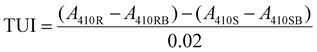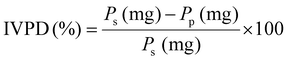Effects of dehulling and boiling on protein digestibility in faba beans with high and low trypsin inhibitor activity
Tianzhen
Xiao
 a,
Patrícia
Duque-Estrada
a,
Patrícia
Duque-Estrada
 b,
Mads Emil
Larsen
b,
Iben Lykke
Petersen
b,
Mads Emil
Larsen
b,
Iben Lykke
Petersen
 b,
Poul Erik
Jensen
b,
Lotte Bach
Larsen
a and
Nina Aagaard
Poulsen
*a
b,
Poul Erik
Jensen
b,
Lotte Bach
Larsen
a and
Nina Aagaard
Poulsen
*a
aDepartment of Food Science, Aarhus University, Agro Food Park 48, 8200 Aarhus N, Denmark. E-mail: nina.poulsen@food.au.dk
bDepartment of Food Science, University of Copenhagen, Rolighedsvej 26, 1958 Frederiksberg, Denmark
First published on 27th November 2025
Abstract
This study investigated how dehulling and boiling affect the in vitro protein digestibility (IVPD) of faba beans with either high (Fuego) or low (Granit) trypsin inhibitor activity (TIA), using the INFOGEST protocol. The results showed that dehulling and boiling affected the TIA and IVPD differently. In cultivar Fuego, dehulling significantly increased the IVPD by 15%, as well as a distinct decrease in TIA by 32%. However, in cultivar Granit, the effect of dehulling on TIA and protein digestibility had no significant effect. Boiling was found to decrease TIA in both cultivars distinctly (with a reduction of 93–96%) and significantly improved the IVPD in cultivar Fuego by 8%, but decreased in cultivar Granit by 13%. In both cultivars, protein hydrolysis during the intestinal phase was most significant, accounting for 58–84% of the total hydrolysis. The boiled faba beans displayed significantly higher levels of hydrolysis compared to raw faba beans during the intestinal phase. The findings suggest that the TIA in different cultivars of faba beans significantly differ and processing results in varied effects on their TIA and digestibility. Beyond the reduction of TIA, thermal treatments can induce comprehensive structural modifications and matrix interactions that affect faba bean protein digestibility differently. Generally, dehulling of faba beans also potentially enhances nutrient availability in faba beans.
1. Introduction
Faba bean (Vicia faba), along with pea (Pisum sativum), chickpea (Cicer arietinum) and lentil (Lens culinaris), belongs to cool-season grain legumes that can germinate at lower temperatures compared to soybeans (Glycine max).1 Among the cool-season legumes, faba beans are outstanding in terms of yield and protein-to-carbohydrate ratio.2 Protein and carbohydrate are the primary components in faba bean seeds, accounting for approximately 25% and 67% respectively of the dry weight, which can vary across the cultivars and growing conditions.3–5 However, as a legume, the protein quality of faba beans has some limits due to the deficiency of sulfur-containing amino acids (S-AAs) as well as a relatively low digestibility compared to animal proteins.6,7 The deficiency of S-AAs can be compensated by the supplementation of cereals in plant-based diets.8,9 The low digestibility, on the other hand, is more complex to resolve. Protein digestibility refers to the susceptibility of protein to be hydrolyzed by our digestive enzymes, which is affected by multiple factors, including food matrix, protein structure, antinutritional factors (ANFs), and processing methods.7 In faba bean seeds, the storage proteins, accounting for 80% of total proteins, are localized within the protein bodies surrounded by the starch granules inside individual cells, forming the microstructure of the cotyledon.2 For the digestive enzymes to access the plant protein substrates, the physical barrier of the plant cell walls must be destroyed, and the protein be released from its interactions with starch. Cell integrity also reduces the availability of unhydrolyzed macronutrients for fermentation by gut microbiota.10 It has been reported that the extent of protein digestion of legume seeds was distinctly limited in intact cells compared to cells being disrupted, e.g. by sonication and cooking.11,12Moreover, the presence of ANFs in faba beans, like trypsin inhibitors (TIs), lectins, phytates, and some polyphenols, can hinder protein digestibility in different ways. After a meal has been ingested, protein digestion begins in the gastric phase, where pepsin initiates the breakdown of proteins in acidic conditions in the stomach. Subsequently, the digest from the gastric phase is released into the intestinal phase, where proteins are continually digested by pancreatic proteases at higher pH. Trypsin is essential in protein digestion because of its activation of zymogens of other important digestive proteases, such as chymotrypsin and elastase, which are necessary for food protein hydrolysis into small peptides.8 However, antinutritional TIs are widespread in legumes, where they evolved as defense proteins against microorganisms and insects, but also hinder excessive proteolysis during seed development.13,14 There are two well-known proteinase inhibitor families among legumes, the Kunitz-type inhibitor (KTI) family, which is mostly found in soybeans and acts specifically against trypsin, and the Bowman-Birk inhibitor (BBI) family, which widely exists in legume seeds and suppresses activity of both trypsin and chymotrypsin independently at two different sites of action.15
In faba beans, the TI belongs to the BBI family, with a molecular weight around 8 kDa.16,17 The TIs have been shown to be enriched in the protein fractions produced by air classification.18 The concentrated TIs were also found in the soluble fraction after isoelectric precipitation, where the in vitro protein digestibility was found to be significantly decreased.19 To minimize the negative effects of BBI in faba beans, thermal treatment, such as boiling and microwaving, is widely used, resulting in enhanced digestibility of legume seed proteins.20 However, the improvement in plant protein digestibility is the combined result of thermal treatments, affecting both the physical structure and chemical composition of the food matrix. As shown by Avilés-Gaxiola et al., the digestibility stayed constant even though the trypsin inhibitor activity (TIA) was significantly decreased by thermal treatment.20 Besides TIs, phytates and polyphenols (mainly condensed tannins) in legume seeds can form complexes by binding to proteins, thereby reducing protein digestibility.21,22 The lectins, also called favins in faba beans, are proteins containing subunits composed of an α-chain (7 kDa) and a β-chain (20 kDa). Both chains contain binding sites for metal ions and specific oligosaccharides, suppressing the protein digestibility and even damaging the intestinal epithelium.7,23
The consumption of faba beans usually occurs after pre-treatments, like soaking and dehulling (removing the seed coat from the grain), which have been commonly applied to enhance the nutrient bioavailability and utilization of seeds before further processing, such as different cooking methods, like boiling and autoclaving. Soaking is widely used because it facilitates shorter cooking times. Soaking has been shown to improve starch digestibility, although the water-soluble nutrients, such as small sugars, minerals, and polyphenols, tend to leach out into the soaking water.24,25 Soaking is normally followed by cooking to make the faba beans edible by mitigating the ANFs distinctly, but processing may also include autoclaving, fermentation and germination, which will also affect ANFs and the nutritional and sensory quality of faba beans.26,27 Both cooking and autoclaving can, via the heat applied during the process, promote protein denaturation and aggregation, thereby potentially affecting protein digestibility.28,29 Duque-Estrada et al. showed that extrusion increased the in vitro digestibility of plant protein blends by approximately 18% by eliminating TIA.30 Fermentation and germination on the other hand may aid in enhancing protein digestibility of faba beans, although the mechanisms are different. Coda et al. found an overall enhancement of protein digestibility after applying Lactobacillus plantarum to faba bean flour.31
Compared to peas, the utilization of faba beans as human food is still relatively low, and the in vitro protein digestibility of faba beans affected by different factors remains largely uninvestigated. In this study, two cultivars of faba beans, which differed in their natural contents of TIA, were investigated to study the effects of TIA on the subsequent protein digestibility. Processes of dehulling and boiling were further applied to the two faba bean cultivars to explore their effects on TIA, and other factors affecting protein digestibility.
2. Materials and methods
2.1 Materials
Two cultivars of faba beans, Fuego and Granit, with the most distinct levels of TIA (Fig. S1) were selected for this study. The pulses of the two cultivars were collected from Danish farmers and processed by dehulling, soaking, and boiling (Fig. 1). For dehulling, the seed coat of faba beans was removed using an ultra-centrifugal mill (ZM 200, Retch, Germany) without a sieve mesh, and the seeds were manually separated from the seed coat. For soaking, faba beans were soaked in MilliQ water (m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v = 1
v = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5) at RT for 16 h. For boiling, the soaked faba beans were patted dry and boiled in MilliQ water (m
5) at RT for 16 h. For boiling, the soaked faba beans were patted dry and boiled in MilliQ water (m![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v = 1
v = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) at 165 °C for 35–45 min with a cover. The same soaking and boiling procedures were conducted on the whole faba beans (WF) and dehulled faba beans (DF), symbolized by soaked whole faba beans (SWF), boiled whole faba beans (BWF), soaked dehulled faba beans (SDF), and boiled dehulled faba beans (BDF). These processed faba bean samples, including dehulled, soaked and boiled faba beans, were freeze-dried for 2 days using a freeze-drier (Lyovapor L-200 Pro, Essen, Germany) and milled into fine powders (0.5 mm) using the same miller as for dehulling, with a 0.5 mm sieve mesh at room temperature (RT). The fine faba bean flours were transferred to 50 mL tubes and stored in a desiccator for further analysis. All chemicals and enzymes used in the present study were purchased from Sigma-Aldrich. Ultrapure water produced by the Milli-Q water system (Millipore Corporation, Merck, Germany) was used to prepare all the solutions.
3) at 165 °C for 35–45 min with a cover. The same soaking and boiling procedures were conducted on the whole faba beans (WF) and dehulled faba beans (DF), symbolized by soaked whole faba beans (SWF), boiled whole faba beans (BWF), soaked dehulled faba beans (SDF), and boiled dehulled faba beans (BDF). These processed faba bean samples, including dehulled, soaked and boiled faba beans, were freeze-dried for 2 days using a freeze-drier (Lyovapor L-200 Pro, Essen, Germany) and milled into fine powders (0.5 mm) using the same miller as for dehulling, with a 0.5 mm sieve mesh at room temperature (RT). The fine faba bean flours were transferred to 50 mL tubes and stored in a desiccator for further analysis. All chemicals and enzymes used in the present study were purchased from Sigma-Aldrich. Ultrapure water produced by the Milli-Q water system (Millipore Corporation, Merck, Germany) was used to prepare all the solutions.
2.2 Protein content measurement
The protein content of all faba beans was determined according to the AACC combustion method 46-30.1 (AACC, 2025) using the Dumatherm N Pro protein analysis system (Gerhardt Gmbh & Co., Germany). The nitrogen to protein conversion factor used was 5.4.2.3 Trypsin inhibitor activity assay
The trypsin inhibitors were extracted according to Liu with small modifications.32 In brief, 1 g of faba bean flour was mixed with 50 ml 10 M NaOH and stirred at 720 rpm for 3 h. The supernatant was collected and stored at −20 °C until analysis. The measurement of TIA followed the American Oil Chemists Society (AOAC) method Ba 12a-2020, which has been tested by Liu.32 Briefly, the trypsin inhibitor extracts were diluted with MilliQ water to reach 30–70% trypsin inhibition. The substrate Nα-benzoyl-DL-arginine-p-nitroanilide (BAPNA) hydrochloride solution (0.4 mg mL−1) in 50 mM Tris buffer containing 20 mM CaCl2 (pH 8.2) and 20 μg mL−1 trypsin solution in 1 mM HCl containing 5 mM CaCl2 was prepared for analysis. All the reactions were conducted in a water bath at 37 °C and followed the sequence of sample, substrate, and enzyme. The reaction was stopped by 30% acetic acid exactly 10 min after adding the enzymes. TIA is expressed as trypsin units inhibited (TUI), where one trypsin unit is defined as an increase of 0.02 absorbance at 410 nm under the 5 mL assay condition, and can be calculated by the following equation:where A410R = reference reading, A410RB = reference blank, A410S = sample reading, A410SB = sample blank. The TIA was further calculated in TIU per mg of faba bean flour on a dry basis.
2.4 In vitro digestion
The raw and boiled faba bean samples were subjected to in vitro digestion according to the INFOGEST static protocol.33 The simulated salivary fluids (SSF) simulated gastric fluids (SGF), and simulated intestinal fluids (SIF) for different digestion phases were prepared according to the protocol. Before digestion, the enzyme activity of pepsin from pig gastric mucosa (P7012, Sigma Aldrich), pancreatin from porcine pancreas (8× USP, P7545, Sigma Aldrich), and the bile content of bovine bile extract (B3883, Sigma Aldrich) were measured. The activity of pepsin and pancreatin was 2817 U mL−1, 6.4 U mL−1, respectively. The pancreatin was dissolved according to Sousa et al.34 Briefly, the weighed pancreatin was mixed with SIF and vortex for 10 s before ultrasound solubilization for 5 min in an ice-water bath. The mixture was centrifuged at 3000g for 5 min at 4 °C and the supernatant was collected and kept on ice before use. The concentration of bile was 1.03 mM g−1. The pH test at the gastric and intestinal phases was conducted before analysis to record the 1 M HCl and 2 M NaOH needed for pH adjustment. For the digestion, samples containing 40 mg of protein were solubilized in MilliQ water and mixed with SSF and CaCl2 solution. The mixture was incubated at 37 °C for 2 min to mimic the oral phase. Then, SGF was added, and the pH was adjusted to 3 with 1 M HCl before adding pepsin to reach 2000 U mL−1 and incubating at 37 °C for 2 h to mimic the gastric phase. Subsequently, the SIF and CaCl2 solution were added, and the pH was adjusted to 7 using 2 M NaOH before adding bile to reach 50 U mL−1 and pancreatin to reach 100 U mL−1. After another 2 h of incubation at 37 °C, the samples were transferred to ice and mixed with ice-cold methanol to a final concentration of 80% to stop the reaction. The solution was incubated at −18 °C for 1 h to precipitate undigested proteins before centrifugation at 4 °C, 4000g for 15 min. To collect samples from the oral and gastric phases, independent experiments were conducted according to the protocol, respectively. Ice-cold methanol was added to the digests after each digestion, reaching the final concentration of 80%, to stop the reaction and precipitate undigested proteins. The supernatant and pellet were collected, respectively. The supernatant was aliquoted and stored at −18 °C for further analysis. The pellet was freeze-dried and weighed before storing at −18 °C for further analysis. All digestion was conducted in triplicate.2.5 Determination of free amino groups and in vitro protein digestibility (IVPD) after digestion
The O-phthaldialdehyde assay (OPA) was conducted according to Michels & Frister with some modifications.35 Briefly, 1 mL of the supernatant from the in vitro digestion was dried by speed vacuum and re-suspended in 100 µL 0.1 M borate solution. Samples from the gastric and intestinal phases were diluted accordingly before analysis. After dilution, 8 µL samples were mixed with 232 µL OPA solution that contains 0.05 M Borate solution, 1% SDS, 0.8 g L−1 OPA in ethanol, 4 g L−1 Na-MES solution, 5 g L−1 Tergitol™ 15-S-9 solution in a glass microplate. The 0.1 M Borate solution was used as a blank, and L-glutamic acid solution (0.25–8 mM L−1) was used as the standard. The plate was incubated for 10 min at 30 °C in the dark and the absorbance at 340 nm was measured by the plate reader (BioTek Gen5, Agilent, CA, US). The results were calculated and expressed as glutamic acid equivalents (Glu-E). All samples were analyzed in technical triplicate. The IVPD was detected according to Ayala-Rodríguez et al.36 Briefly, the protein content in the pellet was measured by determining the nitrogen content as described in section 2.2, and calculated by:where Ps is the protein content before digestion, and Pp is the protein content in the pellet after intestinal digestion.
2.6 Total polyphenolic content (TPC) measurement
The TPC was determined using the Folin–Ciocalteu (F–C) assay37 with slight modifications. Briefly, 200 mg of raw faba bean flour was mixed with 1.5 mL of 70% (v/v) ethanol. The mixture was vortexed for 1 min and shaken at 2000 rpm for 60 min, followed by centrifugation at 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 817g, RT, for 10 min. The supernatant was collected, and the pellet was extracted with 250 μL 70% (v/v) ethanol for another 30 min. The mixture was centrifuged to collect the supernatant. This extraction was repeated once more, and all the supernatants were combined and stored at −20 °C until further analysis. Before analysis, the extracts were centrifuged at 5000g for 5 min. Gallic acid dissolved in MilliQ water at concentrations from 0.01 to 0.5 mg mL−1 was used as the standard. There were 15 μL of samples added to the 96-well plates, followed by 235 μL of MilliQ water, 20 μL of 1 N F–C reagent, and 30 μL of 0.5 M Na2CO3 solution. The plate was incubated for 2 h in the dark at RT. The absorbance was measured at 765 nm using the plate reader with the Gen5 1.11 software (BioTek Gen5, Agilent, CA, US). All extractions were conducted in biological triplicate and further measured in technical triplicate.
817g, RT, for 10 min. The supernatant was collected, and the pellet was extracted with 250 μL 70% (v/v) ethanol for another 30 min. The mixture was centrifuged to collect the supernatant. This extraction was repeated once more, and all the supernatants were combined and stored at −20 °C until further analysis. Before analysis, the extracts were centrifuged at 5000g for 5 min. Gallic acid dissolved in MilliQ water at concentrations from 0.01 to 0.5 mg mL−1 was used as the standard. There were 15 μL of samples added to the 96-well plates, followed by 235 μL of MilliQ water, 20 μL of 1 N F–C reagent, and 30 μL of 0.5 M Na2CO3 solution. The plate was incubated for 2 h in the dark at RT. The absorbance was measured at 765 nm using the plate reader with the Gen5 1.11 software (BioTek Gen5, Agilent, CA, US). All extractions were conducted in biological triplicate and further measured in technical triplicate.
2.7 Scanning electron microscopy (SEM)
The dry and fine faba bean powders were further dried in a desiccator overnight. Minute amounts of dehydrated samples were powdered onto carbon conductive double-sided tape on SEM aluminum stubs with a toothpick. Samples were sputter-coated with gold in a Leica EM ACE200 Vacuum Coater (Leica, Wetzlar, Germany) to a thickness of 8 nm. Scanning electron microscopy images were captured using a FEI Quanta 200 SEM (FEI Company, Hillsboro, Oregon, United States) operating at 10 kV acceleration voltage, using ×T Microscope Control software.2.8 Statistical analysis
Two-way analysis of variance (ANOVA) with fixed factors, cultivar and processes, was used to measure the significance among different faba bean samples by Minitab (version 19.1). One-way ANOVA with Tukey's range test was used to measure the significance among different processes within cultivars. The level of statistical significance was determined at p < 0.05.3. Results
3.1 TIA of faba beans before and after processing
To evaluate the effects of dehulling, soaking, and boiling on TIA, the TIA in two cultivars of faba beans was measured before and after processing, as shown in Table 1. The difference between two factors, cultivar and process, was analyzed by two-way ANOVA analysis and shown in Table 2. The TIA in two cultivars showed a significant difference (p value <0.000), with cultivar Fuego displaying significantly higher TIA than cultivar Granit. The effects of processing (soaking, dehulling, and boiling) on TIA were also significant (p value <0.000). Soaking and boiling showed a significant decrease of TIA, compared to the whole faba beans. In cultivar Fuego, soaking, dehulling, and boiling significantly decreased the TIA by 50%, 32%, and 96%, respectively. Boiling significantly reduced TIA in both whole and dehulled faba beans across both cultivars. In contrast, soaking had no significant effect on TIA in the dehulled beans of either cultivar. The effect of soaking was excluded from the in vitro digestion experiment, since there was no difference in TIA compared to the raw faba beans.| Cultivar | Granit | Fuego | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Process | WF | SWF | BWF | DF | SDF | BDF | WF | SWF | BWF | DF | SDF | BDF |
| WF: whole faba beans; SWF: soaked whole faba beans; BWF: boiled whole faba beans after soaking; DF: dehulled faba beans; SDF: soaked dehulled faba beans; BDF: boiled dehulled faba beans. *The decreased percentage of TIA and TPC, and increased percentage of IVPD% of processed faba beans compared to the WF; different letters show a significant difference (p < 0.05). #Increased percentage of IVPD% of processed faba beans compared to the dehulled faba beans. | ||||||||||||
| TIA (U mg−1 sample) | 3.43 ± 0.31a | 3.47 ± 0.11b | 0.23 ± 0.08c | 3.87 ± 0.09ab | 3.93 ± 0.22ab | 0.43 ± 0.15c | 11.84 ± 0.96a | 5.89 ± 0.53b | 0.45 ± 0.13c | 8.09 ± 0.30ab | 7.69 ± 0.17ab | 0.35 ± 0.18c |
| Reduction* | −9% | 93% | −13% | −15% | 87% | 50% | 96% | 32% | 35% | 97% | ||
| IVPD % | 85.67 ± 4.54b | — | 74.11 ± 5.02b | 91.30 ± 4.32a | — | 85.18 ± 5.95ab | 81.88 ± 6.04b | — | 88.80 ± 1.68b | 93.81 ± 1.93a | — | 87.23 ± 0.21ab |
| Increase* | −13% | 7% | −1%, −7%# | 8% | 15% | 7%, −7%# | ||||||
| TPC (mg GAE per g sample) | 1.17 ± 0.01a | 1.14 ± 0.02ab | 0.91 ± 0.03c | 0.99 ± 0.06bc | 0.88 ± 0.06c | 0.56 ± 0.02d | 2.19 ± 0.13a | 2.13 ± 0.04ab | 1.61 ± 0.03c | 1.82 ± 0.09bc | 1.67 ± 0.08c | 0.90 ± 0.09d |
| Reduction | 3% | 22% | 15% | 25% | 52% | 3% | 26% | 17% | 24% | 59% | ||
| TIA (U mg−1 sample) | IVPD % | TPC (mg GAE per g sample) | Free α-amino groups –oral phase | Free α-amino groups –gastric phase | Free α-amino groups –intestinal phase | |
|---|---|---|---|---|---|---|
| Cultivar | 0.000 | 0.059 | 0.000 | 0.153 | 0.358 | 0.448 |
| Process | 0.000 | 0.004 | 0.000 | 0.000 | 0.015 | 0.000 |
3.2 In vitro digestibility of processed faba beans
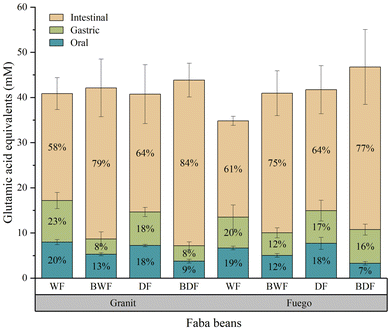
For both cultivars, the highest proportion of free α-amino groups was released during the intestinal phase, independently of the process. On average, there were 5.86, 7.48 and 28.61 mM Glu-E released during oral, gastric, and intestinal digestion, respectively, across all samples. In the oral and gastric phase, the BWF showed significantly lower levels of hydrolysis than raw faba beans in both cultivars. In the gastric phase, the raw Granit faba beans showed the highest hydrolysis, significantly higher than that detected in the boiled faba beans. However, a larger amount of free α-amino groups was released during intestinal digestion in the boiled faba beans, compared to the raw faba beans in both cultivars. During intestinal digestion, the boiled faba beans displayed a higher level of hydrolysis than the raw faba beans with statistical significance. Both cultivars exhibited increased protein hydrolysis after dehulling and boiling processes in the intestinal digestion, with the BDF of Fuego showing the highest concentration of free α-amino groups (52 mM) compared to the others. For both cultivars, DF displayed a lower proportion of free α-amino groups than BWF after intestinal digestion without statistical significance.
3.3 Total polyphenol content (TPC) of different faba beans
The TPC in the analyzed faba beans was measured and is shown in Table 1. The effects of cultivar and process on TPC were of significance as shown in Table 2. The Fuego raw faba beans exhibited significantly higher TPC than Granit (2.19 mg GAE per g versus 1.17 mg GAE per g). After dehulling, the TPC was significantly reduced to 1.82 mg GAE per g in Fuego with no significant decrease observed in the DF of Granit. Boiling significantly decreased TPC in both whole and dehulled faba beans from the two cultivars. The TPC displayed an opposite pattern from the released free α-amino groups during the intestinal phase across the faba beans.3.4 Microstructure of processed faba beans
The effects of soaking, dehulling, and boiling on the microstructure of the cotyledons, primarily composed of starch granules and protein bodies, were visualized by SEM (Fig. 3). The starch granules were embedded with protein bodies and further attached to components such as non-polysaccharide starch and cell walls. As shown in Fig. 3a, the largest particles were observed in the whole faba beans, where the starch granules and protein bodies were less exposed. After soaking, less compact microstructure of starch granules, protein bodies and other components were observed due to the water absorption of starch (Fig. 3b). More starch granules were observed in the soaked faba bean flours than in raw faba beans. After dehulling, the observed particles became smaller, whereas the size of starch granules and protein bodies remained the same (Fig. 3c). Both intact starch granules attached to protein bodies and complicated particles were observed after boiling, where the surface of the starch granule was less smooth with loose structures (Fig. 3d). | ||
| Fig. 3 Scanning electron microscope images of Granit faba beans. (a) Whole faba beans; (b) Soaked faba beans; (c) Dehulled faba beans, (d) Boiled whole faba beans. | ||
4. Discussion
4.1 Effects of dehulling and boiling on trypsin inhibitor activity (TIA)
In this study, two faba bean cultivars with different TIA were analyzed. The TIA ranged from 2.5 (cultivar Granit) to 11.6 TUI per mg (Fuego), which is in the same range as reported in other studies.4,38 Vidal-Valverde et al. reported that dietary TIs reduced animal growth and triggered pancreatic hypertrophy and hyperplasia due to decreased trypsin activity.39 The presence of TIs can also hinder the absorption of free amino acids from the diet and cause an enlargement of the pancreas in rats.40 In a previous study, the relative abundance of the BBI was quantified across different cultivars, with Granit and Fuego showing 4% and 6% of total proteins, respectively.41 The level of TIA in faba beans is affected by different processing methods, among which thermal treatments, like boiling or roasting, can reduce the BBI effectively in faba beans.42,43 Compared to boiling, soaking and dehulling seem less efficient in reducing TIA of faba beans,44 which is consistent with our results (Table 1). In the current study, the free α-amino groups released during intestinal digestion were significantly higher in BDF with a significant reduction of TIA compared to WF in both cultivars. Accordingly, an increase in vitro protein digestibility during intestinal digestion was observed in thermally treated samples, where TIA was decreased.42 Alonso et al. reported that IVPD increased by 23.5% after extrusion of faba beans, with a decrease of 98.9% of TIA compared to the raw samples.45 Our results showed that the boiled dehulled faba beans showed significantly higher hydrolysis in the intestinal digestion compared to raw, dehulled samples. The significant effect of boiling was also observed in raw samples of Granit, but not in Fuego. The higher digestibility induced by thermal treatment is, apart from the effect on TIA, also associated with modifications to the protein structure. It is worth stating the limitations of the in vitro static digestion protocol applied, which may underestimate the level of free amino groups after digestion. According to the INFOGEST protocol, the enzyme activity in the intestinal phase should be 100 U mL−1.33 While in practice, the commercial pancreatin activity is far lower than this requirement, leading to a large amount of inactive pancreatin, including trypsin and chymotrypsin related to protein digestion, added to the digestion system. The active TIs in the faba bean samples may potentially bind to inactive pancreatin and subsequently result in an underestimation of the effect of TIA.During digestion, food is physically broken down in the oral phase, releasing the nutrients before entering the gastric phase, where the chemical breakdown occurs, the acid starts to denature the released starch and proteins of plant foods, enhancing their susceptibility to pepsin degradation by exposing peptide bonds.46,47 Pepsin mainly hydrolyzes proteins into large peptides or oligopeptides and thus primes further breakdown of protein in the intestine.48 The degree of protein hydrolysis was less in the gastric compared to the intestinal stage, which was as expected and consistent with the results of other studies.49,50
4.2 Protein digestibility affected by protein composition
In this study, the IVPD of raw faba beans was above 80%, which was comparable to previous research.21 Faba bean protein digestibility can be up to 94% after autoclaving, compared to only 31% in dry-processed beans treated at 70 °C.36 In the current study, the highest level of IVPD (94%) was detected in the dehulled Fuego faba beans. For cultivar Granit, the IVPD of boiled faba beans was decreased, which might result from multiple factors, including the protein composition, other components that can interact with proteins, like TIs, polyphenols, and effects related to the food matrix. Martineau-Côté et al. also found that boiling had a negative effect on faba bean protein digestibility, indicating the importance of studying the impact of food processing on protein quality.51 The storage protein in faba bean seeds is predominantly globulins, which are composed of 11S legumins and 7S vicilins and convicilins.41 Legumins are hexamers with a molecular weight (MW) of 360–400 kDa, consisting of disulfide-linked polypeptides.52 Vicilin and convicilin are trimers with a lower MW around 220–290 kDa, which are assembled by sulfur-free subunits.53 Nielsen et al. reported that vicilin was more susceptible to digestion compared to other legume storage proteins.54 Contardo et al. reported that vicilin showed 99% protein hydrolysis during the intestinal stage, which was significantly higher than protein hydrolysis of legumin (84%) in chickpeas, which may be attributed to the lower MW of vicilin and the higher exposure of polar groups in its structure.50 Legumin has higher heat coagulation at neutral pH compared to vicilin, leading to a lower digestibility.55 The legumin to vicilin ratio was determined in our previous study, where the L/V value of Granit and Fuego was 1.03 and 0.22, respectively.41 This can explain the lower content of free amino groups determined in the boiled samples of cultivar Granit during the gastric phase compared to Fuego, and the lower protein digestibility in the boiled faba beans due to higher levels of legumins.4.3 Polyphenols
Polyphenols are widely distributed in plant foods, showing antioxidant ability. Recently, polyphenols in faba beans have been increasingly studied due to their health-promoting properties. Abu-Reidah et al. characterized 104 polyphenols in faba beans, dominated by flavonoids.56 Valente et al. found the TPC ranged from 2.62 to 4.3 mg GAE per g dry weight in faba beans across different cultivars, which was positively correlated with the antioxidant activity.57 The condensed tannins are the predominant polyphenols in the seed coat of legume seeds, which tend to diffuse to the cotyledon more easily, compared to the monomeric polyphenols during boiling.58 Alonso & Marzo showed that dehulling significantly decreased the content of condensed tannins and total polyphenols,45 which was also observed in the current study (Table 1). When entrapped within the food matrix, polyphenols can form complexes with lipids, carbohydrates, and proteins, among which polyphenol-protein interactions can induce astringency in the food and affect the enzyme activity and protein digestibility.59 The interaction between protein and polyphenols is largely affected by the type of phenols, including their molecular weight, number of hydroxyl groups, and protein structure, at different temperatures and pH values.59,60 At neutral pH, proteins and polyphenols tend to bind by non-covalent bonds, affected by the secondary structure of proteins. The interaction of protein and polyphenols reduces the α-helix content, but increases the random coil content, leading to a loose secondary structure of proteins. More hydrophobic groups and enhanced polarity are observed in the tertiary structure.61 The bioaccessibility of protein and polyphenols in their interaction depends on the type of bonding. Proteins are more bioaccessible when bound non-covalently, whereas polyphenols exhibit higher availability in the intestine when covalently bound due to the escape of gastric digestion.624.4 Microstructure
The effect of different processing steps on the microstructure of chickpea was studied by Aguilera et al., showing similar results as those found in this study and demonstrating increased starch availability after cooking.63 The seed coat of faba beans is generally thicker than that of pea, chickpeas and lentils,64,65 which may hinder the physical breakdown of the cotyledons, reducing surface area available for enzymatic digestion. The current study also found smaller particles after dehulling, indicating more extensive damage to the cotyledons compared to the whole beans. It was reported that dehulling resulted in smaller particle sizes in faba beans than in whole seeds.66 Generally, the cotyledon cells are broken by thermal treatment, generating increased available starch and proteins during digestion.49,675. Conclusion
In this study, the effects of dehulling and boiling on the in vitro protein digestibility of faba beans with high (Fuego) and low TIA (Granit) were investigated by applying the INFOGEST protocol. The effects of dehulling and soaking on TIA were different between the two cultivars. In the low TIA cultivar Granit, the impact of dehulling on TIA was of no significance, leading to no significant difference observed in the IVPD between the whole and dehulled faba beans. In the high TIA cultivar Fuego, dehulling significantly decreased TIA and distinctly enhanced the IVPD up to 94%, which makes faba beans a promising source of plant protein. Boiling of the beans significantly decreased TIA in both cultivars to below 0.45 TUI per mg, which further mitigates the negative effects of TIs on faba bean protein digestibility. However, the IVPD result of boiled faba beans showed a different trend compared to the raw faba beans. In cultivar Fuego, the IVPD of boiled whole faba beans was significantly increased, while the IVPD was significantly decreased in the boiled dehulled faba beans. In cultivar Granit, the IVPD of whole faba beans was also significantly decreased by boiling, implying that factors beyond TIA affect faba bean protein digestibility. The TPC was significantly lower in Granit than in Fuego. Dehulling and boiling significantly decreased TPC in both cultivars. However, the interactions between protein and polyphenol, which may better explain their effects on protein digestibility, were not included in the current study. The smaller starch granules attached to protein bodies were observed after dehulling, and boiled samples displayed an increased surface area of starch granules and protein bodies. Overall, dehulling showed improvement in the IVPD of faba bean, which may result from the removal of physical barriers that limit nutrient bioavailability. Moreover, these findings indicate that beyond the reduction of TIA, thermal treatments can induce comprehensive structural modifications and matrix interactions that affect faba bean protein digestibility, highlighting the need to understand and eventually elucidate the underlying mechanisms by which heat processing modulates protein digestibility.Conflicts of interest
The authors declare no competing interest.Data availability
Data will be made available from the corresponding author on reasonable request.Supplementary information (SI) is available. Trypsin inhibitor activity (TIA) of four faba bean cultivars. See DOI: https://doi.org/10.1039/d5fo03653d.
Acknowledgements
The authors thank Elena Bergamasco and Yueyue Liu from the University of Copenhagen for testing the method for trypsin inhibitor activity measurement. We express our gratitude to the suppliers of faba beans for providing the materials for this study. This study was part of the project ØkoFaba financed by the Danish Organic Farming Levy Foundation. The work was also supported by the China Scholarship Council (CSC).References
- P. Williams, R. Bhatty, S. Deshpande, L. Hussein and G. Savage, Improving nutritional quality of cool season food legumes, in Expanding the Production and Use of Cool Season Food Legumes: A global perspective of peristent constraints and of opportunities and strategies for further increasing the productivity and use of pea, lentil, faba bean, chickpea and grasspea in different farming systems, Springer, 1994, pp. 113–129 Search PubMed.
- S. Sharan, G. Zanghelini, J. Zotzel, D. Bonerz, J. Aschoff, A. Saint-Eve and M. N. Maillard, Fava bean (Vicia faba L.) for food applications: From seed to ingredient processing and its effect on functional properties, antinutritional factors, flavor, and color, Compr. Rev. Food Sci. Food Saf., 2021, 20(1), 401–428 CrossRef CAS PubMed.
- G. Duc, P. Marget, R. Esnault, J. Le Guen and D. Bastianelli, Genetic variability for feeding value of faba bean seeds (Vicia faba): Comparative chemical composition of isogenics involving zero-tannin and zero-vicine genes, J. Agric. Sci., 1999, 133(2), 185–196 CrossRef CAS.
- I. C. M. Labba, H. Frokiær and A. S. Sandberg, Nutritional and antinutritional composition of fava bean (Vicia faba L., var. minor) cultivars, Food Res. Int., 2021, 140, 110038 CrossRef PubMed.
- P. J. Pritchard, E. A. Dryburgh and B. J. Wilson, Carbohydrates of spring and winter field beans (Vicia faba L.), J. Sci. Food Agric., 1973, 24(6), 663–668 CrossRef CAS PubMed.
- M. A. Kniskern and C. S. Johnston, Protein dietary reference intakes may be inadequate for vegetarians if low amounts of animal protein are consumed, Nutrition, 2011, 27(6), 727–730 CrossRef CAS PubMed.
- A. G. A. Sa, Y. M. F. Moreno and B. A. M. Carciofi, Food processing for the improvement of plant proteins digestibility, Crit. Rev. Food Sci. Nutr., 2020, 60(20), 3367–3386 CrossRef CAS PubMed.
- P. B. Geil and J. W. Anderson, Nutrition and health implications of dry beans: a review, J. Am. Coll. Nutr., 1994, 13(6), 549–558 CrossRef CAS PubMed.
- A. Iqbal, I. A. Khalil, N. Ateeq and M. S. Khan, Nutritional quality of important food legumes, Food Chem., 2006, 97(2), 331–335 CrossRef CAS.
- N. Guan, X. He, S. Wang, F. Liu, Q. Huang, X. Fu, T. Chen and B. Zhang, Cell Wall Integrity of Pulse Modulates the in Vitro Fecal Fermentation Rate and Microbiota Composition, J. Agric. Food Chem., 2020, 68(4), 1091–1100 CrossRef CAS PubMed.
- S. A. Junejo, L. Ding, X. Fu, W. Xiong, B. Zhang and Q. Huang, Pea cell wall integrity controls the starch and protein digestion properties in the INFOGEST in vitro simulation, Int. J. Biol. Macromol., 2021, 182, 1200–1207 CrossRef CAS PubMed.
- C. Melito and J. Tovar, Cell walls limit in vitro protein digestibility in processed legume seeds, Food Chem., 1995, 53(3), 305–307 CrossRef CAS.
- W. E. Brown, K. Takio, K. Titani and C. A. Ryan, Wound-induced trypsin inhibitor in alfalfa leaves: identity as a member of the Bowman-Birk inhibitor family, Biochem, 1985, 24(9), 2105–2108 CrossRef CAS PubMed.
- P. A. Divekar, V. Rani, S. Majumder, S. G. Karkute, K. A. Molla, K. K. Pandey, T. K. Behera and G.-P.-P. Govindharaj, Protease Inhibitors: An Induced Plant Defense Mechanism Against Herbivores, J. Plant Growth Regul., 2022, 42(10), 6057–6073 CrossRef.
- M. Laskowski Jr. and M. Qasim, What can the structures of enzyme-inhibitor complexes tell us about the structures of enzyme substrate complexes?, Biochim. Biophys. Acta, Protein Struct. Mol. Enzymol., 2000, 1477(1–2), 324–337 CrossRef PubMed.
- T. Asao, F. Imai, I. Tsuji, M. Tashiro, K. Iwami and F. Ibuki, The amino acid sequence of a Bowman-Birk type proteinase inhibitor from faba beans (Vicia faba L.), J. Biochem., 1991, 110(6), 951–955 CrossRef CAS PubMed.
- E. Guillamón, M. M. Pedrosa, C. Burbano, C. Cuadrado, M. de Cortes Sánchez and M. Muzquiz, The trypsin inhibitors present in seed of different grain legume species and cultivar, Food Chem., 2008, 107(1), 68–74 CrossRef.
- M. Vogelsang-O'Dwyer, I. L. Petersen, M. S. Joehnke, J. C. Sorensen, J. Bez, A. Detzel, M. Busch, M. Krueger, J. A. O'Mahony and E. K. Arendt, et al., Comparison of Faba Bean Protein Ingredients Produced Using Dry Fractionation and Isoelectric Precipitation: Techno-Functional, Nutritional and Environmental Performance, Foods, 2020, 9(3), 322 CrossRef PubMed.
- M. Krause, J. C. Sorensen, I. L. Petersen, P. Duque-Estrada, C. Cappello, A. Z. A. Tlais, R. Di Cagno, L. Ispiryan, A. W. Sahin and E. K. Arendt, et al., Associating Compositional, Nutritional and Techno-Functional Characteristics of Faba Bean (Vicia faba L.), Protein Isolates and Their Production Side-Streams with Potential Food Applications, Foods, 2023, 12(5), 919 CrossRef CAS PubMed.
- S. Aviles-Gaxiola, C. Chuck-Hernandez and S. O. Serna Saldívar, Inactivation, Methods of Trypsin Inhibitor in Legumes: A Review, J. Food Sci., 2018, 83(1), 17–29 CrossRef CAS PubMed.
- G. S. Gilani, K. A. Cockell and E. Sepehr, Effects of antinutritional factors on protein digestibility and amino acid availability in foods, J. AOAC Int., 2005, 88(3), 967–987 CrossRef CAS.
- A. Van der Poel, Effect of processing on antinutritional factors and protein nutritional value of dry beans (Phaseolus vulgaris L.). A review, Anim. Feed Sci. Technol., 1990, 29(3–4), 179–208 CrossRef CAS.
- G. N. Reeke Jr. and J. W. Becker, Three-dimensional structure of favin: saccharide binding-cyclic permutation in leguminous lectins, Science, 1986, 234(4780), 1108–1111 CrossRef CAS PubMed.
- A. Duhan, N. Khetarpaul and S. Bishnoi, Improvement in starch digestibility (in vitro) of various Pigeonpea cultivars through processing and cooking, Ecol. Food Nutr., 1998, 37(6), 557–568 CrossRef.
- J. Frias, M. Prodanov, I. Sierra and C. Vidal-Valverde, Effect of Light on Carbohydrates and Hydrosoluble Vitamins of Lentils during Soaking, J. Food Prot., 1995, 58(6), 692–695 CrossRef CAS PubMed.
- S. B. Dhull, M. K. Kidwai, R. Noor, P. Chawla and P. K. Rose, A review of nutritional profile and processing of faba bean (Vicia faba L.), Legume Sci., 2022, 4(3), e129 CrossRef CAS.
- I. C. Ohanenye, F. C. Ekezie, R. A. Sarteshnizi, R. T. Boachie, C. U. Emenike, X. Sun, I. D. Nwachukwu and C. C. Udenigwe, Legume Seed Protein Digestibility as Influenced by Traditional and Emerging Physical Processing Technologies, Foods, 2022, 11(15), 2299 CrossRef CAS PubMed.
- R. A. Habiba, Changes in anti-nutrients, protein solubility, digestibility, and HCl-extractability of ash and phosphorus in vegetable peas as affected by cooking methods, Food Chem., 2002, 77(2), 187–192 CrossRef CAS.
- Z. Ma, J. I. Boye, B. K. Simpson, S. O. Prasher, D. Monpetit and L. Malcolmson, Thermal processing effects on the functional properties and microstructure of lentil, chickpea, and pea flours, Food Res. Int., 2011, 44(8), 2534–2544 CrossRef CAS.
- P. Duque-Estrada, K. Hardiman, A. Bogebjerg Dam, N. Dodge, M. D. Aaslyng and I. L. Petersen, Protein blends and extrusion processing to improve the nutritional quality of plant proteins, Food Funct., 2023, 14(16), 7361–7374 RSC.
- R. Coda, L. Melama, C. G. Rizzello, J. A. Curiel, J. Sibakov, U. Holopainen, M. Pulkkinen and N. Sozer, Effect of air classification and fermentation by Lactobacillus plantarum VTT E-133328 on faba bean (Vicia faba L.) flour nutritional properties, Int. J. Food Microbiol., 2015, 193, 34–42 CrossRef CAS PubMed.
- K. S. Liu, Comparison of ISO 14902
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2001 with AOCS Ba 12a-2020 for determining trypsin inhibitor activity in protein products, J. Am. Oil Chem. Soc., 2021, 98(12), 1115–1129 CrossRef CAS.
2001 with AOCS Ba 12a-2020 for determining trypsin inhibitor activity in protein products, J. Am. Oil Chem. Soc., 2021, 98(12), 1115–1129 CrossRef CAS. - A. Brodkorb, L. Egger, M. Alminger, P. Alvito, R. Assuncao, S. Ballance, T. Bohn, C. Bourlieu-Lacanal, R. Boutrou and F. Carriere, et al. INFOGEST static in vitro simulation of gastrointestinal food digestion, Nat. Protoc., 2019, 14(4), 991–1014 CrossRef CAS PubMed.
- R. Sousa, I. Recio, D. Heimo, S. Dubois, P. J. Moughan, S. M. Hodgkinson, R. Portmann and L. Egger, In vitro digestibility of dietary proteins and in vitro DIAAS analytical workflow based on the INFOGEST static protocol and its validation with in vivo data, Food Chem., 2023, 404(Pt B), 134720 CrossRef CAS PubMed.
- W. Michels and H. Frister, The modified OPA method with an alternative thiol component, Zentr. Steril., 2004, 12(2), 117–118 Search PubMed.
- V. A. Ayala-Rodriguez, A. A. Lopez-Hernandez, M. L. C. Lomeli, B. E. Gonzalez-Martinez and J. A. Vazquez-Rodriguez, Nutritional quality of protein flours of fava bean (Vicia faba L.) and in vitro digestibility and bioaccesibility, Food Chem.:X, 2022, 14, 100303 CAS.
- J. C. Sánchez-Rangel, J. Benavides, J. B. Heredia, L. Cisneros-Zevallos and D. A. Jacobo-Velázquez, The Folin–Ciocalteu assay revisited: improvement of its specificity for total phenolic content determination, Anal. Methods, 2013, 5(21), 5990–5999 RSC.
- P. Valdebouze, E. Bergeron, T. Gaborit and J. Delortlaval, Content and Distribution of Trypsin-Inhibitors and Hemagglutinins in Some Legume Seeds, Can. J. Plant Sci., 1980, 60(2), 695–701 CrossRef CAS.
- C. Vidal-Valverde, J. Frias, C. Diaz-Pollan, M. Fernandez, M. Lopez-Jurado and G. Urbano, Influence of processing on trypsin inhibitor activity of faba beans and its physiological effect, J. Agric. Food Chem., 1997, 45(9), 3559–3564 CrossRef CAS.
- F. M. Lajolo and M. I. Genovese, Nutritional significance of lectins and enzyme inhibitors from legumes, J. Agric. Food Chem., 2002, 50(22), 6592–6598 CrossRef CAS PubMed.
- T. Xiao, I. S. Roland, S. D. Nielsen, T. T. Le, E. Olesen, L. B. Larsen and N. A. Poulsen, Variation in composition of storage proteins, minor proteins and amino acids across faba bean cultivars, Food Chem., 2025, 483, 144225 CrossRef CAS PubMed.
- L. J. Corzo-Ríos, C. Jiménez-Martínez, M. S. Cid-Gallegos, A. Cardador-Martínez, J. Martínez-Herrera and X. M. Sánchez-Chino, Chemical and non-nutritional modification of faba bean (Vicia faba) due to the effect of roasting and boiling, Int. J. Gastron. Food Sci., 2022, 30, 100622 CrossRef.
- N. M. A. Moneam, Effects of Presoaking on Faba Bean Enzyme-Inhibitors and Polyphenols after Cooking, J. Agric. Food Chem., 1990, 38(7), 1479–1482 CrossRef CAS.
- A. Sharma and S. Sehgal, Effect of domestic processing, cooking and germination on the trypsin inhibitor activity and tannin content of faba bean (Vicia faba), Plant Foods Hum. Nutr., 1992, 42(2), 127–133 CrossRef CAS.
- R. Alonso, A. Aguirre and F. Marzo, Effects of extrusion and traditional processing methods on antinutrients and in vitro digestibility of protein and starch in faba and kidney beans, Food Chem., 2000, 68(2), 159–165 CrossRef CAS.
- G. M. Bornhorst, M. J. Ferrua and R. P. Singh, A proposed food breakdown classification system to predict food behavior during gastric digestion, J. Food Sci., 2015, 80(5), R924–R934 CrossRef CAS PubMed.
- C. Tanford, Protein denaturation, Adv. Protein. Chem., 1968, 23, 121–282 CrossRef CAS PubMed.
- B. M. Dunn, Overview of pepsin-like aspartic peptidases, Curr. Protoc. Protein Sci., 2001, 25(1), 21–23 CrossRef PubMed.
- U. Choe, J. M. Osorno, J. B. Ohm, B. Chen and J. Rao, Modification of physicochemical, functional properties, and digestibility of macronutrients in common bean (Phaseolus vulgaris L.) flours by different thermally treated whole seeds, Food Chem., 2022, 382, 132570 CrossRef CAS PubMed.
- I. Contardo, S. Gutierrez, J. Hurtado-Murillo and N. Escobar, Understanding the structural differences in chickpea globulins and their relationship with in vitro protein digestibility, Food Res. Int., 2025, 202, 115702 CrossRef CAS PubMed.
- D. Martineau-Côté, A. Achouri, M. Pitre, J. Wanasundara, S. Karboune and L. L’Hocine, Investigation of the nutritional quality of raw and processed Canadian faba bean (Vicia faba L.) flours in comparison to pea and soy using a human in vitro gastrointestinal digestion model, Food Res. Int., 2023, 173(Pt 1), 113264 CrossRef PubMed.
- R. R. Croy, J. A. Gatehouse, I. M. Evans and D. Boulter, Characterisation of the storage protein subunits synthesised in vitro by polyribosomes and RNA from developing pea (Pisum sativum L.) : I, Legumin, Planta, 1980, 148(1), 49–56 CrossRef CAS PubMed.
- K. Müntz, C. Horstmann and B. Schlesier, Vicia globulins, in Seed proteins, Springer, 1999, pp. 259–284 Search PubMed.
- S. S. Nielsen, S. S. Deshpande, M. A. Hermodson and M. P. Scott, Comparative Digestibility of Legume Storage Proteins, J. Agric. Food Chem., 1988, 36(5), 896–902 CrossRef CAS.
- T. D. Oluwajuyitan and R. E. Aluko, Structural and functional properties of fava bean legumin and vicilin protein fractions, Int. J. Food Sci. Technol., 2024, 59(9), 6698–6716 CrossRef CAS.
- I. M. Abu-Reidah, M. del Mar Contreras, D. Arraez-Roman, A. Fernandez-Gutierrez and A. Segura-Carretero, UHPLC-ESI-QTOF-MS-based metabolic profiling of Vicia faba L. (Fabaceae) seeds as a key strategy for characterization in foodomics, Electrophoresis, 2014, 35(11), 1571–1581 CrossRef CAS PubMed.
- I. M. Valente, A. R. J. Cabrita, N. Malushi, H. M. Oliveira, L. Papa, J. A. Rodrigues, A. J. M. Fonseca and M. R. G. Maia, Unravelling the phytonutrients and antioxidant properties of European Vicia faba L. seeds, Food Res. Int., 2019, 116, 888–896 CrossRef CAS PubMed.
- Y. Aoyama, K. Masuda, S. Mitsui and T. Yokoi, Diffusion of adzuki bean (Vigna angularis) oligomeric polyphenols from seed coats to cotyledons during boiling, Biosci., Biotechnol., Biochem., 2024, 88(12), 1453–1464 CrossRef CAS PubMed.
- L. Jakobek, Interactions of polyphenols with carbohydrates, lipids and proteins, Food Chem., 2015, 175, 556–567 CrossRef CAS PubMed.
- X. Yan, J. Zhao, Z. Zeng, M. Ma, J. Xia, W. Tian, G. Zhang, X. Gong, D. Gong and P. Yu, Effects of preheat treatment and polyphenol grafting on the structural, emulsifying and rheological properties of protein isolate from Cinnamomum camphora seed kernel, Food Chem., 2022, 377, 132044 CrossRef CAS PubMed.
- Y. J. Kim, D. M. Shin, E. J. Oh, Y. G. Chun, J. K. Shin, Y. S. Choi and B. K. Kim, Mechanisms underlying the changes in the structural, physicochemical, and emulsification properties of porcine myofibrillar proteins induced by prolonged pulsed electric field treatment, Food Chem., 2024, 456, 140024 CrossRef CAS PubMed.
- S. D. Zhou, Y. F. Lin, X. Xu, L. Meng and M. S. Dong, Effect of non-covalent and covalent complexation of (-)-epigallocatechin gallate with soybean protein isolate on protein structure and digestion characteristics, Food Chem., 2020, 309, 125718 CrossRef CAS PubMed.
- Y. Aguilera, R. M. Esteban, V. Benitez, E. Molla and M. A. Martin-Cabrejas, Starch, functional properties, and microstructural characteristics in chickpea and lentil as affected by thermal processing, J. Agric. Food Chem., 2009, 57(22), 10682–10688 CrossRef CAS PubMed.
- S.Ç Karataş, D. Günay and S. Sayar, In vitro evaluation of whole faba bean and its seed coat as a potential source of functional food components, Food Chem., 2017, 230, 182–188 CrossRef PubMed.
- S. Sayar, M. Turhan and S. Gunasekaran, Analysis of chickpea soaking by simultaneous water transfer and water-starch reaction, J. Food Eng., 2001, 50(2), 91–98 CrossRef.
- C. S. do Carmo, P. Silventoinen, C. T. Nordgård, C. Poudroux, T. Dessev, H. Zobel, A. K. Holtekjølen, K. I. Draget, U. Holopainen-Mantila and S. H. Knutsen, et al. Is dehulling of peas and faba beans necessary prior to dry fractionation for the production of protein- and starch-rich fractions? Impact on physical properties, chemical composition and techno-functional properties, J. Food Eng., 2020, 278, 109937 CrossRef.
- C. M. Chigwedere, T. F. Olaoye, C. Kyomugasho, Z. Jamsazzadeh Kermani, A. Pallares Pallares, A. M. Van Loey, T. Grauwet and M. E. Hendrickx, Mechanistic insight into softening of Canadian wonder common beans (Phaseolus vulgaris) during cooking, Food Res. Int., 2018, 106, 522–531 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |