Potential lipid-lowering effects of Northern Thai vegetable extracts on the inhibition of cholesterol absorption: emerging in vitro and in vivo models
Worarat
Rojanaverawong
 ab,
Pannita
Holasut
ab,
Pannita
Holasut
 a,
Jakkapong
Inchai
a,
Kornwalai
Tunkaew
a,
Thanthakan
Saithong
a,
Matina
Chaiwarut†
a,
Sirinya
Taya
c,
Rawiwan
Wongpoomchai
d,
Supakit
Chaipoot
c and
Chutima S.
Vaddhanaphuti
a,
Jakkapong
Inchai
a,
Kornwalai
Tunkaew
a,
Thanthakan
Saithong
a,
Matina
Chaiwarut†
a,
Sirinya
Taya
c,
Rawiwan
Wongpoomchai
d,
Supakit
Chaipoot
c and
Chutima S.
Vaddhanaphuti
 *a
*a
aInnovative Research Unit of Epithelial Transport and Regulation, Department of Physiology, Faculty of Medicine, Chiang Mai University, 50200, Thailand. E-mail: chutima.srimaroeng@cmu.ac.th; Fax: +66-53-935-365; Tel: +66-53-935-362
bOffice of Research Administration, Chiang Mai University, 50200, Thailand
cMultidisciplinary Research Institute, Chiang Mai University, Chiang Mai 50200, Thailand
dDepartment of Biochemistry, Faculty of Medicine, Chiang Mai University, 50200, Thailand
First published on 19th November 2025
Abstract
Obesity has emerged as a major health issue caused by the alteration of lipid profiles, including hypercholesterolemia, hypertriglyceridemia, and dyslipidemia, necessitating the prevention of hyperlipidemia. Several local Thai vegetables have been recognized for their potent anti-oxidant properties. However, there is limited information regarding their lipid-lowering effects. Therefore, this study aims to investigate and identify the mechanisms underlying the cholesterol-reducing effects of functional ingredients derived from the aqueous extracts of seven local Thai vegetables using in vitro, ex vivo, and in vivo models. The findings suggest that the antioxidant-rich extracts have potential to serve as functional ingredients due to their ability to inhibit cholesterol absorption in intestinal colorectal adenocarcinoma (Caco-2) cells. Among these, the aqueous extracts from Piper sarmentosum Roxb. (PSRAE), Solanum torvum Sw. (STSAE), and Acmella oleracea L. (APAE) showed the most potent lipid-lowering effects through increasing cholesterol micelle size and interfering its formation. Moreover, these extracts modulated lipid homeostasis through distinct mechanisms, including the inhibition of cholesterol synthesis and absorption and stimulation of lipid secretion. Lipid-lowering effects were further confirmed in intact jejunal loops, where PSRAE and STSAE demonstrated the highest cholesterol-lowering efficacies. However, a single oral administration in rats revealed that PSRAE decreased the cholesterol levels only in the serum, intestinal and hepatic tissues. Therefore, PSRAE designated as the most potent lipid-lowering candidate. Nonetheless, long-term efficacy and safety of these extracts require further investigation. Thus, a promising set of functional ingredients derived from local Thai vegetables could serve as future foods for controlling hyperlipidemia and preventing non-communicable diseases.
1. Introduction
Obesity is a chronic, complex disease that affects people worldwide, leading to impaired health. According to the World Health Organization (WHO), obesity is measured from a person's weight and height by calculating the body mass index (BMI): weight (kg)/height2 (m2). A BMI score greater than or equal to 30 kg m−2 indicates obesity.1 Obesity occurs due to several interacting factors. Previous studies have suggested that factors contributing to pathophysiological processes include inflammation, energy expenditure, lipid metabolism, and lipid absorption.2 In addition, the consequences could include type 2 diabetes mellitus, cardiovascular disease, hypertension, cancer, and non-alcoholic fatty liver.3,4Lipid transporters have been suggested to play an important role in regulating lipid homeostasis in intestinal, hepatic, and renal epithelial cells. For instance, Niemann-Pick C1-Like 1 (NPC1L1), a polytopic transmembrane protein, is enriched on the apical membrane of proximal jejunum in humans and rodents.3,5 In addition, NPC1L1 is abundantly expressed on the canalicular membrane of hepatocytes in humans.6,7 NPC1L1 is localized on the absorptive enterocytes, mediating the transport of sterol from the extracellular space into the intracellular compartments via the brush border membrane in humans and rodents.3,8 This is consistent with a previous study showing that the inhibition of NPC1L1 led to a decrease in dietary cholesterol absorption, resulting in reduced lipid levels in the plasma of mice.3 Furthermore, the upregulation of NPC1L1 increased cholesterol absorption and lipid accumulation in the liver of mice.3,9,10 Moreover, downregulating the NPC1L1 expression by the activation of peroxisome proliferator-activated receptor (PPAR) and liver X receptor (LXR) results in reduced cholesterol absorption in human colorectal adenocarcinoma (Caco-2) cells and in mice.11,12 Thus, it is suggested that NPC1L1 has high efficacy in inhibiting cholesterol absorption, leading to lower blood cholesterol and resulting in advantageous effects on metabolic syndromes, such as diabetes, obesity, atherosclerosis, and fatty liver. Furthermore, 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR), a rate-limiting enzyme in lipid metabolism that activates cholesterol synthesis, is the main target for lowering cholesterol levels.13,14
Currently, lipid-lowering drugs are the most commonly used for controlling lipid profiles in patients with metabolic syndrome. Ezetimibe is a lipid-lowering drug that blocks NPC1L1, inhibiting cholesterol absorption.15 A previous study also demonstrated that cholesterol, triglyceride, and low-density lipoprotein-cholesterol (LDL) levels decreased, while high-density lipoprotein-cholesterol (HDL) levels increased in obese rats treated with ezetimibe.16 In addition, ezetimibe reduced cholesterol permeability in Caco-2 cells.17 Moreover, ezetimibe was found to reduce LDL and non-HDL levels in dyslipidemia patients.18 Even though drug therapy is recommended to improve hyperlipidemia, drug–drug interactions and toxicity require attention. Thus, investigation of natural products for lipid-lowering effects to reduce risk factors associated with synthetic drugs is desirable.
The local Thai vegetables used in this study are ingredients in North Thai Lanna food, which include Acacia pennata L. Willd (AP), Piper sarmentosum Roxb. (PSR), Eryngium foetidum L. (EFL), Solanum torvum Sw. (STS), Acmella oleracea L. (AO), Lentinus polychrous Berk (LPB), and Coccinia grandis Linn. Voig (CG). These local Thai vegetables have been widely grown in Northern Thailand and exhibit several beneficial effects, including anti-oxidant, anti-diabetic, anti-cancer, anti-microbial, anti-apoptotic, and anti-inflammatory effects.19–26 For example, AP extracts have been shown to inhibit cyclooxygenase 1 (COX-1) and 2 (COX-2) in in vitro assays, thereby reducing inflammation.19 While PSR has been reported to inhibit reactive oxygen species (ROS) production in human neuroblastoma SH-SY5Y cells20 and exert anti-oxidant effects by decreasing malondialdehyde (MDA), inducible nitric oxide synthase (iNOS), nitric oxide (NO), tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6) in gastric ulcer rats.21 Significant anti-cancer and anti-oxidant effects have been shown in human breast cancer MCF7 cells treated with 3 mg mL−1 of EFL.22 The aqueous extract of 1000 µg mL−1 STS exhibited anti-inflammatory effects by inhibiting albumin denaturation and protease activity, as well as stabilizing the human red blood cell membrane.23 The STS aqueous extract at the same concentration also had anti-diabetic effects, inhibiting α-amylase and α-glucosidase and enhancing glucose uptake in in vitro models.23 Consistently, STS improved diabetic conditions by regenerating the morphology of β-cells of islets of Langerhans, leading to increased insulin secretion and decreasing blood glucose in streptozotocin-induced diabetic rats.24 The flavonoid-rich AO exhibited anti-inflammation activity by inhibiting COX-1 and COX-2 in an in vitro study.19 Similarly, LPB reduced TNF-α production and lowered the expression of IL-1β and IL-6 in the macrophages induced by lipopolysaccharide (LPS).25 Furthermore, CG aqueous extract has the potential to serve as an anti-diabetic agent by inhibiting α-glucosidase, α-amylase, and dipeptidyl peptidase IV enzymes.26 Nonetheless, the mechanisms of action of native vegetables on cholesterol absorption are still not fully understood. Therefore, the present study investigated the potential lipid-lowering effects of native vegetables extracted using a water extraction method and the selected fractions were further identified to determine the mechanisms involved. The findings from this study could yield a new set of ingredients for future use as functional ingredients and functional foods with the added benefit of lipid-lowering properties.
2. Materials and methods
2.1. The preparation of local Thai vegetable aqueous extracts
The seven local vegetables were obtained from Terdwalai and local farms in Chiang Mai, Thailand. All local vegetable voucher specimens were deposited at the Faculty of Pharmacy, Chiang Mai University, as shown in Table S1. The aqueous extraction of seven local vegetables was carried out as follows. Fresh vegetables were washed, chopped into small pieces and then blended with water until a homogeneous mixture was achieved. After 15 minutes of boiling, the blended mixture was filtered and then centrifuged at 9000 rpm for 10 minutes. Finally, the supernatant was freeze-dried to provide the aqueous extracts for further investigation.2.2. Chemical constituents of local Thai vegetable aqueous extracts
The contents and compositions of phenolic compounds of local Thai vegetable aqueous extracts were analyzed using high-performance liquid chromatography (HPLC) (Shimadzu, Kyoto, Japan) equipped with an SPD-M20A photodiode array detector and an Inertsil C18 column (250 × 4.6 mm, GL Sciences, CA, USA). The mobile phases consisted of 2% acetic acid in water (A) and 100% acetonitrile (B). Gradient elution followed the method reported by Chaipoot et al.,27 with a total run time of 85 minutes. The flow rate was 1 mL min−1, and the column temperature was set at 30 °C. Nineteen phenolic standards were detected at 280 nm, including gallic acid, theobromine, protocatechuic acid, p-hydroxybenzoic acid, catechin, chlorogenic acid, caffeine, vanillic acid, caffeic acid, syringic acid, epicatechin, vanillin, p-coumaric acid, ferulic acid, sinapic acid, rutin, myricetin, quercetin, and trans-cinnamic acid.2.3. Evaluation of the physicochemical properties of cholesterol micelles
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min, and the supernatant was filtered with a 0.2 µm filter membrane to separate the bound and free bile acids. The free bile acids were mixed with reaction reagents (0.133 M tris-buffer pH 9.5, 1 M hydrazine hydrate, 7.7 mM nicotinamide adenine dinucleotide (NAD), and 1 unit per mL 3α-hydroxysteroid dehydrogenase), then incubated at 37 °C for 2 h. Thereafter, the reaction was monitored at 340 nm to detect the rate of thio-NADH formation. The data will be presented as a percentage of control, as previously reported by Ontawong A. et al.28
000 rpm for 10 min, and the supernatant was filtered with a 0.2 µm filter membrane to separate the bound and free bile acids. The free bile acids were mixed with reaction reagents (0.133 M tris-buffer pH 9.5, 1 M hydrazine hydrate, 7.7 mM nicotinamide adenine dinucleotide (NAD), and 1 unit per mL 3α-hydroxysteroid dehydrogenase), then incubated at 37 °C for 2 h. Thereafter, the reaction was monitored at 340 nm to detect the rate of thio-NADH formation. The data will be presented as a percentage of control, as previously reported by Ontawong A. et al.28
2.4. Cell culture
Human colorectal adenocarcinoma (Caco-2) cell cultures were maintained in Dulbecco's modified Eagle's medium (DMEM), which contained 1% antibiotic-antimycotic and 10% fetal bovine serum (FBS). The cells were grown in an incubator containing 95% O2, 5% CO2, and at a temperature of 37 °C. For additional testing, cells were cultivated in 75 cm2 flasks and 96-well plates.2.5. Determination of cell viability
Cell viability of Caco-2 cells was detected with a 3-[4,5-dimethylthiazol-2-yl]-2,3-diphenyl tetrazolium bromide (MTT) assay. First, Caco-2 cells were cultured on 96-well plates for 21 days, followed by treating with AP, PSR, EFL, STS, AO, LPB, and CG aqueous extracts at a dosage of 1 mg mL−1 and ezetimibe at a dosage of 30 μg mL−1 for 24 h. Thereafter, the MTT solution was added to the cells containing the culture solution and incubated for 4 hours. Subsequently, the culture solution was removed and the formazan precipitate was dissolved in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH (1
EtOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v). Eventually, it was measured with a microplate reader at a wavelength of 570 nm.
1 v/v). Eventually, it was measured with a microplate reader at a wavelength of 570 nm.
2.6. Determination of cholesterol transport in human intestinal Caco-2 cells
Caco-2 cells were seeded at 5 × 104 cells per well in a 24-well plate and cultured for 21 days. After that, the cells were incubated with APAE, PSRAE, EFLAE, STSAE, AOAE, LPBAE, CGAE, and ezetimibe at a dosage of 30 μg mL−1, which incorporated the micelle and 10 µM 25-[N-[(7-nitro-2-1,3-benzoxadiazol-4-yl) methyl] amino]-27-norcholesterol (25-NBD-cholesterol) (Avanti, Alabama, USA) for 2 h. After that, the treated medium was removed and washed 2 times with Dulbecco's phosphate-buffered saline (DPBS). The fluorescence intensity was measured with a high content microscope (ImageXpress Micro 4 High Content Imaging System) (Molecular Devices, CA, USA) at Ex/Em = 482 and 536 nm, respectively.2.7. Determination of cholesterol transport in ex vivo rat jejunal loops
To investigate the inhibitory effect of local Thai vegetable aqueous extracts on cholesterol transport through intestinal epithelial cells in rats, [3H]-cholesterol, a substance with properties similar to regular cholesterol as previously reported,37 was applied in both ex vivo and in vivo cholesterol absorption. Male Wistar rats from Nomura Siam International (Bangkok, Thailand), weighing 300–350 g, were used. In this study, the facilities and protocols for the animals were approved by the Laboratory Animal Care and Use Committee of the Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand (Protocol no: 03/2567). After sacrifice, the jejunum segments were separated and cleaned. Then, the jejunum segments were tied into 1.5 cm segments and injected with 2 μM cholesterol containing 1 μCi mL−1 of radiolabelled cholesterol micelles ([3H]-cholesterol, specificity 49 Ci mmol−1) combined with 1 mg mL−1 of local Thai vegetable aqueous extracts and ezetimibe (30 μg mL−1), and incubated for 30 min at room temperature. After that, the jejunum segments were cut, the epithelial tissues were separated, and the radioactivity was measured using a liquid scintillation counter (PerkinElmer, Waltham, MA, USA).2.8. Determination of cholesterol levels in hypercholesterolemic rats
To confirm whether the most potent local Thai vegetable aqueous extracts inhibited cholesterol absorption in rats, a single dose of the tested compound and [3H]-cholesterol micelle solution was administered to rats. After fasting for 6–8 hours, 2 μM cholesterol containing 10 μCi mL−1 of radiolabelled cholesterol micelles ([3H]-cholesterol, specificity 49 Ci mmol−1) with 1 mg mL−1 of tested compounds (details shown in figure legend) and 30 μg per kg BW of ezetimibe, an effective inhibitor for cholesterol absorption,29 were administered to the experimental animals. After that, blood was collected from the lateral tail vein at 0, 4, 8, 12, 24, 28, 32, and 48 hours after feeding. At the end of this experiment, the animals were euthanized to collect the blood, liver, and jejunum tissues. The tissues were lysed with 1 N sodium hydroxide, and the blood samples were centrifuged to separate the plasma. The plasma and lysed tissues were measured for radioactivity using a liquid scintillation counter (PerkinElmer, Waltham, MA, USA).2.9. Determination of cholesterol transporter gene expression in rat intestinal epithelial cells
The quantitative real-time polymerase chain reaction (qPCR) technique was used to analyze the cholesterol transporter gene expression. The total RNA was extracted with TRIzol™ Reagent (Thermo Fisher Scientific, MA, USA) and converted to first-strand cDNA by a commercial first-strand cDNA kit (Bio-Rad, PA, USA). In addition, the primers listed in Table 1 were used to amplify the target genes, and gene expression was examined by using the SYBR green real-time PCR master mix (Bioline, London, UK).| cDNA | GenBank accession no. | Forward primers 5′ to 3′ | Reverse primers 5′ to 3′ | Amplicon size (bp) | Ref. |
|---|---|---|---|---|---|
| Human primers | |||||
| hNPC1L1 | NM_013389.3 | TGCTGTGTCCCTCATCAACC | TCTGAATGAGCTGGGCCTTG | 218 | — |
| hABCG5 | NM_022436.3 | CGCGTAGGTCTCCTTTACCA | AGTGCATAGGCCAGCATCAT | 152 | 30 |
| hABCG8 | NM_022437.3 | GCCGCCCTCTTGTTCATG | TAACATTTGGAGATGACATCCAGAA | 71 | 31 |
| hPPARα | NM_005036.6 | CAGAACAAGGAGGCGGAGGTC | TTCAGGTCCAAGTTTGCGAAGC | 119 | 32 |
| hHMGRC | NM_000859.3 | CACCAAGAAGACAGCCTGAA | CATCCTCCACAAGACATTGC | 121 | 33 |
| hGAPDH | NM_001357943.2 | CGGAGTCAACGGATTTGGTCG | AGCCTTCTCCATGGTGGTGAAGAC | 253 | 34 |
| Rat primers | |||||
| rABCG5 | NM_053754.2 | GGGAAGTGTTTGTGAACGGC | GTGTATCTCAGCGTCTCCCG | 121 | 35 |
| rABCG8 | NM_130414.2 | CGTCAGATTTCCAATGACTTCCG | TCCGTCCTCCAGTTCATAGTACA | 243 | 36 |
| rHMGCR | NM_013134.2 | TGCTGCTTTGGCTGTATGT | TGAGCGTGAACAAGAACCA | 230 | — |
| rLXRα | NM_031627.2 | GCTCTCCGAGATCTGGG | TCCTCAGTCTGCTCCACC | 109 | 28 |
| rNPC1L1 | NM_001002025.1 | GAGGCCTCACGTCACTATCC | AAAAGTGGTATTGGGGCCGT | 128 | — |
| rPPARα | NM013196.1 | AATCCACGAAGCCTACCTGA | GTCTTCTCAGCCATGCACAA | 132 | 37 |
| rβ-actin | NM_031144.3 | CCTAAGGCCAACCGTGAAAA | GGAGCGCGTAACCCTCATAC | 181 | 37 |
2.10. Determination of protein expression of cholesterol synthesis and transporters in rat intestinal epithelial cells
The protein expression was detected using the western blotting technique. The cells were lysed with cold lysis buffer (CelLytic MT, Sigma-Aldrich, USA) containing 1% protease inhibitor (Roche, MA, Germany) in order to obtain subcellular fractions. After homogenizing the lysate sample, it was centrifuged at 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 10 minutes at 4 °C. The supernatant was termed the whole cell lysate. Additionally, from the entire cell lysate, cell membrane and cytosolic fractions were obtained by centrifugation at 100
000g for 10 minutes at 4 °C. The supernatant was termed the whole cell lysate. Additionally, from the entire cell lysate, cell membrane and cytosolic fractions were obtained by centrifugation at 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 2 hours at 4 °C. In this phase, the pellet was resuspended in the same lysis buffer and labelled as the membrane fraction, while the supernatant was collected and labelled as the cytosolic fraction. Before use in additional investigations, the samples were stored at −80 °C. Protein concentrations in the samples were ascertained using the Bradford protein assay (BioRad, PA, USA).
000g for 2 hours at 4 °C. In this phase, the pellet was resuspended in the same lysis buffer and labelled as the membrane fraction, while the supernatant was collected and labelled as the cytosolic fraction. Before use in additional investigations, the samples were stored at −80 °C. Protein concentrations in the samples were ascertained using the Bradford protein assay (BioRad, PA, USA).
2.11. Statistical analysis
All statistical analyses were performed with GraphPad Prism 10 software (San Diego, CA, USA). The data were expressed as the mean ± standard error of mean (SEM). Statistical significance was analyzed by one-way analysis of variance (ANOVA) followed by Tukey's method for multiple comparisons. P-Value < 0.05 was considered statistically significant.3. Results
3.1. The contents and compositions of phenolic compounds in local Thai vegetable aqueous extracts
The composition and contents of phenolic compounds in seven local Thai vegetable aqueous extracts were investigated using HPLC, as shown in Table 2. The results indicated that aqueous extracts from various Thai local vegetables had diverse profiles and levels of phenolic compounds, with composition and total phenolic content differing depending on the kind of vegetable. These included gallic acid, theobromine, protocatechuic acid, p-hydroxybenzoic acid, catechin, chlorogenic acid, caffeine, vanillic acid, caffeic acid, syringic acid, epicatechin, vanillin, p-coumaric acid, ferulic acid, sinapic acid, rutin, myricetin, quercetin, and trans-cinnamic acid. These results revealed that the local Thai vegetable aqueous extracts had high anti-oxidant capacity and total phenolic content in the 1.05–138.85 mg g−1 range, contingent upon the species.| Phenolic compounds | Local Thai vegetable aqueous extracts (mg g−1 sample) | ||||||
|---|---|---|---|---|---|---|---|
| APAE | PSRAE | EFLAE | STSAE | AOAE | LPBAE | CGAE | |
| Data represented as mean ± SEM, ND = not detected. Acacia pennata L. Willd (AP), Piper sarmentosum Roxb. (PSR), Eryngium foetidum L. (EFL), Solanum torvum Sw. (STS), Acmella oleracea L. (AO), Lentinus polychrous Berk (LPB), Coccinia grandis Linn. Voig (CG), and aqueous extraction (AE). | |||||||
| 1. Gallic acid | 0.36 ± 0.01 | 0.44 ± 0.01 | ND | ND | 0.89 ± 0.01 | ND | 0.97 ± 0.01 |
| 2. Theobromine | 0.20 ± 0.02 | ND | ND | ND | 0.03 ± 0.01 | ND | ND |
| 3. Protocatechuic acid | ND | ND | ND | ND | 0.38 ± 0.01 | ND | ND |
| 4. p-Hydroxybenzoic acid | ND | ND | ND | ND | 0.72 ± 0.04 | ND | 1.98 ± 0.02 |
| 5. Catechin | ND | ND | ND | 2.53 ± 0.04 | 0.12 ± 0.01 | ND | 3.83 ± 0.11 |
| 6. Chlorogenic acid | ND | 2.75 ± 0.06 | ND | ND | 8.83 ± 0.02 | 0.82 ± 0.01 | ND |
| 7. Caffeine | ND | 1.13 ± 0.03 | ND | ND | ND | ND | ND |
| 8. Vanillic acid | 28.34 ± 0.06 | ND | 4.51 ± 0.03 | 10.75 ± 0.06 | 1.84 ± 0.22 | ND | 7.11 ± 0.27 |
| 9. Caffeic acid | ND | ND | ND | ND | 1.02 ± 0.05 | ND | ND |
| 10. Syringic acid | ND | ND | ND | ND | ND | 0.15 ± 0.01 | ND |
| 11. Epicatechin | ND | 5.80 ± 0.01 | ND | ND | ND | ND | ND |
| 12. Vanillin | ND | ND | ND | 0.23 ± 0.01 | 0.05 ± 0.01 | ND | ND |
| 13. p-Coumaric acid | 0.28 ± 0.01 | ND | 0.12 ± 0.01 | 0.13 ± 0.01 | 0.27 ± 0.05 | ND | 0.14 ± 0.01 |
| 14. Ferulic acid | ND | ND | ND | 0.12 ± 0.01 | 0.35 ± 0.01 | ND | ND |
| 15. Sinapic acid | 2.02 ± 0.01 | ND | ND | 0.59 ± 0.02 | ND | ND | ND |
| 16. Rutin | ND | ND | ND | ND | 2.65 ± 0.44 | ND | ND |
| 17. Myricetin | ND | ND | 134.21 ± 1.53 | 25.70 ± 1.08 | 23.30 ± 2.14 | ND | ND |
| 18. Quercetin and trans-cinnamic acid | 0.44 ± 0.01 | 14.51 ± 0.18 | ND | 0.19 ± 0.01 | 0.25 ± 0.13 | 0.08 ± 0.01 | 0.06 ± 0.01 |
| Total phenolic compound | 31.63 | 24.63 | 138.85 | 40.26 | 40.69 | 1.05 | 14.09 |
3.2. Effect of local Thai vegetable aqueous extracts on cholesterol transport and cell viability in human intestinal Caco-2 cells
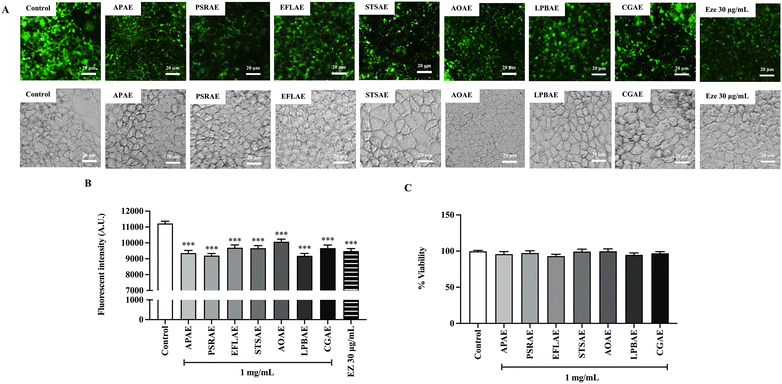
Fig. 1A and B illustrates the fluorescence intensity in Caco-2 cells treated with aqueous extracts of local Thai vegetables. Results showed that all seven local Thai vegetables had significantly reduced fluorescence intensities similar to those in the ezetimibe group, indicating that the aqueous extracts of all seven species have the potential to reduce cholesterol absorption by intestinal cells. Subsequently, cell viability assays were performed to measure cell survival following treatment with aqueous extracts of local Thai vegetables. Fig. 1C shows that the Caco-2 cell viability did not differ among experimental groups. Thus, the existing potential cholesterol-lowering effect of seven local Thai vegetables was not due to cell death.3.3. Effect of local Thai vegetables on physiochemical parameters in the intestinal Caco-2 cells
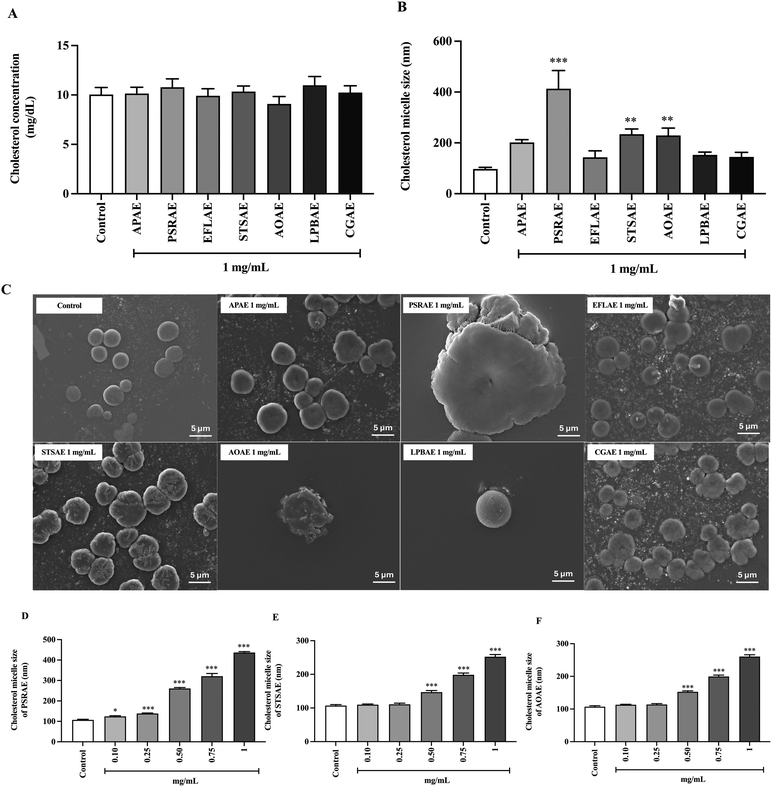
To determine the effect of seven local Thai vegetable aqueous extracts on cholesterol solubility in cholesterol micelles, an in vitro assay was applied. Fig. 2A shows that the cholesterol concentration did not change when treated with APAE, PSRAE, EFLAE, STSAE, AOAE, LPBAE, and CGAE. Thus, the aqueous extracts did not influence cholesterol solubility in lipid-mixed micelles. On the other hand, data on cholesterol micelle size are presented in Fig. 2B, showing that the aqueous extracts of PSR, STS, and AO significantly increased the cholesterol micelle size when compared with the control group. Fig. 2C depicts representative images of cholesterol micelle size in each experimental group obtained using scanning electron microscopy. Consistently, PSRAE, STSAE, and AOAE showed a dose-dependent increase in cholesterol micelle size, with the highest potency and degree of enlargement observed in PSRAE treatment (Fig. 2D–F). These data implied that 3 species of local Thai vegetable extracts, including PSRAE, STSAE, and AOAE, had lipid-lowering activity, partly by increasing micellar cholesterol content.3.4. Effect of local Thai vegetable aqueous extracts on bile acid binding activity
All seven aqueous extracts of local Thai vegetables have the potential to inhibit cholesterol absorption in Caco-2 cells, and the most potent of the three vegetables, PRPAE, STSAE, and AOAE, exerted their effect by increasing cholesterol micelle size. Therefore, PRPAE, STSAE, and AOAE were selected to identify the possible mechanisms by which these species interfere with the physicochemical properties of micelle cholesterol. The affinity of the Thai local species, PRPAE, STSAE, and AOAE, with three bile acids, including taurocholic acid, taurodeoxycholate, and glycodeoxycholate, was then determined. As presented in Table 3, AOAE significantly bound to all of the bile acids, similar to the effect of cholestyramine at a dosage of 1 mg mL−1, while STSAE significantly bound to both taurocholic acid and glycodeoxycholate, and PSR markedly bound to only taurocholic acid. This finding indicated that the selected local Thai vegetables, PSRAE, STSAE, and AOAE, possessed high binding affinity for bile acids, thereby interfering with cholesterol micellar formation and further reducing cholesterol absorption.| Samples | (mg mL−1) | Bile acids (%) | ||
|---|---|---|---|---|
| Taurocholic acid | Glycodeoxycholate | Taurodeoxycholate | ||
| Acacia pennata L. Willd (AP), Piper sarmentosum Roxb. (PSR), Eryngium foetidum L. (EFL), Solanum torvum Sw. (STS), Acmella oleracea L. (AO), Lentinus polychrous Berk (LPB), and Coccinia grandis Linn. Voig (CG), and aqueous extraction (AE). Data presented as % of control, **p < 0.01 and ***p < 0.001 vs. control. | ||||
| Control | 1 | 0.00 | 0.00 | 0.00 |
| PSRAE | 1 | 88.26 ± 18.51** | 0.00 | 0.00 |
| STSAE | 1 | 174.29 ± 21.38*** | 107.73 ± 9.51*** | 0.00 |
| AOAE | 1 | 117.99 ± 34.46*** | 160.90 ± 22.88*** | 95.18 ± 30.35*** |
| Cholestyramine | 1 | 105.22 ± 9.16** | 156.38 ± 11.76*** | 118.01 ± 9.07*** |
3.5. Effect of local Thai vegetable aqueous extracts on genes involved in lipid metabolism and transporter expression in intestinal Caco-2 cells
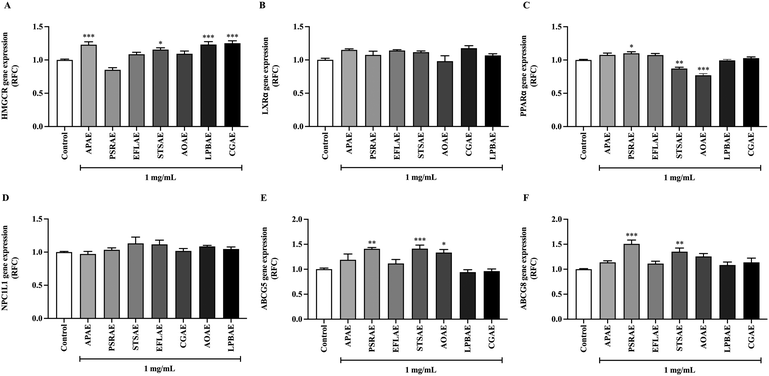
Expression of genes related to lipid metabolism and transporters was determined by the qPCR technique. As shown in Fig. 3, the extracts from local Thai vegetables, APAE, STSAE, LPBAE, and CGAE, showed a comparable degree of increasing the expression of the HMGCR gene, a rate-limiting step for cholesterol synthesis, while PSRAE tended to decrease the HMGCR gene expression. On the other hand, the expression of PPARα, a gene related to β-oxidation, was increased when treated with PSRAE and decreased when treated with STSAE and LPBAE. In addition, the expression of genes encoding efflux transporters, ABCG5 and ABCG8, was enhanced when treated with PRSAE and STSAE, while the treatment with AOAE solely increased the expression of ABCG8. However, there were no significant changes observed in LXRα and influx transporter, the NPC1L1 gene expression by any compounds, suggesting that local Thai vegetables could possibly modulate lipid metabolism and transport.3.6. Effect of local Thai vegetables on total cholesterol and triglyceride levels in media
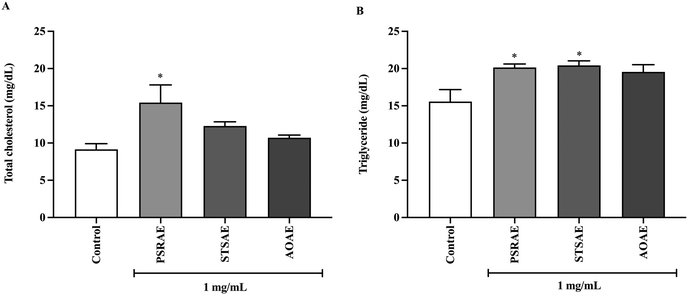
Since previous data showed alterations in genes encoding efflux lipid transporters, possible mechanisms by which local Thai vegetables reduce cholesterol absorption through stimulation of lipid efflux were proposed. Thus, the total cholesterol and triglyceride levels were determined in cell culture media. As expected, total cholesterol and triglyceride levels in the media were increased when treated with PSRAE, while treatment with STSAE and AOAE enhanced triglyceride efflux in the media with no significant change in total cholesterol levels. Hence, PSRAE had higher efficacy in stimulating lipid efflux than STSAE and AOAE (Fig. 4).3.7. Effect of local Thai vegetable aqueous extracts on cholesterol transport in ex vivo rat jejunal loops
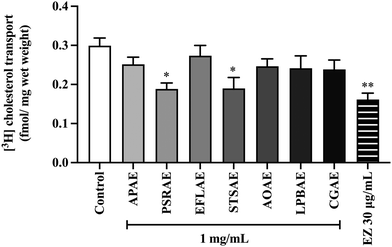
As shown in Fig. 5, cholesterol absorption was measured by cholesterol transport into the intact jejunal loops. Among the tested compounds, PSRAE and STSAE significantly decreased the level of [3H]-cholesterol uptake compared with the control group. Similarly, ezetimibe at the dose of 30 μg mL−1 showed a significantly higher degree of reduction of [3H]-cholesterol levels in comparison with the control group. This data suggested that PSRAE and STSAE were the most efficient extracts in lowering cholesterol levels among other local Thai vegetable extracts.3.8. Effect of local Thai vegetable aqueous extracts on serum cholesterol levels in rats
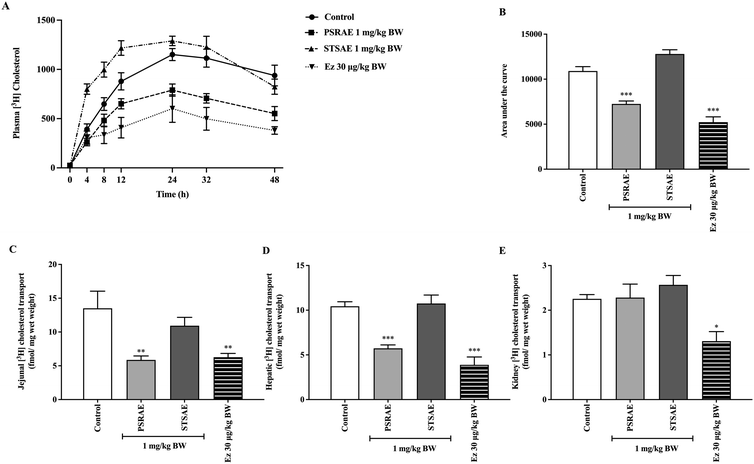
According to prior experiments, PSRAE and STSAE dose-dependently increased the size of cholesterol micelles, interfered with bile acid binding capacity, and stimulated lipid efflux transport, which, in turn, reduced the intestinal cholesterol absorption in both Caco-2 cells and rat jejunal epithelium. To confirm this hypothesis in an in vivo model, co-administration of the most potent local Thai vegetable aqueous extracts, PSRAE and STSAE, with [3H]-cholesterol micelles was performed. Surprisingly, PSRAE significantly decreased cholesterol levels in serum, intestinal, and liver tissues without any effect on accumulation in the kidney. This data was consistent with the effects of ezetimibe. However, there was no significant change in cholesterol content in both serum and tissues, as shown in Fig. 6. This finding indicated that PSRAE was the most potent lipid-lowering candidate among the local Thai vegetables in this study.3.9. Effect of local Thai vegetable aqueous extracts on the expression of genes and proteins involved in lipid metabolism and lipid transporters in the intact rat intestinal epithelial cells
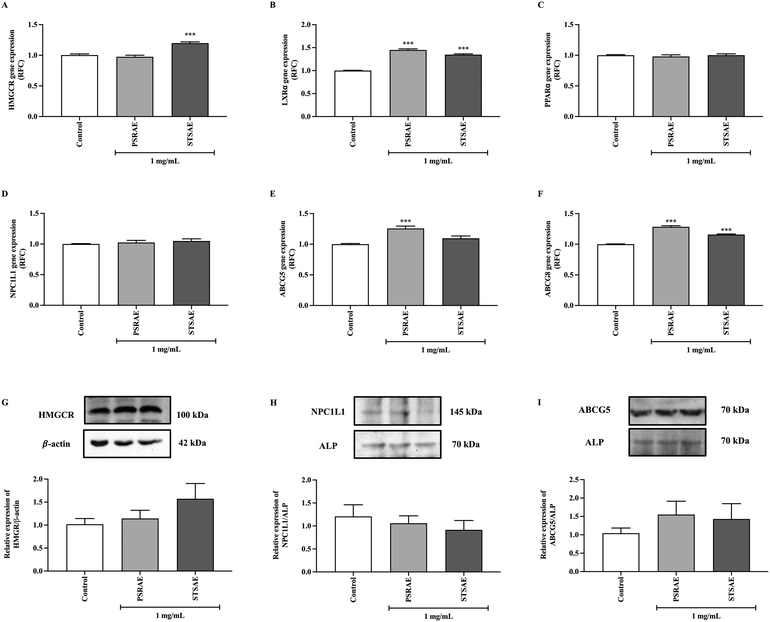
To further confirm the cholesterol-lowering effects of local Thai vegetable aqueous extracts in intact tissues, the expression of genes and proteins associated with lipid metabolism and lipid transporters was assessed by qPCR and western blotting, respectively. As shown in Fig. 7A, the HMGCR gene was significantly increased in the STSAE group when compared with the control group, while its protein expression tended to increase (Fig. 7G). While LXRα gene was increased in both PSRAE and STSAE groups when compared with control (Fig. 7B), no significant alteration of genes encoding PPARα and NPC1L1 was observed in any tested compounds (Fig. 7C and D). Genes involved in cholesterol efflux transporters, including the heterodimer of ABCG5 and ABCG8, were upregulated. The former gene was induced by PSRAE, while the latter was induced by both STSAE and PSRAE (Fig. 7E and F). Similarly, the cholesterol influx transporter NPC1L1 did not differ among experimental groups (Fig. 7H). Although there was no reliable antibody for ABCG8 protein detection, the ABCG5 protein tended to increase by both STSAE and PSRAE when compared with control cells (Fig. 7I). These data indicate that acute exposure to selective local Thai vegetables, PSRAE and STSAE, could alter genes and proteins involved in lipid metabolism and lipid excretion.4. Discussion
Lipids are significant biomolecules in the human body. For instance, cholesterol is an important element of the cell membrane and is a major component in the synthesis of steroid hormones and bile acids.38 However, excessive cholesterol levels mostly lead to adverse health events. Thus, a lipid-lowering strategy aims to reduce the risk of widespread diseases, such as diabetes, obesity, atherosclerosis, and fatty liver disease. In this research, we evaluated the beneficial effects of aqueous extracts from seven species of local Thai vegetables used in North Thai Lanna food. These included Acacia pennata L. Willd (AP), Piper sarmentosum Roxb. (PSR), Eryngium foetidum L. (EFL), Solanum torvum Sw. (STS), Acmella oleracea L. (AO), Lentinus polychrous Berk (LPB), and Coccinia grandis Linn. Voig (CG). According to a previous study, these local Thai vegetables exhibit anti-oxidant, anti-diabetic, anti-cancer, anti-apoptotic, and anti-inflammatory properties.19–26 Among these, some of the active compounds identified in the local Thai vegetable aqueous extracts have previously been reported to have lipid-lowering effects through several mechanisms. For instance, gallic acid, found in PSRAE and AOAE in this study, at a dose of 0.2 mg g−1 downregulated PPARα expression, a key transcription factor inducing β-oxidation in diabetic NAFLD mice.39 The coffee fruit extracts rich in gallic acid increased the cholesterol micelle sizes and decreased the cholesterol micelle solubility, leading to reduced cholesterol absorption.40 In addition, gallic acid in grape seed has been shown to reduce the formation of cholesterol micelles and cholesterol micelle solubility.41 Besides gallic acid, this study revealed the enrichment of chlorogenic acid in PSRAE and AOAE. This active compound at a dose of 100 mg g−1 day−1 could potentially maintain cholesterol homeostasis in both hypercholesterolemic mice and human HepG2 and Caco2 cell lines by reducing the expression of NPC1L1 and HMGCR mediated by two mechanisms: directly targeted to AMP-activated protein kinase (AMPK)/sterol regulatory element-binding protein 2 (SREBP2) pathway and indirectly activated AMPK/SREBP2/heat shock protein 90 (HSP90)/pregnane X receptor (PXR) pathway.42 Furthermore, catechin, a major flavonoid group found in STSAE and AOAE of this study, has also been shown to decrease cholesterol micelle solubility in an in vitro study, which then could potentially reduce cholesterol absorption.43The present study initially evaluated the potential of local Thai vegetable aqueous extracts in lowering cholesterol absorption using in vitro studies. As proper physicochemical characteristics of cholesterol micelles are a critical step for intestinal absorption in humans,28,44 this study illustrates the disruption of cholesterol micellar formation by 3 major local Thai vegetables in aqueous extracts – PSRAE, STSAE, and AOAE – leading to increased cholesterol micelle size, thereby reducing cholesterol absorption. In fact, it has been recognized that the absorbable emulsified lipid droplets driven by NPC1L1 typically range from 0.1–100 μm.45 Hence, the presence of bulky cholesterol micelles, approximately 2–4-fold larger than control micelles due to the 3 extracts in this study, could enwrap the binding site of the NPC1L1 pocket, hindering cholesterol absorption. Since these 3 candidates also interfered with the binding affinity for both primary and secondary bile acids, they could decrease the bile acid pool and micelle stability, reflecting unfavorable bile salt and phospholipid compositions and concomitantly reduced cholesterol uptake into enterocytes.46 This evidence is in agreement with several studies. For instance, an increase in cholesterol micelle size due to egg-yolk phospholipids led to attenuation of cholesterol absorption in Caco-2 cells.47 Our previous study also reported that the increment of cholesterol micelle size, decrement of cholesterol micelle solubility, and disturbance of bile acid binding capacity resulted in reduced cholesterol absorption.32 Similarly, the earlier study by Trisat et al. (2016) found that the Acacia pennata L. Willd was able to effectively bind with bile acids, leading to decreased lipid absorption.48 The use of water extracts of local vegetables from this study provides a unique advantage by reflecting the traditional mode of preparation and consumption in Northern Thailand, where boiling and aqueous cooking are common. Unlike isolated dietary fibers, the water extract contains a complex mixture of soluble polysaccharides and phenolic compounds that can synergistically influence cholesterol micelle formation and bile acid binding. In relative comparison with other previous works on plant aqueous extracts in the literature, including green tea, bitter melon, and okra, the extracts examined in this study demonstrated modest or comparable or higher inhibition of cholesterol micellar solubility, suggesting that these underexplored vegetables could serve as promising candidates for the development of culturally relevant functional ingredients.49–51 Likewise, the soluble fiber from oat bran water extract exerted a lipid-lowering effect by binding to bile acids and cholesterol, leading to decreased cholesterol absorption and increased rate of cholesterol excretion out of the body in feces.52 Although the aqueous extract from other 4 local Thai vegetables – APAE, EFLAE, LPBAE, and CGAE – had no effect on the changes of physicochemical properties of the cholesterol micelle, it cannot exclude the other possible mechanisms of a modest degree on lipid-lowering action in the Caco-2 cells.
While lipid synthesis plays a crucial role in maintaining cellular structure, membrane integrity, and overall lipid homeostasis,46,53 lipid transporters are specialized proteins responsible for facilitating the movement of lipids across cellular membranes throughout the body and regulating lipoprotein metabolism.54 Dysregulation of either one or both processes has been implicated in the pathogenesis of various metabolic disorders, such as atherosclerosis, obesity, and metabolic syndrome.46 Although our tested compounds – APAE, STSAE, LPBAE, and CGAE – exhibited minimal gene-inducible effects (1.08–1.25 folds vs. control) on the expression of HMGCR, the rate-limiting enzyme for cholesterol synthesis, while STSAE and AOAE down-regulated PPARα, a gene involved in β-oxidation, the most potent compound – PSRAE – showed the positively opposed effects on reducing the lipid levels by downregulating HMGCR and upregulating PPARα gene expression. Likewise, a previous study showed that the reduction of the HMGCR gene expression by black chokeberry led to the attenuation of lipid synthesis in Caco-2 cells.55 Apart from that, a previous study showed that Lactobacillus rhamnosus BFE 5264 and Lactobacillus plantarum NR74 increased ABCG5 and ABCG8 gene and protein expressions, thereby facilitating cholesterol efflux and decreasing intracellular cholesterol levels in the Caco-2 cells.56 The upregulation of PPARα expression by Lactobacillus rhamnosus ATCC 53103 and Lactiplantibacilus plantarum MGEL20154 resulted in the downregulation of NPC1L1 expression, leading to a lower content of cholesterol in Caco-2 cells.57 Corresponding to the effect of cholesterol efflux transporters, the 3 local Thai vegetable aqueous extracts, particularly PSRAE, activated the cholesterol and triglyceride excretion in Caco-2 cells in this study. The ability to inhibit cholesterol absorption in rat intestinal epithelial cells and in the plasma by the most potent extract, PSRAE, was consistent with its effects in Caco-2 cells. This data was consistent with up-regulation of ABCG5 and ABCG8, gene expression by PSRAE in intestinal epithelial cells in vivo. Furthermore, induction of LXRα by PSRAE, the upstream transcription factor for ABCG5 and ABCG8 protein synthesis, was also observed in this study.58,59 This evidence strongly supported the novel and concomitant mechanisms of local Thai vegetables, particularly PSRAE, in inhibiting cholesterol absorption by altering cholesterol micelle size and formation, along with accelerating lipid secretion through major lipid efflux transporters. The findings of this present study clearly illustrate a scientific advancement beyond that of other vegetables found in various regions. For instance, flavonoids isolated from foxtail millet showed strong potential in managing blood lipid levels and reducing lipid absorption mainly through suppression of the free fatty acid uptake transporter, fatty acid translocase (CD36) expression.56 In addition, the pectic polysaccharide extracted from unripe fruits of Rubus chingii Hu significantly reduced serum triglyceride levels, inhibited intestinal lipid absorption, and effectively ameliorated dyslipidemia in rats.57 In addition, catechins from green tea extract reduced gene expression of NPC1L1 and increased the expression of ABCG5/8, thereby inhibiting cholesterol absorption and promoting cholesterol excretion in high-fat diet-fed rats.60 From the aforementioned studies, it is evident that these extracts primarily possess the ability to reduce lipid absorption, whereas coexisting mechanisms contributing to lipid excretion and inhibition of lipid synthesis were observed with that of the potent local Thai vegetable extract, PSRAE. Nonetheless, the cholesterol-lowering effects of PSRAE and its coexistent mechanisms in long-term conditions and/or obesity-induced hyperlipidemic conditions require further investigation. As a promising local Thai vegetable extract, particularly PSRAE, display novel mechanisms and are more comprehensive and effective in regulating lipid homeostasis; therefore, it has the potential to be developed as functional ingredients and/or functional foods.
5. Conclusion
Based on the overall results of this study, it can be summarized that local Thai vegetable aqueous extracts have the potential to reduce cholesterol absorption, particularly PSRAE, STSAE, and AOAE. These effects could be due to an increase in cholesterol micelle size and cholesterol excretion, and to a high binding affinity for bile acids, resulting in a decrease in cholesterol levels. Moreover, the local Thai vegetables regulated cholesterol homeostasis by interfering with cholesterol synthesis, cholesterol absorption and secretion, leading to a decrease in plasma cholesterol. This study also suggests that this promising set of local Thai vegetables, which could be further developed as functional ingredients, could have beneficial effects in protecting against hypercholesterolemia and subsequently in preventing several risk pathological conditions, including diabetes, metabolic syndrome, and cardiovascular diseases. However, further studies are needed to investigate the detailed mechanisms of action and the efficacy of local Thai vegetables in both animal and clinical studies.Author contributions
Worarat Rojanaverawong: methodology, investigation, formal analysis, data curation, visualization, writing – original draft, review & editing. Pannita Holasut: methodology, investigation, formal analysis, visualization, data curation, writing – review & editing. Jakkapong Inchai: methodology, investigation, formal analysis, visualization, data curation writing – review & editing. Thanthakan Saithong: methodology, investigation, formal analysis, visualization, data curation, writing – review & editing. Kornwalai Tunkaew: methodology, investigation, formal analysis, visualization, data curation, writing – review & editing. Matina Chaiwarut: investigation, writing – review & editing. Sirinya Taya: methodology, investigation, formal analysis, visualization, data curation, writing – review & editing. Rawiwan Wongpoomchai: conceptualization, supervision. Chutima S. Vaddhanaphuti: visualization, conceptualization, supervision, data curation, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors confirm that the data supporting the findings of this study are available within the article. Raw data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5fo03247d.
Acknowledgements
The authors would like to thank Mr Nobphaphit Saeyang and Mrs Umaporn Phusaen for technical assistance and the Medical Science Research Equipment Center (MSREC) for access to research facilities. This research project was supported by the Fundamental Fund 2023, Chiang Mai University (grant number FF66/027 to W.R., S.T., R.W., C.S.V), the Chiang Mai University, Chiang Mai, Thailand (to W.R. and C.S.V), the Research and Researcher for Industry (RRi) from the National Research Council of Thailand (NRCT) (no. N41A640337 to C. S. V. and J. I.), the NSRF via the Program Management Unit for Human Resources & Institutional Development, Research and Innovation (grant number B05F640169 to C.S.V), the Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand (no. 1/2566 to C. S. V. and J. I.).References
- World Health Organization , Obesity and overweight. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight. Accessed, 2024.
- M. Huang, J. Zheng, L. Chen, S. You and H. Huang, Role of apolipoproteins in the pathogenesis of obesity, Clin. Chim. Acta, 2023, 545, 117359 CrossRef CAS.
- B. Liu, J. Zhang, P. Sun, R. Yi, X. Han and X. Zhao, Raw Bowl Tea (Tuocha) Polyphenol Prevention of Nonalcoholic Fatty Liver Disease by Regulating Intestinal Function in Mice, Biomolecules, 2019, 9(9), 435 CrossRef CAS PubMed.
- B. Nussbaumerova and H. Rosolova, Obesity and Dyslipidemia, Curr. Atheroscler. Rep., 2023, 25(12), 947–955 CrossRef CAS PubMed.
- R. Hang, W. Liu and J. Zeng, et al., Niemann-Pick C1-Like 1 inhibitors for reducing cholesterol absorption, Eur. J. Med. Chem., 2022, 230, 114111 CrossRef.
- L. Jia, J. L. Betters and L. Yu, Niemann-pick C1-like 1 (NPC1L1) protein in intestinal and hepatic cholesterol transport, Annu. Rev. Physiol., 2021, 73, 239–259 CrossRef.
- L. J. Wang and B. L. Song, Niemann-Pick C1-Like 1 and cholesterol uptake, Biochim. Biophys. Acta, 2012, 1821(7), 964–972 CrossRef CAS PubMed.
- J. L. Betters and L. Yu, NPC1L1 and cholesterol transport, FEBS Lett., 2010, 584(13), 2740–2747 CrossRef CAS.
- H. Liu, L. Huang and X. Pei, Effects of sorghum rice and black rice on genes associated with cholesterol metabolism in hypercholesterolemic mice liver and intestine, Food Sci. Nutr.: Curr. Issues Answers, 2020, 9(1), 217–229 CrossRef.
- Y. T. Yeh, Y. Y. Cho, S. C. Hsieh and A. N. Chiang, Chinese olive extract ameliorates hepatic lipid accumulation in vitro and in vivo by regulating lipid metabolism, Sci. Rep., 2018, 8(1), 1057 CrossRef.
- C. Duval, V. Touche and A. Tailleux, et al., Niemann-Pick C1 like 1 gene expression is down-regulated by LXR activators in the intestine, Biochem. Biophys. Res. Commun., 2006, 340(4), 1259–1263 CrossRef CAS PubMed.
- S. N. Mathur, K. R. Watt and F. J. Field, Regulation of intestinal NPC1L1 expression by dietary fish oil and docosahexaenoic acid, J. Lipid Res., 2007, 48(2), 395–404 CrossRef CAS.
- L. J. Sharpe and A. J. Brown, Controlling cholesterol synthesis beyond 3-hydroxy-3-methylglutaryl-CoA reductase (HMGCR), J. Biol. Chem., 2013, 288(26), 18707–18715 CrossRef CAS PubMed.
- J. Li, X. Tang and J. Xu, et al., HMGCR gene polymorphism is associated with residual cholesterol risk in premature triple-vessel disease patients treated with moderate-intensity statins, BMC Cardiovasc. Disord., 2023, 23(1), 317 CrossRef CAS.
- B. A. Smith, C. Wright and M. Davidson, Role of Ezetimibe in Lipid-Lowering and Cardiovascular Disease Prevention, Curr. Atheroscler. Rep., 2015, 17(12), 72 CrossRef PubMed.
- X. X. Li, L. Zhao and Y. Chang, et al., Ezetimibe prevents myocardial remodeling in an obese rat model by inhibiting inflammation, Acta Biochim. Pol., 2018, 65(3), 465–470 CrossRef CAS PubMed.
- A. Ressaissi and M. L. M. Serralheiro, Hydroxycinnamic acid derivatives effect on hypercholesterolemia, comparison with ezetimibe: Permeability assays and FTIR spectroscopy on Caco-2 cell line, Curr. Res. Pharmacol. Drug Discov., 2022, 3, 100105 CrossRef PubMed.
- S. Miura and K. Saku, Ezetimibe, a selective inhibitor of the transport of cholesterol, Intern. Med., 2008, 47(13), 1165–1170 CrossRef PubMed.
- A. B. Dongmo, T. Miyamoto, K. Yoshikawa, S. Arihara and M. A. Lacaille-Dubois, Flavonoids from Acacia pennata and their cyclooxygenase (COX-1 and COX-2) inhibitory activities, Planta Med., 2007, 73(11), 1202–1207 CrossRef CAS.
- E. W. L. Chan, E. T. Y. Yeo and K. W. L. Wong, et al., Piper sarmentosum Roxb. Attenuates Beta Amyloid (Aβ)-Induced Neurotoxicity Via the Inhibition of Amyloidogenesis and Tau Hyperphosphorylation in SH-SY5Y Cells, Curr. Alzheimer Res., 2021, 18(1), 80–87 CrossRef CAS PubMed.
- M. N. Akmal, I. Abdel Aziz and M. F. Nur Azlina, Piper sarmentosum Roxb. methanolic extract prevents stress-induced gastric ulcer by modulating oxidative stress and inflammation, Front. Pharmacol., 2023, 13, 971443 CrossRef PubMed.
- J. Daimari and A. K. Deka, Anticancer, antimicrobial and antioxidant activity of CuO-ZnO bimetallic nanoparticles: green synthesised from Eryngium foetidum leaf extract, Sci. Rep., 2024, 14(1), 19506 CrossRef CAS.
- S. Manikandaselvi, R. S. Divya and R. Thinagarbabu, In vitro antidiabetic activity of aqueous fruit extract of Solanum torvum Sw, Int. Res. J. Pharm., 2018, 9(9), 145–151 CrossRef CAS.
- N. Satyanarayana, S. V. Chinni, R. Gobinath, P. Sunitha, A. Uma Sankar and B. S. Muthuvenkatachalam, Antidiabetic activity of Solanum torvum fruit extract in streptozotocin-induced diabetic rats, Front. Nutr., 2022, 9, 987552 CrossRef PubMed.
- N. Fangkrathok, J. Junlatat and B. Sripanidkulchai, In vivo and in vitro anti-inflammatory activity of Lentinus polychrous extract, J. Ethnopharmacol., 2013, 147(3), 631–637 CrossRef CAS.
- N. D. De Silva, A. P. Attanayake, D. N. Karunaratne, L. D. A. M. Arawwawala and G. K. S. Pamunuwa, Synthesis and bioactivity assessment of Coccinia grandis L. extract encapsulated alginate nanoparticles as an antidiabetic drug lead, J. Microencapsulation, 2023, 41(1), 1–17 CrossRef PubMed.
- S. Chaipoot, P. Wiriyacharee and R. Phongphisutthinant, et al., Changes in Physicochemical Characteristics and Antioxidant Activities of Dried Shiitake Mushroom in Dry-Moist-Heat Aging Process, Foods, 2023, 12(14), 2714 CrossRef CAS PubMed.
- A. Ontawong, A. Duangjai and C. Muanprasat, et al., Lipid-lowering effects of Coffea arabica pulp aqueous extract in Caco-2 cells and hypercholesterolemic rats, Phytomedicine, 2019, 52, 187–197 CrossRef CAS PubMed.
- M. van Heek, C. Farley, D. S. Compton, L. Hoos and H. R. Davis, Ezetimibe selectively inhibits intestinal cholesterol absorption in rodents in the presence and absence of exocrine pancreatic function, Br. J. Pharmacol., 2001, 134(2), 409–417 CrossRef CAS.
- O. Renner, D. Lütjohann and D. Richter, et al., Role of the ABCG8 19H risk allele in cholesterol absorption and gallstone disease, BMC Gastroenterol., 2013, 13, 30 CrossRef CAS PubMed.
- D. R. Michael, J. W. Moss, D. L. Calvente, I. Garaiova, S. F. Plummer and D. P. Ramji, Lactobacillus plantarum CUL66 can impact cholesterol homeostasis in Caco-2 enterocytes, Benefic. Microbes, 2016, 7(3), 443–451 CAS.
- R. Farahzadi, E. Fathi, S. A. Mesbah-Namin and N. Zarghami, Zinc sulfate contributes to promote telomere length extension via increasing telomerase gene expression, telomerase activity and change in the TERT gene promoter CpG island methylation status of human adipose-derived mesenchymal stem cells, PLoS One, 2017, 12(11), e0188052 CrossRef PubMed.
- M. Pierdomenico, A. F. G. Cicero and M. Veronesi, et al., Effect of Citrus bergamia extract on lipid profile: A combined in vitro and human study, Phytother. Res., 2023, 37(9), 4185–4195 CrossRef CAS PubMed.
- J. S. Joshi, H. H. Vora, N. R. Ghosh, R. N. Tankshali, D. H. Jetly and T. I. Trivedi, Nonhomologous end joining repair pathway molecules as predictive biomarkers for patients with oral squamous cell carcinoma, J. Cancer Res. Ther., 2021, 17(4), 1031–1038 CrossRef CAS PubMed.
- S. Wang, G. Xiaoling, L. Pingting, L. Shuqiang and Z. Yuaner, Chronic unpredictable mild stress combined with a high-fat diets aggravates atherosclerosis in rats, Lipids Health Dis., 2014, 13, 77 CrossRef PubMed.
- A. Ghanbari-Niaki, S. Rahmati-Ahmadabad and N. Zare-Kookandeh, ABCG8 Gene Responses to 8 Weeks Treadmill Running With or Without Pistachia atlantica (Baneh) Extraction in Female Rats, Int. J. Endocrinol. Metab., 2012, 10(4), 604–610 CrossRef PubMed.
- A. Ontawong, O. Boonphang and T. Pasachan, et al., Hepatoprotective effect of coffee pulp aqueous extract combined with simvastatin against hepatic steatosis in high-fat diet-induced obese rats, J. Funct. Foods, 2019, 54, 568–577 CrossRef CAS.
- V. A. Cortes, D. Busso, A. Maiz, A. Arteaga, F. Nervi and A. Rigotti, Physiological and pathological implications of cholesterol, Front. Biosci., 2014, 19(3), 416–428 CrossRef CAS.
- J. Chao, H. Y. Cheng and M. L. Chang, et al., Gallic Acid Ameliorated Impaired Lipid Homeostasis in a Mouse Model of High-Fat Diet-and Streptozotocin-Induced NAFLD and Diabetes through Improvement of β-oxidation and Ketogenesis, Front. Pharmacol., 2021, 11, 606759 CrossRef PubMed.
- A. Duangjai, P. Pontip and S. Sumhem, et al., Phenolic acids from Coffea arabica L. suppress intestinal uptake of glucose and cholesterol, Biomed. Res., 2020, 31, 59–66 CAS.
- S. Ngamukote, K. Mäkynen, T. Thilawech and S. Adisakwattana, Cholesterol-lowering activity of the major polyphenols in grape seed, Molecules, 2011, 16(6), 5054–5061 CrossRef CAS PubMed.
- C. Meng, L. Zhou and L. Huang, et al., Chlorogenic acid regulates the expression of NPC1L1 and HMGCR through PXR and SREBP2 signaling pathways and their interactions with HSP90 to maintain cholesterol homeostasis, Phytomedicine, 2024, 123, 155271 CrossRef CAS PubMed.
- C. Kirana, P. F. Rogers, L. E. Bennett, M. Y. Abeywardena and G. S. Patten, Naturally derived micelles for rapid in vitro screening of potential cholesterol-lowering bioactives, J. Agric. Food Chem., 2005, 53(11), 4623–4627 CrossRef CAS.
- L. A. Woollett, Y. Wang and D. D. Buckley, et al., Micellar solubilisation of cholesterol is essential for absorption in humans, Gut, 2006, 55(2), 197–204 CrossRef CAS PubMed.
- M. Armand, P. Borel and B. Pasquier, et al., Physicochemical characteristics of emulsions during fat digestion in human stomach and duodenum, Am. J. Physiol., 1996, 271(1 Pt 1), G172–G183 CAS.
- D. Q. Wang, New concepts of mechanisms of intestinal cholesterol absorption, Ann. Hepatol., 2003, 2(3), 113–121 CrossRef.
- F. Yang, G. Chen, M. Ma, N. Qiu, L. Zhu and J. Li, Egg-Yolk Sphingomyelin and Phosphatidylcholine Attenuate Cholesterol Absorption in Caco-2 Cells, Lipids, 2018, 53(2), 217–233 CrossRef CAS.
- K. Trisat, M. Wong-on, P. Lapphanichayakool, W. Tiyaboonchai and N. Limpeanchob, Vegetable Juices and Fibers Reduce Lipid Digestion or Absorption by Inhibiting Pancreatic Lipase, Cholesterol Solubility and Bile Acid Binding, Int. J. Veg. Sci., 2016, 23(3), 260–269 CrossRef.
- J. Su, H. Wang and C. Ma, et al., Hypocholesterolaemic mechanism of bitter melon aqueous extracts via inhibition of pancreatic cholesterol esterase and reduction of cholesterol micellar solubility, Int. J. Food Sci. Nutr., 2016, 67(1), 20–28 CrossRef PubMed.
- M. Cornelia and M. Anggraini, Healthy drinks made from okra and green tea extract for reducing blood cholesterol, Food Res., 2020, 4, 468–473 Search PubMed.
- S. I. Koo and S. K. Noh, Green tea as inhibitor of the intestinal absorption of lipids: potential mechanism for its lipid-lowering effect, J. Nutr. Biochem., 2007, 18(3), 179–183 CrossRef CAS PubMed.
- G. V. Vahouny, Dietary fiber, lipid metabolism, and atherosclerosis, Fed. Proc., 1982, 41(11), 2801–2806 CAS.
- K. Eliza, Lipids and their roles in physiological processes, Am. J. Physiol., Biochem. Pharmacol., 2023, 13(11), 01–02 Search PubMed.
- N. A. Abumrad and N. O. Davidson, Role of the gut in lipid homeostasis, Physiol. Rev., 2012, 92(3), 1061–1085 CrossRef CAS.
- B. Kim, Y. Park, C. J. Wegner, B. W. Bolling and J. Lee, Polyphenol-rich black chokeberry (Aronia melanocarpa) extract regulates the expression of genes critical for intestinal cholesterol flux in Caco-2 cells, J. Nutr. Biochem., 2013, 24(9), 1564–1570 CrossRef CAS PubMed.
- H. S. Yoon, J. H. Ju and H. Kim, et al., Lactobacillus rhamnosus BFE 5264 and Lactobacillus plantarum NR74 Promote Cholesterol Excretion Through the Up-Regulation of ABCG5/8 in Caco-2 Cells, Probiotics Antimicrob. Proteins, 2011, 3(3–4), 194–203 CrossRef CAS PubMed.
- S. Y. Park, J. W. Choi and D. N. Oh, et al., Anti-Obesity Potential through Regulation of Carbohydrate Uptake and Gene Expression in Intestinal Epithelial Cells by the Probiotic Lactiplantibacillus plantarum MGEL20154 from Fermented Food, J. Microbiol. Biotechnol., 2023, 33(5), 621–633 CrossRef CAS PubMed.
- J. J. Repa, K. E. Berge, C. Pomajzl, J. A. Richardson, H. Hobbs and D. J. Mangelsdorf, Regulation of ATP-binding cassette sterol transporters ABCG5 and ABCG8 by the liver X receptors alpha and beta, J. Biol. Chem., 2002, 277(21), 18793–18800 CrossRef CAS PubMed.
- B. Wang and P. Tontonoz, Liver X receptors in lipid signalling and membrane homeostasis, Nat. Rev. Endocrinol., 2018, 14(8), 452–463 CrossRef CAS PubMed.
- E. Susanti and E. Susilowati, The Effect of Green Tea on the Expression of NPC1L1, ABCG5, and ABCG8 in the Intestine of High Fat Diets-induced Rats, Indones. Biomed. J, 2021, 13(2), 147–154 CrossRef.
Footnote |
| † Medical Student. |
| This journal is © The Royal Society of Chemistry 2026 |