observed. This phenomenon is not without precedent. For instance, glycerol monolaurate (300 mg kg−1), despite demonstrating significant hypoglycemic effects in HFD-fed mice, was also reported to reduce HDL-C levels.29 Further literature review reveals that potent metabolic interventions, including certain lipid-lowering agents and dietary supplements, can sometimes paradoxically lower HDL-C concentrations.30,31 This is often interpreted in the context of “HDL remodeling”, a process where the composition and function of HDL particles are enhanced—such as through improved cholesterol efflux capacity—even as their plasma concentrations decrease. We hypothesize that high-dose WP may promote such functional remodeling, potentially leading to more efficient reverse cholesterol transport.
Dysregulation of lipid metabolism promotes systemic inflammation, a significant contributor to the development of insulin resistance.32,33 Research indicates that HFD consumption induces metabolic endotoxemia, marked by elevated circulating lipopolysaccharide (LPS) levels in serum.34 This condition results from enhanced LPS transport from the gut lumen, mediated by enterocyte-derived chylomicrons synthesized in response to dietary fat intake.34 Consistent with this mechanism, our study observed a significant increase in serum LPS concentrations in mice following 16 weeks of HFD feeding. Intervention with WP effectively reduced LPS levels, demonstrating its beneficial impact on mitigating metabolic endotoxemia. Furthermore, elevated circulating LPS is known to induce low-grade inflammation, subsequently leading to insulin resistance.35 Consequently, we quantified serum levels of the pro-inflammatory cytokine TNF-α. As anticipated, the abnormal rise in TNF-α induced by HFD was normalized upon WP administration. IL-1β, another key pro-inflammatory cytokine, was also significantly downregulated by WP treatment. This reduction in IL-1β aligns with findings reported by W. Makena et al., where interventions (genistein and Momordica charantia L.) also demonstrated suppressive effects on IL-1β levels alongside improvements in diabetic parameters.36 Collectively, these results indicate that WP intervention effectively ameliorates HFD-induced metabolic endotoxemia and suppresses the ensuing inflammatory response.
The gut microbiota is intricately linked to the pathogenesis of metabolic syndrome.37 Numerous bioactive compounds known to ameliorate glucose and lipid metabolism disorders exert their effects through modulation of the gut microbiome, as demonstrated by studies on substances such as L-arabinose combined with lactulose and Lactobacillus plantarum,38 and phenolic-enriched Rosa rugosa extract.39 In line with this, various protein-derived peptides have been reported to influence gut microbiota composition and function. For instance, bovine α-lactalbumin hydrolysates were found to alleviate endotoxemia and systemic inflammation in high-fat diet-induced obese mice by altering the overall gut microbial structure and reducing the Firmicutes/Bacteroidetes ratio.40 Similarly, Han et al. showed that pepsin-hydrolyzed peptides from soybean 7S globulin lowered body weight and influenced gut microbiota composition in obese individuals.41 Wang et al. showed that collagen peptides from walleye pollock skin mitigate obesity and modulate the gut microbiota in high-fat diet models, enhancing the abundance of beneficial bacteria such as Lactobacillus and Akkermansia, while suppressing pro-inflammatory taxa.42 Interestingly, in our investigation, while medium-dose WP intervention elicited a significant anti-diabetic effect, it did not induce statistically significant alterations in the gut microbiota structure (assessed via alpha and beta diversity indices). This observation suggests that modulation of the overall gut microbial community structure may not be the primary mechanism underlying WP's anti-diabetic efficacy. Nevertheless, despite the lack of significant structural changes, analysis at the phylum level revealed that medium-dose WP significantly attenuated the HFD-induced elevation in the Firmicutes-to-Bacteroidetes (F/B) ratio. An elevated F/B ratio is a recognized hallmark associated with obesity and metabolic dysfunction.23 Consequently, WP-mediated reduction of the F/B ratio likely contributes to the alleviation of obesity, thereby indirectly improving hyperglycemia. Furthermore, WP administration enhanced the relative abundance of beneficial bacterial genera, including Bifidobacterium, Lactobacillus, and Akkermansia. Increased levels of Bifidobacterium and Lactobacillus genera are frequently correlated with improved diabetic parameters, consistent with findings where metformin treatment increased the abundance of these beneficial bacteria alongside diabetes amelioration.43 Notably, Akkermansia is increasingly recognized as a next-generation probiotic bacterium inversely associated with markers of intestinal inflammation.44,45 This increase in Akkermansia abundance following WP supplementation mirrors results observed in our prior research on fermented sea buckthorn.13 Beyond complex protein hydrolysates, certain single peptide sequences have also demonstrated the capacity to exert health-promoting effects via modulation of the gut microbiota and related metabolites. For example, rice protein-derived peptides such as IQP, VEP, and IPQ have been shown to alleviate obesity-related metabolic disorders in high-fat diet-fed mice through activation of AMPK and regulation of gut microbial metabolites.12 These findings highlight that specific peptide sequences, in addition to composite hydrolysates, can participate in food matrix–microbiota interactions to influence host metabolism. Collectively, although the gut microbiota may not constitute the principal target of WP's anti-diabetic action, our data indicate that WP intervention confers beneficial modulatory effects on specific HFD-induced dysbiosis within the gut microbial ecosystem.
Metabolomics provides a comprehensive profile of small-molecule metabolites within biological systems. Our analysis revealed that the WP-M group significantly elevated levels of specific phospholipids, namely PE(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1/20
1/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), PE(14
3), PE(14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0/18
0/18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), PG(18
0), PG(18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0/20
0/20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4), and PS(22
4), and PS(22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6/22
6/22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The enzyme phosphatidylethanolamine N-methyltransferase (PEMT) has emerged as a key modulator in type 2 diabetes pathogenesis. This is supported by observed associations between the PEMT genetic variant rs4646404 and insulin resistance in obese individuals.46 Moreover, adipose tissue PEMT expression correlates positively with insulin resistance in this population.46 Hepatic PEMT activity is also upregulated in diabetic states, as demonstrated in streptozotocin-treated rat models.47 Streptozotocin-induced diabetic rat studies further implicate PEMT in diabetic nephropathy progression.48 Notably, our data demonstrate significant inverse correlations between circulating PE levels and key metabolic parameters including body weight, AUC (glucose tolerance), and fasting blood glucose (FBG). Phosphatidylglycerol (PG), recognized for its superior emulsifying properties as a functional lipid, exhibited similar negative correlations with weight, AUC, and FBG. This aligns with research showing the metabolic benefits of n-3 PUFA-bound PG in diabetic/obese models.49 Phosphatidylserine (PS), another essential membrane phospholipid with documented bioactivity,50 also showed significant inverse relationships with body weight and AUC in our cohort. Supplementation studies further suggest that PS may improve vascular function in type 2 diabetes.50 These consistent patterns support the hypothesis that WP ameliorates hyperglycemia, at least partially, through modulation of these functional phospholipids. Beyond phospholipids, WP intervention influenced the tryptophan metabolic pathway, specifically reversing the HFD-induced reduction in 5-hydroxyindoleacetate (5-HIAA). This metabolite is known to alleviate HFD-induced insulin resistance via activation of the hepatic AhR/TSC2/mTORC1 signaling axis and by mitigating adipose tissue inflammation.51 Mirroring the phospholipid findings, 5-HIAA levels in our study were inversely correlated with weight, AUC, and FBG. Taken collectively, the metabolomic profile suggests that WP-M's anti-diabetic effects involve coordinated modulation of functional lipid metabolism and the tryptophan pathway.
1). The enzyme phosphatidylethanolamine N-methyltransferase (PEMT) has emerged as a key modulator in type 2 diabetes pathogenesis. This is supported by observed associations between the PEMT genetic variant rs4646404 and insulin resistance in obese individuals.46 Moreover, adipose tissue PEMT expression correlates positively with insulin resistance in this population.46 Hepatic PEMT activity is also upregulated in diabetic states, as demonstrated in streptozotocin-treated rat models.47 Streptozotocin-induced diabetic rat studies further implicate PEMT in diabetic nephropathy progression.48 Notably, our data demonstrate significant inverse correlations between circulating PE levels and key metabolic parameters including body weight, AUC (glucose tolerance), and fasting blood glucose (FBG). Phosphatidylglycerol (PG), recognized for its superior emulsifying properties as a functional lipid, exhibited similar negative correlations with weight, AUC, and FBG. This aligns with research showing the metabolic benefits of n-3 PUFA-bound PG in diabetic/obese models.49 Phosphatidylserine (PS), another essential membrane phospholipid with documented bioactivity,50 also showed significant inverse relationships with body weight and AUC in our cohort. Supplementation studies further suggest that PS may improve vascular function in type 2 diabetes.50 These consistent patterns support the hypothesis that WP ameliorates hyperglycemia, at least partially, through modulation of these functional phospholipids. Beyond phospholipids, WP intervention influenced the tryptophan metabolic pathway, specifically reversing the HFD-induced reduction in 5-hydroxyindoleacetate (5-HIAA). This metabolite is known to alleviate HFD-induced insulin resistance via activation of the hepatic AhR/TSC2/mTORC1 signaling axis and by mitigating adipose tissue inflammation.51 Mirroring the phospholipid findings, 5-HIAA levels in our study were inversely correlated with weight, AUC, and FBG. Taken collectively, the metabolomic profile suggests that WP-M's anti-diabetic effects involve coordinated modulation of functional lipid metabolism and the tryptophan pathway.
DPP-IV represents a promising therapeutic target for diabetes management. Studies demonstrate that DPP-IV deficient mice (DPP-IV−/−) exhibit resistance to diet-induced obesity and improved postprandial glucose control.52,53 These mice also exhibit enhanced insulin sensitivity, reduced islet hypertrophy, and protection against streptozotocin-induced β-cell loss and hyperglycemia.53 Pharmacological inhibition of DPP-IV activity ameliorates glucose intolerance in wild-type mice, while showing no additional benefit in DPP-IV−/− animals,52 confirming the enzyme's central role in glucose regulation. Previous studies have shown that peptide segments composed of a second or C-terminal proline possess potential DPP-IV inhibitory activity.54 Interestingly, our analysis of WP identified multiple sequences containing LPQ and LPQF motifs – structures characterized by a proline residue at the second position. Our findings indicate that LPQF is likely a stable digestive fragment derived from WP following gastrointestinal digestion. Our in vitro simulated gastrointestinal digestion experiment provided direct insight into this issue. While the seven longer parent peptides containing the LPQ/LPQF motifs (LPQFEELLNR, LPQFEELR, LPQFAELR, LPQFEALR, LPQFEELNR, LPQFEF, and LPQQL) identified prior to digestion were degraded, we consistently detected the shorter peptides LPQF, LPQFE, and LPQQL in the final digest. A key observation is that we did not detect any further degradation products of these core sequences, such as PQF, LP, or PQ. This absence suggests that the LPQF motif, particularly the peptide bonds within it, exhibits notable resistance to cleavage by gastrointestinal proteases. Therefore, we propose that the LPQF core sequence represents a stable, digestion-resistant fragment liberated from larger parent peptides. Molecular docking revealed high binding affinity between these peptides and the DPP-IV active site, correlating with potent in vitro DPP-IV inhibition. Subsequent zebrafish models confirmed their significant glucose-lowering efficacy. Comparative studies demonstrated that LPQ/LPQF peptides exhibit superior DPP-IV inhibitory activity and in vivo hypoglycemic effects compared with structurally similar peptides (LP, FP, VP, and LPT) with analogous proline positioning. This finding aligns with previous research on wheat gluten hydrolysates—proline-rich proteins that generate DPP-IV inhibitory peptides, particularly those featuring proline at the second position or C-terminal position.24 DPP-IV inhibition mechanisms include competitive, non-competitive, and mixed modes. Competitive inhibitors mimic the substrate's Xaa-Pro/Glu sequence, reversibly binding the catalytic triad (Ser630, Asp708, and His740) to block substrate access. Non-competitive inhibitors induce conformational changes via allosteric binding (e.g., to the β-propeller domain), reducing catalytic efficiency and often exhibiting slow dissociation kinetics.55 Mixed-type inhibitors combine both mechanisms, as exemplified by peptides LPIIDI and APGPAGP, which interact with both the active site and regulatory domains.56 Kinetic analyses (Lineweaver–Burk plots) further classify inhibitory peptides: QPY, WPEYL and YPPQVM function competitively, while LPAAP and IPAPSFPRL exhibit mixed-type inhibition.57 Our structural modeling indicates that LPQ primarily engages DPP-IV through van der Waals forces, hydrogen bonding, and electrostatic interactions (salt bridges). LPQF interacts with DPP-IV through multiple non-covalent interactions, including van der Waals forces, hydrogen bonding, hydrophobic interactions (primarily alkyl packing), and electrostatic forces encompassing both salt bridges and charge–charge attraction.
Collectively, our findings demonstrate WP's efficacy in counteracting HFD-induced hyperglycemia and metabolic disturbances through a potentially synergistic, multi-target mechanism: suppressing inflammation, restoring lipid metabolism (PE/PG/PS) and tryptophan profiles, modulating the gut microbiota (reduced F/B and enriched Bifidobacterium/Lactobacillus), and potently inhibiting DPP-IV via LPQ/LPQF peptides. This positions WP as a promising multi-target strategy for diabetes intervention. It is important to note that this study primarily explores potential mechanisms; the precise and detailed molecular crosstalk underlying these effects warrants further in-depth investigation. Future studies could address clinical translation, precise mechanistic crosstalk, component specificity, long-term effects, and individual variability to realize its full therapeutic potential.
Author contributions
Fei Shen: conceptualization, methodology, investigation, formal analysis, writing – original draft, and financial support. Qian Deng: investigation and writing – original draft. Yishan Song: investigation. Guanghua He: conceptualization and financial support. Xiaojun Chu: conceptualization. Mingjie Zhao: conceptualization. Juan Du: investigation. Fengqin Feng: conceptualization. Xi Zhang: conceptualization, financial support, supervision, and financial support. Hao Zhong: conceptualization, investigation, supervision, project administration, and financial support.Conflicts of interest
The authors declare that they have no conflicts of interest to disclose.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information mainly include the main component content of WP, the relative molecular weight distribution of WP, the amino acid composition of WP, and the secondary mass spectrum of some peptides. See DOI: https://doi.org/10.1039/d5fo03412d.Acknowledgements
We thank Yingniang Li from the Core Facilities, Zhejiang University School of Medicine, for their technical support. This research was funded by Zhejiang Provincial Leading Geese plan (2025C04040), Zhejiang University of Science and Technology Research Initiation Project (2024F701103P08), the Yunnan Young & Elite Talents Project (No. YNWR-QNBJ-2020-213), the National Natural Science Foundation of China (No. 32402088) and the Science and Technology Development Plan Project of Hangzhou City (20241203A02).References
- P. Saeedi, I. Petersohn and P. Salpea, et al., Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the international diabetes federation diabetes atlas, 9(th) edition, Diabetes Res. Clin. Pract., 2019, 157, 107843 CrossRef PubMed.
- S. Klein, A. Gastaldelli, H. Yki-Jarvinen and P. E. Scherer, Why does obesity cause diabetes?, Cell Metab., 2022, 34, 11–20 CrossRef CAS PubMed.
- J. M. Forbes and M. E. Cooper, Mechanisms of diabetic complications, Physiol. Rev., 2013, 93, 137–188 CrossRef CAS PubMed.
- J. Cai, C. Li, Z. Liu, J. Du, J. Ye, Q. Gu and J. Xu, Predicting DPP-IV inhibitors with machine learning approaches, J. Comput.-Aided Mol. Des., 2017, 31, 393–402 CrossRef CAS PubMed.
- P. M. Jedlowski, M. F. Jedlowski and M. T. Fazel, DPP-4 inhibitors and increased reporting odds of bullous pemphigoid: a pharmacovigilance study of the FDA adverse event reporting system (FAERS) from 2006 to 2020, Am. J. Clin. Dermatol., 2021, 22, 891–900 CrossRef PubMed.
- K. Sato, Metabolic fate and bioavailability of food-derived peptides: are normal peptides passed through the intestinal layer to exert biological effects via proposed mechanisms?, J. Agric. Food Chem., 2022, 70, 1461–1466 CrossRef CAS PubMed.
- S. B. Mada, C. P. Ugwu and M. M. Abarshi, Health promoting effects of food-derived bioactive peptides: a review, Int. J. Pept. Res. Ther., 2020, 26, 831–848 CrossRef CAS.
- W. Wang, W. Yang, Y. Dai, J. Liu and Z.-Y. Chen, Production of food-derived bioactive peptides with potential application in the management of diabetes and obesity: a review, J. Agric. Food Chem., 2023, 71, 5917–5943 CrossRef CAS PubMed.
- Z. Weng, Y. Chen, T. Liang, Y. Lin, H. Cao, H. Song, L. Xiong, F. Wang, X. Shen and J. Xiao, A review on processing methods and functions of wheat germ-derived bioactive peptides, Crit. Rev. Food Sci. Nutr., 2023, 63, 5577–5593 CrossRef CAS PubMed.
- F. Rivero-Pino, F. J. Espejo-Carpio and E. M. Guadix, Identification of dipeptidyl peptidase IV (DPP-IV) inhibitory peptides from vegetable protein sources, Food Chem., 2021, 354, 129473 CrossRef CAS PubMed.
- T. Hatanaka, Y. Inoue, J. Arima, Y. Kumagai, H. Usuki, K. Kawakami, M. Kimura and T. Mukaihara, Production of dipeptidyl peptidase IV inhibitory peptides from defatted rice bran, Food Chem., 2012, 134, 797–802 CrossRef CAS PubMed.
- L. Liu, J. Jian, S. Yu, S. Li, J. Wang, T. Bu and J. Wu, Rice protein-derived hypoglycemic peptides IQP,VEP and IPQ alleviate obesity-related metabolic disorders in high-fat diet (HFD)-induced C57BL/6J mice through activation of AMPK and modulation of gut microbiota and related metabolites, Food Biosci., 2025, 72, 107427 CrossRef CAS.
- F. Shen, J. Zhuang, Q. Wang, J. Zhang, Y. Huang, Q. Mo, M. Zhao, J. Wang, H. Zhong and F. Feng, Enhancement in the metabolic profile of sea buckthorn juice via fermentation for its better efficacy on attenuating diet-induced metabolic syndrome by targeting gut microbiota, Food Res. Int., 2022, 162, 111948 CrossRef CAS PubMed.
- F. Shen, Q. Wang, H. Zhong, Y. Pan, J. Zhang, J. Wang, M. Chen, F. Feng and M. Zhao, Combination of galacto-oligosaccharides and wheat peptides synergistically relieved constipation via regulating water transport, gut barrier, inflammation and gut microbiota, Food Biosci., 2023, 56, 103208 CrossRef CAS.
- J. Zhang, H. Yu, Q. Wang, H. Cai, F. Shen, S. Ruan, Y. Wu, T. Liu, F. Feng and M. Zhao, Dietary additive octyl and decyl glycerate modulates metabolism and inflammation under different dietary patterns with the contribution of the gut microbiota, Food Funct., 2023, 14, 525–540 RSC.
- F. Shen, Q. Wang, S. Ullah, Y. Pan, M. Zhao, J. Wang, M. Chen, F. Feng and H. Zhong, Ligilactobacillus acidipiscis YJ5 modulates the gut microbiota and produces beneficial metabolites to relieve constipation by enhancing the mucosal barrier, Food Funct., 2024, 15, 310–325 RSC.
- F. Shen, H. Zhu, Z. Bao, D. Han, J. Du, M. Zhao, F. Feng, G. He and Q. Mo, Lactiplantibacillus plantarum T34 alleviates constipation by enhancing intestinal barrier and modulating gut homeostasis, Food Biosci., 2025, 66, 106195 CrossRef CAS.
- Q. Wang, J. Zhang, F. Shen, J. Du and F. Feng, Sea cucumber (Acaudina leucoprocta) peptides exhibit anti-aging effect in Drosophila melanogaster via regulation of microbiota dysbiosis and metabolic disorder, Food Biosci., 2024, 60, 104476 CrossRef CAS.
- J. Lan, K. Dang, Y. Wang, L. Du, D. Pan, Y. Dang and X. Gao, Evaluation of novel angiotensin I-converting enzyme and dipeptidyl peptidase IV inhibitory peptides derived from yak milk based on peptidomics and network pharmacology, Food Biosci., 2025, 67, 106030 CrossRef CAS.
- I. Kim, S. H. Seok and H.-Y. Lee, Development of a zebrafish larvae model for diabetic heart failure with reduced ejection fraction, Korean Circ. J., 2023, 53, 34–46 CrossRef CAS PubMed.
- Z. Wang, C. Yuan, T. Huang and B. Lu, Early nutritional interventions for chronic low-grade inflammation, Trends Endocrinol. Metab., 2025, 27, S1043276025001249 Search PubMed.
- J.-C. Peng, W.-J. Qi, H.-Y. Wang, W. Zhou, X.-J. Yu and L. Wang, Blumeatin inhibits LPS-induced inflammation of TLR4/NF-κB signaling pathway via targeting TLR4/MD-2, J. Asian Nat. Prod. Res., 2025, 27, 1496–1509 CrossRef CAS PubMed.
- S. Stojanov, A. Berlec and B. Strukelj, The influence of probiotics on the firmicutes/bacteroidetes ratio in the treatment of obesity and inflammatory bowel disease, Microorganisms, 2020, 8, 1715 CrossRef CAS PubMed.
- A. B. Nongonierma, M. Hennemann, S. Paolella and R. J. FitzGerald, Generation of wheat gluten hydrolysates with dipeptidyl peptidase IV (DPP-IV) inhibitory properties, Food Funct., 2017, 8, 2249–2257 RSC.
- Y. Pan, Y. Song, M. Zhao, M. Yang, N. Xiao, J. Wang and F. Feng, Mulberry leaf extract ameliorates high-fat diet-induced obesity in mice by regulating the gut microbiota and metabolites, Food Bioscience, 2024, 62, 105359 CrossRef CAS.
- X. Luo, W. Liu, H. Zhong, Y. Yan, F. Feng and M. Zhao, Synergistic effect of combined oyster peptide and ginseng extracts on anti-exercise-fatigue and promotion of sexual interest activity in male ICR mice, J. Funct. Foods, 2021, 86, 104700 CrossRef CAS.
- Y. Wang, J. Wu, L. Lu, B. Fang, M. Zhao, F. Feng and J. Wang, The hypouricemic effect and mechanism of tuna (Thunnus albacares) peptide and screening its functional peptide segments, Food Res. Int., 2025, 221, 117181 CrossRef CAS.
- Z.-h. Xia, S.-y. Zhang, Y.-s. Chen, K. Li, W.-b. Chen and Y.-q. Liu, Curcumin anti-diabetic effect mainly correlates with its anti-apoptotic actions and PI3K/Akt signal pathway regulation in the liver, Food Chem. Toxicol., 2020, 146, 111803 CrossRef CAS PubMed.
- M. Zhao, H. Cai, Z. Jiang, Y. Li, H. Zhong, H. Zhang and F. Feng, Glycerol-monolaurate-mediated attenuation of metabolic syndrome is associated with the modulation of gut microbiota in high-fat-diet-fed mice, Mol. Nutr. Food Res., 2019, 63, 1801417 CrossRef PubMed.
- S. Ganjali, G. F. Watts, M. Banach, Z. Reiner, P. Nachtigal and A. Sahebkar, The Yin and Yang of high-density lipoprotein and atherosclerotic cardiovascular disease: focusing on functionality and cholesterol efflux to reframe the HDL hypothesis, Curr. Med. Chem., 2021, 28, 6066–6081 CrossRef CAS.
- A. Gluba-Sagr, R. Olszewski, B. Franczyk, E. Mlynarska, M. Rysz-Gorzynska, J. Rysz, S. Surma, S. Sohum, M. Banach and P. P. Toth, High-density lipoproteins. Part 2. Impact of disease states on functionality, Am. J. Prev. Cardiol., 2025, 23, 101073–101073 CrossRef PubMed.
- D. B. Savage, K. F. Petersen and G. I. Shulman, Disordered lipid metabolism and the pathogenesis of insulin resistance, Physiol. Rev., 2007, 87, 507–520 CrossRef CAS PubMed.
- P. Bjornstad and R. H. Eckel, Pathogenesis of lipid disorders in insulin resistance: a brief review, Curr. Diabetes Rep., 2018, 18, 127 CrossRef PubMed.
- A. C. E. Vreugdenhil, C. H. Rousseau, T. Hartung, J. W. M. Greve, C. van ‘t Veer and W. A. Buurman, Lipopolysaccharide (LPS)-binding protein mediates LPS detoxification by chylomicrons, J. Immunol., 2003, 170, 1399–1405 CrossRef CAS PubMed.
- P. D. Cani, J. Amar, M. A. Iglesias, M. Poggi, C. Knauf, D. Bastelica, A. M. Neyrinck, F. Fava, K. M. Tuohy, C. Chabo, A. l. Waget, E. Delmée, B. a. Cousin, T. Sulpice, B. Chamontin, J. Ferrières, J.-F. o. Tanti, G. R. Gibson, L. Casteilla, N. M. Delzenne, M. C. Alessi and R. m. Burcelin, Metabolic endotoxemia initiates obesity and insulin resistance, Diabetes, 2007, 56, 1761–1772 CrossRef CAS PubMed.
- W. Makena, A. I. Iliya, J. O. Hambolu, J. A. Timbuak, U. E. Umana and N. I. Dibal, Genistein and Momordica charantia L. prevent oxidative stress and upregulate proglucagon and insulin receptor mRNA in diabetic rats, Appl. Physiol., Nutr., Metab., 2022, 47, 41–50 CrossRef CAS.
- A. Everard, C. Belzer, L. Geurts, J. P. Ouwerkerk, C. Druart, L. B. Bindels, Y. Guiot, M. Derrien, G. G. Muccioli, N. M. Delzenne, W. M. de Vos and P. D. Cani, Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 9066–9071 CrossRef CAS PubMed.
- Q. Wang, J. Xiong, Y. He, J. He, M. Cai, Z. Luo, T. Zhang and X. Zhou, Effect of l-arabinose and lactulose combined with Lactobacillus plantarum on obesity induced by a high-fat diet in mice, Food Funct., 2024, 15, 5073–5087 RSC.
- S. T. Im, W. C. Kim, Y.-S. Lee, J.-Y. Kim, K. Lee, G.-W. Oh, J. M. Lee, M.-J. Yim, D.-S. Lee, S.-C. Ko and S.-H. Lee, Phenolic-enriched Rosa rugosa extract ameliorates obesity-associated metabolic disorders and regulates gut microbiota in high-fat diet-fed mice, J. Funct. Foods, 2024, 122, 106548 CrossRef CAS.
- T. Li, J. Gao, M. Du and X. Mao, Bovine α-lactalbumin hydrolysates ameliorate obesity-associated endotoxemia and inflammation in high-fat diet-fed mice through modulation of gut microbiota, Food Funct., 2019, 10, 3368–3378 RSC.
- K. Han, D. Luo, Y. Zou, S. Dong, Z. Wan and X. Yang, Modulation of gut microbiota by soybean 7S globulin peptide that involved lipopolysaccharide-peptide interaction, J. Agric. Food Chem., 2019, 67, 2201–2211 CrossRef CAS PubMed.
- S. Wang, Z. Lv, W. Zhao, L. Wang and N. He, Collagen peptide from Walleye pollock skin attenuated obesity and modulated gut microbiota in high-fat diet-fed mice, J. Funct. Foods, 2020, 74, 104194 CrossRef CAS.
- N. M. Mindrescu, C. Guja, V. Jinga, S. Ispas, A. Curici, R. E. D. Miulescu, A. N. Twakor and A. M. P. Stoian, SGLT-2 inhibitors and metabolic outcomes: a primary data study exploring the microbiota-diabetes connection, Metabolites, 2025, 15, 411 CrossRef CAS PubMed.
- H. Plovier, A. Everard, C. Druart, C. Depommier, M. Van Hul, L. Geurts, J. Chilloux, N. Ottman, T. Duparc, L. Lichtenstein, A. Myridakis, N. M. Delzenne, J. Klievink, A. Bhattacharjee, K. C. H. van der Ark, S. Aalvink, L. O. Martinez, M.-E. Dumas, D. Maiter, A. Loumaye, M. P. Hermans, J.-P. Thissen, C. Belzer, W. M. de Vos and P. D. Cani, A purified membrane protein from Akkermansia muciniphila or the pasteurized bacterium improves metabolism in obese and diabetic mice, Nat. Med., 2017, 23, 107–113 CrossRef CAS.
- R. Zhai, X. Xue, L. Zhang, X. Yang, L. Zhao and C. Zhang, Strain-specific anti-inflammatory properties of two Akkermansia muciniphila strains on chronic colitis in mice, Front. Cell. Infect. Microbiol., 2019, 9, 239 CrossRef CAS PubMed.
- C. Sun, D. J. F. Holstein, N. Garcia-Cubero, Y. Moulla, C. Stroh, A. Dietrich, M. R. Schoen, D. Gaertner, T. Lohmann, M. Dressler, M. Stumvoll, M. Blueher, P. Kovacs and E. Guiu-Jurado, The role of phosphatidylethanolamine N-Methyltransferase (PEMT) and its waist-hip-ratio-associated locus rs4646404 in obesity-related metabolic traits and liver disease, Int. J. Mol. Sci., 2023, 24, 16850 CrossRef CAS PubMed.
- C. S. Hartz, K. M. Nieman, R. L. Jacobs, D. E. Vance and K. L. Schalinske, Hepatic phosphatidylethanolamine n-methyltransferase expression is increased in diabetic rats, J. Nutr., 2006, 136, 3005–3009 CrossRef CAS PubMed.
- M. Watanabe, A. Nakatsuka, K. Murakami, K. Inoue, T. Terami, C. Higuchi, A. Katayama, S. Teshigawara, J. Eguchi, D. Ogawa, E. Watanabe, J. Wada and H. Makino, Pemt deficiency ameliorates endoplasmic reticulum stress in diabetic nephropathy, PLoS One, 2014, 9, e92647 CrossRef PubMed.
- L. Chen, N. Takatani, F. Beppu, K. Miyashita and M. Hosokawa, The effect of n-3 PUFA binding phosphatidylglycerol on metabolic syndrome-related parameters and n-3 PUFA accretion in diabetic/obese KK-Ay mice, Nutrients, 2019, 11, 2866 CrossRef PubMed.
- B.-K. Kim and S.-K. Park, Phosphatidylserine modulates response to oxidative stress through hormesis and increases lifespan via DAF-16 in Caenorhabditis elegans, Biogerontology, 2020, 21, 231–244 CrossRef CAS PubMed.
- W. Du, S. Jiang, S. Yin, R. Wang, C. Zhang, B. C. Yin, J. Li, L. Li, N. Qi, Y. Zhou and B.-C. Ye, The microbiota- dependent tryptophan metabolite alleviates high- fat diet-induced insulin resistance through the hepatic AhR/TSC2/mTORC1 axis, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2400385121 CrossRef CAS.
- D. Marguet, L. Baggio, T. Kobayashi, A. M. Bernard, M. Pierres, P. F. Nielsen, U. Ribel, T. Watanabe, D. J. Drucker and N. Wagtmann, Enhanced insulin secretion and improved glucose tolerance in mice lacking CD26, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 6874–6879 CrossRef CAS PubMed.
- S. L. Conarello, L. Stacey, Z. H. Li, J. Ronan, R. S. Roy, L. Zhu, G. Q. Jiang, F. Liu, J. Woods, E. Zycband, D. E. Moller, N. A. Thornberry and B. B. Zhang, Mice lacking dipeptidyl peptidase IV are protected against obesity and insulin resistance, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 6825–6830 CrossRef CAS PubMed.
- L. Zheng, Q. Xu, L. Lin, X.-A. Zeng, B. Sun and M. Zhao, In vitro metabolic stability of a casein-derived dipeptidyl peptidase-IV (DPP-IV) inhibitory peptide VPYPQ and its controlled release from casein by enzymatic hydrolysis, J. Agric. Food Chem., 2019, 67, 10604–10613 CrossRef CAS PubMed.
- Q. Xu, L. Zheng, M. Huang and M. Zhao, Collagen derived Gly-Pro-type DPP-IV inhibitory peptides: Structure-activity relationship, inhibition kinetics and inhibition mechanism, Food Chem., 2024, 441, 138370 CrossRef CAS PubMed.
- Y. Zhang, R. Chen, X. Chen, Z. Zeng, H. Ma and S. Chen, Dipeptidyl Peptidase IV-Inhibitory Peptides Derived from Silver Carp (Hypophthalmichthys molitrix Val.), Proteins, J. Agric. Food Chem., 2016, 64, 831–839 CrossRef CAS PubMed.
- Y. Xie, J. Wang, S. Wang, R. He, Z. Wang, L. Zhao and W. Ge, Preparation, characterization, and mechanism of DPP-IV inhibitory peptides derived from Bactrian camel milk, Int. J. Biol. Macromol., 2024, 277, 134232 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |