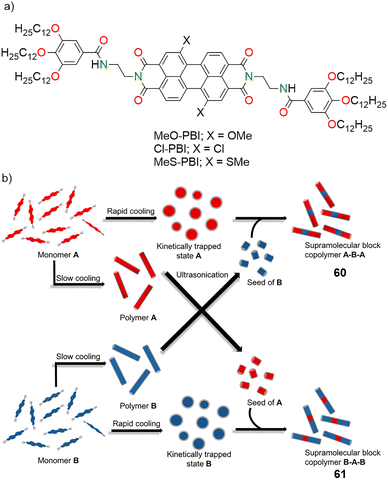
Advances in heterochromophoric perylene bisimide self-assemblies towards supramolecular electronics
Vidushi
Gupta†
,
Samaresh
Samanta†
and
Sanchita
Sengupta
 *
*
Department of Chemical Sciences, Indian Institute of Science Education and Research (IISER) Mohali, Knowledge City, Sector 81, P.O. Manauli, Punjab 140306, India. E-mail: sanchita@iisermohali.ac.in
First published on 26th March 2026
Abstract
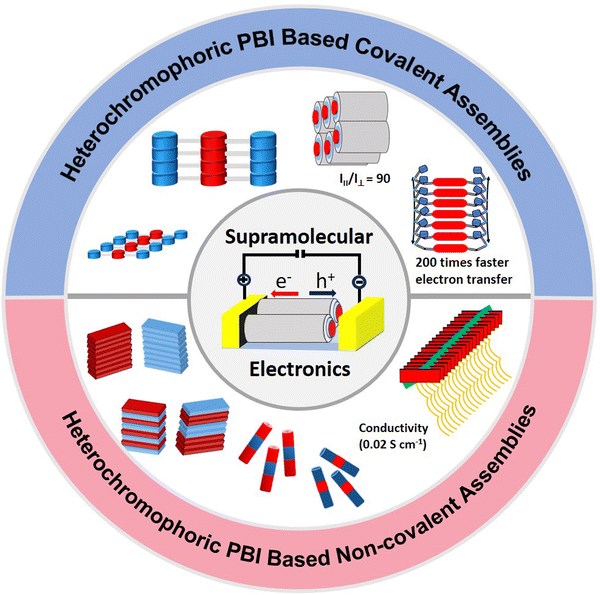
Perylene bisimide (PBI) dyes are the quintessential and most well-studied class of dyes for designing biomimetic light harvesting (LH) self-assembled systems owing to their exceptional photophysical features such as excellent absorption, high molar extinction coefficient and high fluorescence quantum yield, thermal robustness, high charge carrier mobilities, and efficient exciton diffusion properties in their assemblies. Although excellent reviews spanning many aspects of PBI dyes and their applications are available in the literature, none of these to date have focused on heterochromophoric PBI assemblies in solution and their applications in electronics. To bridge the gap, this review focuses on heterochromophoric PBI dye self-assemblies, their optical properties in solution, morphological aspects and applications in solar light harvesting and charge and energy transfer, and their eventual integration in supramolecular electronic devices. The PBI heterochromophoric assemblies have been categorized into two major categories, namely, (i) covalent heterochromophoric self-assemblies and (ii) heterochromophoric PBI systems assembled via non-covalent interactions such as π–π stacking, hydrogen bonding, charge transfer interactions or co-assembly of PBIs with other chromophores and scaffolds. The detailed structural intricacies, photophysical properties in the solution phase, and morphologies of these assemblies are discussed followed by their established supramolecular electronic applications and future potential in the frontier areas of materials science.
1. Introduction
Natural light harvesting (LH) systems of purple bacteria and green sulphur bacteria have served as inspiration for supramolecular chemists to design bioinspired chromophores and their aggregates that can emulate the exceptional structural intricacies and functional properties of natural LH systems.1 In purple bacteria, the cyclic arrangement of pigment–protein complexes that function in LH, ultrafast exciton transport and eventual electron transfer to the reaction centre has inspired generations of chemists to design and synthesize aesthetic and complex artificial cyclic LH arrays and macrocycles.2–4 On the other hand, LH systems of green sulphur bacteria consisting of chlorosomes possess an elevated level of structural complexity and sophistication compared to that of purple bacteria.5 In chlorosomes, bacteriochlorophylls are spatially organized in cyclic tubular arrangement through π–π stacking, directional hydrogen bonding and metal–ligand coordination,6,7 where chromophores in a slip-stacked arrangement lead to J-aggregation, resulting in efficient excitonic coupling and long-range energy transport to the reaction centre.8,9 These intriguing findings provided impetus for the development of several pioneering bio-inspired artificial LH molecules pre-programmed for self-assembly.10–15 Accordingly, seminal progress has been achieved in the area of supramolecular polymers11,12,16 over several decades, including the in-depth understanding of their structural and functional properties and their applications in supramolecular electronics.12,17–20In the context of chromophore selection for designing biomimetic LH self-assembled systems, perylene bisimide (PBI) dyes have been the quintessential and most well-studied class of dyes, rich with exceptional photophysical properties such as excellent absorption and fluorescence quantum yield, high thermal robustness, high charge carrier mobility and efficient exciton diffusion properties in their assemblies13,15,21–24 Numerous benchmark reviews on the structures, properties and applications of PBIs are known in the literature.13,15,21,22,25 While some of the comprehensive reviews have summarized the synthetic strategies of PBI core modifications such as functionalization at bay and ortho positions, substitution at bay positions, direct functionalization of core-unsubstituted PBIs, core expansion and annulations,21,26 others have summarized core enlargement strategies leading to higher rylene diimide derivatives.22 Likewise, several strategies for the PBI self-assembly into J-aggregates,27–32 their self-sorting,13,33 PBI supramolecular34–36 and block co-polymer37 formation, one-dimensional (1D) nanostructures,38 polymorphs,39 host–guest PBI cyclophanes,40 metallosupramolecular squares,41,42 PBI catenanes43 and macrocycles43,44 have been investigated and reviewed systematically. Furthermore, several pioneering reviews on the applications of PBI J-aggregates in organic electronics and photonics such as solar cells45 and photodetectors,46 aggregation induced emissive PBIs for photothermal conversions,47 photocatalytic water oxidation,42 supramolecular photocatalysis38,48 have inspired studies towards their ever expanding application port-folio in materials science research frontiers.
Self-assembly of PBIs is principally driven by non-covalent interactions such as π–π stacking, and other non-covalent interactions such as van der Waals forces and hydrogen bonding are added through molecular design to achieve desired self-assembled structures.28,49–51 The geometry and strength of these interactions dictate the formation of H- and J-aggregates each with characteristic optical signatures.28 While H-aggregates are characterized by sandwich type stacking arrangement of chromophores leading to blue-shifted absorption and quenched fluorescence, J-aggregates involve a head-to-tail arrangement, resulting in red-shifted absorption and enhanced fluorescence.28 In this context, heterochromophoric assemblies are of particular importance as they facilitate directional charge and energy transfer through segregated channels52 and ambipolar charge transport properties, enabling applications in organic and supramolecular electronics.19 Moreover, heterochromophoric assemblies undergo self-sorting,13 whereby different chromophores can self-sort either narcissistically (each chromophore prefers its own type for assembly) or socially (distinct chromophores co-assemble in a controlled sequence). Heterochromophoric assemblies thus provide a versatile platform for designing LH, stimuli-responsive and optoelectronic materials with distinct properties. Heterochromophoric assemblies by virtue of their well-organized segregated p–n stacks serve as efficient conduits for charge transport and are vital for supramolecular electronics.19,53
With regard to applications in organic electronics, PBIs are among the most important classes of π-conjugated dyes that exhibit high charge-carrier mobility,54 low threshold voltage,55 and sufficiently high on–off current ratios (Ion/Ioff).56 Compared with conventional inorganic semiconductors, PBI-based materials offer several advantages for their incorporation in devices, such as low fabrication cost, high-throughput manufacturing, low-temperature processability on flexible substrates, and tunable electronic properties.57 These characteristics make PBI based materials attractive for a wide range of next-generation electronic applications, including low-power flexible displays, cost-effective radio-frequency identification (RFID) tags, printable sensors, bipolar transistors,58 organic p–n junctions, polymer solar cells,59–62 light-emitting diodes,63 and organic thin-film transistors (OTFTs).64–71 Some of the most important features of PBI self-assemblies include their pronounced J-aggregate formation characterized by sharp and bathochromically shifted absorption originating due to long-range Coulomb coupling.72,73 Recent improved understanding of J-aggregation revealed that PBIs preferably form charge transfer (CT) mediated J-aggregates while some other dye classes such as squaraines prefer CT coupled J aggregation.29,46,72,73 CT coupled J-aggregates by virtue of their red-shifted absorption bands and efficient charge transport behaviour have proven to be efficient light harvesters for organic solar cells (OSCs) with excellent photocurrent. Furthermore, such J-aggregates have also succeeded as efficient materials for wavelength-dependent photodiodes and photodetectors.46 PBI aggregation studies have provided a new paradigm in the realm of supramolecular electronics, where factors such as controlling the aggregation domains, ensuring macroscopic alignment of assemblies, ensuring the monodispersity of aggregation and insights into the aggregation dynamics play key roles.19,74–76
PBI aggregation behaviour has also been reported in aqueous systems wherein various water soluble PBIs77,78 and amphiphilic dendron functionalized PBI–porphyrin dyads formed micellar and liposomal aggregates with prominent optical behaviour and superstructure morphologies.79–81 A thorough understanding of self-assembly processes of PBIs in aqueous systems will lead to better rational design of bio-mimetic synthetic molecules and their assemblies in the future.77,78 By virtue of stimuli responsiveness of optical properties of π-assemblies of PBIs where subtle changes in parameters such as temperature, pH, solvent composition, and pressure lead to perturbation in the chromophore arrangement, resulting in pronounced changes in optical properties, they have found applications as stimuli responsive smart materials,75 fluorescence turn-on sensors82,83 for volatile organic compounds (VOCs) and nitroaromatics, biosensors and diagnostic probes.84
In recent years, PBI supramolecular polymers have found prevalent applications in photocatalytic organic transformations where several strategies such as precipitation, charge screening, surfactant assisted self-assembly, H-bond aided assembly and interaction with metals or macromolecules have been adopted.38 Several of these strategies have been successfully adopted for organic transformations, photocatalytic hydrogen evolution and photocatalytic CO2 reduction to value added chemicals such as CO, CH4 and so on.85 Our group has recently reviewed the development of PBI based photocatalysts for hydrogen evolution,86 where self-assembly of PBIs is a vital strategy towards achieving the best photocatalytic H2 evolution efficiencies among all categories of photocatalysts.87,88
Surprisingly however, compared to the large number of comprehensive reviews on PBI dyes and their homo-assemblies, PBI heterochromophoric assemblies are much less covered in the literature.47,89 In this context however, heterochromophoric compounds of other important rylene diimides, namely, naphthalene diimides, have been thoroughly reviewed in the literature.90–92 While one of the recent reviews discussed important examples of heterochromophoric assemblies of PBIs,89 their solution self-assembly was not reviewed in detail. Herein, we have reviewed the examples of PBI heterochromophoric self-assemblies in solution and presented the chronological developments over the last two decades. While the present review summarizes the development of heterochromophoric self-assemblies containing PBI as the central chromophore/one of the chromophores, the examples discussed are focussed on solution phase self-assemblies only. Herein, the heterochromophoric PBI self-assemblies have been categorised into two major groups, namely, (i) covalent heterochromophoric self-assemblies and (ii) non-covalent heterochromophoric PBI assemblies and co-assemblies of PBIs with other chromophores or scaffolds. A few minor categories like heterochromophoric foldamers, oligomers and CT hetero-assemblies of PBIs have also been discussed in the context of their established or potential applications in (supra)molecular electronics. For the major categories (Fig. 1), detailed structural intricacies, photophysical studies and charge/electron transfer properties of the aggregates in the solution phase are discussed followed by in-depth discussion of the assembly morphologies. Finally, the possible implications of those studies and the applications of heterochromophoric PBI assemblies in supramolecular electronics are discussed.
 | ||
| Fig. 1 Schematic representation of heterochromophoric PBI self-assemblies according to two major categories with applications in supramolecular electronics. | ||
To keep the article succinct and focussed, heterochromophoric self-assemblies of PBIs in the solid-state and condensed phase such as xerogels, hydrogels, liquid crystals and chiral self-assemblies93–100 have not been included except for a few important representative examples, since there are comprehensive reviews on each of these topics.13,15 Furthermore, studies on heterochromophoric PBI self-assemblies in the solid state and those focussed exclusively on surface self-assemblies101,102 are not included. Finally, heterochromophoric rotaxanes, catenanes, macrocycles of PBIs and their metallosupramolecular polymers15 have been excluded, as pioneering reviews are available on this topic.40,42–44 The self-assembly studies limited to the solid/condensed phase,103 thin films104,105 and π-stacks3,106,107 performed using transient spectroscopy reviewed comprehensively elsewhere4,103–105,108,109 are also not covered here. With the availability of excellent reviews spanning many aspects of PBI dyes and their applications, the present review aims to provide a comprehensive overview of the heterochromophoric PBI assemblies in solution, serving as a guiding tool for the development of newer heterochromophoric systems for self-assembly and their cutting-edge applications and future potential in supramolecular electronics and other areas.
2. Heterochromophoric PBI self-assemblies
2.1. Covalent Heterochromophoric PBI self-assemblies
Heterochromophoric covalently connected systems can self-organize through H and J-aggregate formation driven by chromophore–chromophore coupling and by their intermolecular interactions. Such self-organization results in formation of well-defined J- and H-aggregates including core–shell architectures as summarized in Fig. 2. There are specific advantages of covalent linkage of donor (D) and acceptor (A) type chromophores such as fixing the donor–acceptor (D–A) ratio and mutual orientation, eliminating phase separation and maximising the D–A interface, thereby reducing compositional inhomogeneity that is common in blended systems.109 Furthermore, covalent connectivity ensures controlled spatial disposition and proximity of different chromophores, leading to reproducible morphology, facile pathways for holes and electrons and desirable (opto)electronic properties. The fixed D–A distances in such systems enable efficient interchromophoric coupling, rapid and efficient photoinduced charge separation, improved charge diffusion length and longer charge recombination time, and overall high charge carrier yield.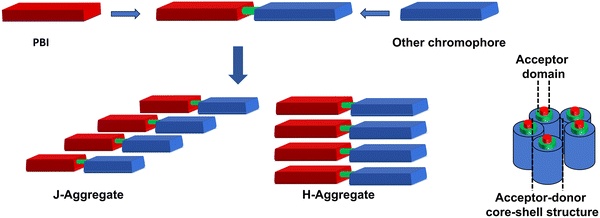 | ||
| Fig. 2 General representation of self-assemblies of heterochromophoric covalently connected PBI systems. | ||
Würthner, Schenning, and co-workers (2004) studied the co-assembly of D–A dye systems using oligo(p-phenylenevinylene) (OPV) and PBI chromophores to form supramolecular p–n heterojunctions through non-covalent interactions.110 They synthesized and compared two systems: covalently linked OPV–PBI–OPV triad 1 and hydrogen-bonded OPV–PBI arrays 2a–c and 3 (Fig. 3a), where OPV units were decorated with chiral aliphatic chains. Both formed J-type π-stacked aggregates in aliphatic solvents such as methylcyclohexane (MCH), evident from bathochromic shifts (∼600 nm) in absorption spectra caused by exciton coupling. In temperature-dependent UV/vis studies, all three hydrogen bonded complexes 2a–c (spectral changes are shown in Fig. 3b(i) for array 2b) and covalently bonded compound 1 exhibited spectral changes similar to those in the concentration-dependent experiments (Fig. 3b(ii)). Importantly, only the hydrogen-bonded systems self-assembled into hierarchical, left-handed helical co-aggregates in solution, as shown by circular dichroism (CD) spectroscopy. Aggregates of 2b further organized into right-handed supercoils in the solid state, visualized by atomic force microscopy (AFM). The systems were aggregated through an isodesmic pathway that exhibited reversible thermal behaviour, with improved stability and reversibility attributed to hydrogen bonding. Van’t Hoff analysis showed that self-assembly was enthalpy-driven, indicating stronger binding (more negative ΔH and ΔS values) and tighter packing in hydrogen-bonded systems such as 2b compared to covalent triad 1.110 The extent of aggregation increased with the conjugation length of OPV. Nonetheless, both covalent and non-covalent systems had similar binding constants, suggesting that π–π stacking was the major driving force. However, hydrogen bonding resulted in more planar packing and sharper melting transitions, like deoxyribonucleic acid (DNA) base stacking. CD studies confirmed chiral excitonic coupling (indicated by bisignate Cotton effects) in helically stacked aggregates of 2b and 2c, with the strongest chiral induction observed in array 3. While aggregates in array 2a were unable to show the CD signal, the transfer of chirality was attributed not only to side chains but mainly to cooperative helical stacking, which was missing in the twisted covalent system such as 1. Molecular modelling supported the formation of left-handed helices and provided evidence for the diastereomeric enrichment of the M enantiomer of PBI (Fig. 3c). This work therefore offered a strategy to build chiral, hierarchical p–n junctions via self-organization.
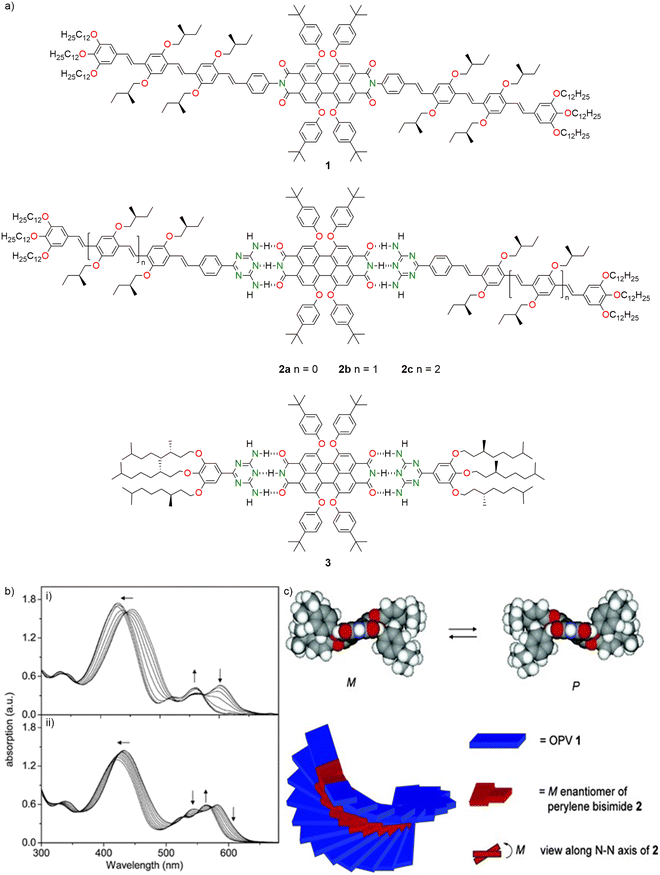 | ||
| Fig. 3 (a) Molecular structures of the compounds 1–3. (b) Temperature dependent UV/vis spectra from 10 to 80 °C of (i) 2b and (ii) 1 in MCH. Arrows denote changes upon increasing temperature. (c) Energy minimized structures of the M (left) and the P (right) enantiomers of PBI and the left-handed helical stacking model for the 2b complex. Adapted from Fig. 3b and c with permission from ref. 110. Copyright 2004 American Chemical Society. | ||
Janssen and co-workers investigated (2006) how intermolecular orientation within self-assembled architectures of OPV–PBI arrays affects the kinetics of light-induced CT processes in these systems. Three homologous OPV–PBI–OPV arrays with the OPV donor and the PBI acceptor, 4, 5 and 6, each differing in the substituents on the perylene core, were designed and synthesized (Fig. 4a).111 Arrays formed either J-type (slipped) or H-type (face-to-face) aggregates, depending on their substituents. UV/vis absorption (Fig. 4b) and CD spectroscopy were utilized to probe aggregate type and structure, and their charge separation and recombination dynamics were studied by transient absorption (TA) spectroscopy. At lower temperature, all three arrays displayed spectral features characteristic of aggregated species, while heating led to shifts consistent with supramolecular dissociation. Array 6 (with bulky bay substituents) formed J-type aggregates, while array 5 (less steric hindrance) formed H-type aggregates. J-type aggregates (array 4 and array 6) enabled very fast intermolecular charge separation (kCS = 1.2 × 1012 s−1 and 2.9 × 1012 s−1) due to reduced D–A distances (∼4 Å), much faster than that in a molecularly dissolved state. However, H-type aggregate array 5 did not show enhanced kinetics; electron transfer occurred at similar rates as in the molecularly dissolved state, since intermolecular spacing was not reduced. Faster recombination was observed in J-type aggregates of arrays 4 and 6 (kCR = 2 × 109 s−1 and 8.3 × 1010 s−1), consistent with Marcus inverted region behaviour, i.e., a shorter D–A distance led to lower negative free energy for recombination and hence faster kinetics.111
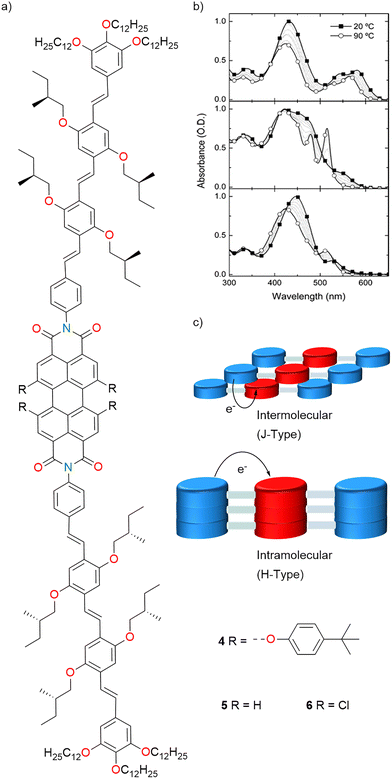 | ||
| Fig. 4 (a) Chemical structure of OPV-PBI-OPV arrays 4–6. (b) Absorption spectra of 4 (top 5 × 10−5 M), 5 (middle, 2 × 10−6 M), and 6 (bottom, 1.3 × 10−6 M). (c) Cartoon of 1D packing of arrays 4–6 to give J-type and H-type aggregates. Fig. 4b is adapted with permission from ref. 111. Copyright 2006 American Chemical Society. | ||
However, due to the larger distances in H-type structures in array 5, such CT behaviour was not detected (Fig. 4c). Molecular mechanics calculations provided insights into the molecular packing and corroborated the experimental observations.111 This study showed that shorter D–A distances in J-type aggregates are responsible for accelerated CT and recombination, thus providing key insights for functional material design.
Müllen and co-workers (2012) synthesized a series of covalently linked dyads (7–11) composed of hexa-peri-hexabenzocoronene (HBC) and PBI as the electron donor and acceptor respectively, connected via a rigid bridging unit (Fig. 5a).112 To investigate the effect of HBC and PBI, and the linker length, on self-assembly behaviour, the ratios of HBC to PBI and the spacer lengths were systematically varied. Scanning tunneling microscopy (STM) and two-dimensional (2D) wide-angle X-ray scattering (2D-WAXS) revealed well-ordered 2D supramolecular structures for all dyads, both in solution and in the solid state. These structures enabled tunable D–A orientations and separations at the nanometre scale. The self-assembled morphologies depend strongly on the molecular symmetry of the dyads, with dyads 7 and 8 (HBC–6PBI) forming either columnar or star-shaped nanostructures with nano-segregated HBC and PBI stacks. In contrast, dyads 9 (HBC–2PBI) and 10 (HBC–PBI) formed interdigitating networks characterized by alternating arrangements of HBC and PBI moieties (Fig. 5b–e).112 UV/vis absorption spectroscopy revealed aggregate formation in toluene for dyads 7 and 8 and linear dyads 9, 10 and 11 at a concentration of 10−5 M. A noticeable decrease in the A0–0 band intensity relative to the A0–1 band was observed for PBI, indicating H-type aggregation, which is commonly associated with exciton coupling and hypsochromically shifted absorption. After photoexcitation of the donor HBC, a photoinduced electron transfer (PET) was observed between HBC and PBI. However, only the nano-segregated stacks showed charge transport pathways in bulk, highlighting the importance of this system for optoelectronic applications such as in organic photovoltaics and field-effect transistors (OFETs).
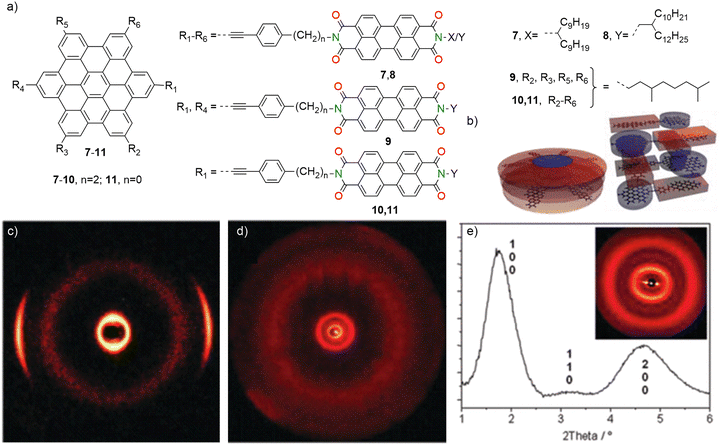 | ||
| Fig. 5 (a) Chemical structures of the HBC–PBI dyads 7–11. (b) Schematic representation of the columnar stacks of dyads 7 (left) and 8 and interdigitating stacks of dyad 10 (right). (c) 2D-WAXS of extruded fibre 10, (d) 2D-WAXS and (e) 2D-small-angle X-ray scattering (SAXS) (inset) of 7 as powder with the corresponding integration (Miller's indices are used to assign the reflections). Fig. 5b–e is adapted with permission from ref. 112 copyright 2012 American Chemical Society. | ||
Fukuzumi, Kim, and co-workers (2013) studied how the solvent environment influences the photoinduced charge separation in a molecular triad 12 (An2PBI), which consists of two anthracene (An) donors and one PBI acceptor (Fig. 6a).81 In methanol (MeOH), 12 exhibited monomeric behaviour similar to its individual chromophores, whereas in water, the triad formed polymeric self-assemblies. In MeOH, the triad remained molecularly dissolved, and no stable charge-separated species were detected. Fast charge recombination caused inefficient electron transfer, and the singlet excited state of PBI was quenched, preventing the formation of long-lived charge-separated states. In water, 12 self-assembled into H-aggregates because of hydrophilic–hydrophobic interactions. These aggregates facilitated π-stacking of PBI units and folding of the flexible linkers. The close and more rigid D–A configuration allowed for efficient and ultrafast photoinduced charge separation (Fig. 6b). The charge separation rate in MeOH was about 1.2 × 1010 s−1, whereas in water, it was roughly 2.5 × 1012 s−1, about 200 times faster. The charge-separated state in water lasted around 30 ps. It is hypothesized that π-stacked PBIs stabilized the radical anion, while folding of the linker proximally positioned the anthracene, facilitating charge separation and reducing the charge recombination. This work shows that non-covalent interactions and solvent-induced self-assembly can significantly boost the efficiency of photodriven charge separation in D–A systems, with implications for artificial photosynthesis and organic photovoltaics.
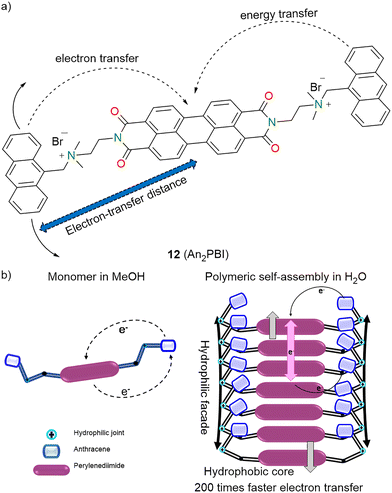 | ||
| Fig. 6 (a) Molecular structures of 12 (An2PBI). (b) Comparison of the photoinduced processes of 12 in MeOH and water with illustration of its conformational arrangements. | ||
Ma and co-workers (2014) investigated how side-chain functionalities (amphiphilic vs. lipophilic) and linker lengths influence the self-assembly, phase separation, and morphologies of D–A dyads composed of 4,7-bis(2-thienyl)benzothiadiazole (T2BTZ) as the donor and PBI as the acceptor (Fig. 7a and b).113 Four D–A dyads were synthesized, including amphiphilic side chains with a short linker 13a (Samphi) and long linker 13b (Lamphi) and lipophilic chains with a short linker 14a (Slipo) and long linker 14b (Llipo). UV/vis absorption (Fig. 7c) and nuclear magnetic resonance (NMR) spectroscopy revealed that the dyads with long-linkers, 14b and 13b (Llipo and Lamphi), could fold intramolecularly resulting in pronounced D–A interactions even in dilute solutions. Conversely, dyads with short-linkers, 14a and 13a (Slipo and Samphi), did not fold intramolecularly but exhibited strong intermolecular D–A aggregation at higher concentrations (Fig. 7d). Amphiphilic side chains helped in weakening of D–A interactions in Lamphi thereby encouraging phase separation. SAXS and electron microscopy revealed that amphiphilic dyads 13b and 13a (Lamphi and Samphi) formed thin nanofibres (∼10 nm), while lipophilic dyads 14b and 14a (Slipo, Llipo) formed thicker fibres or nanoparticles (∼50 nm). Dyad 13b (Lamphi) showed better phase separation due to incompatible side chains, whereas 13a (Samphi) formed a gel upon solvent annealing, indicating strong self-assembly. Cyclic voltammetry (CV) was used to evaluate the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels of these dyads, with a sufficient energy offset (∼0.62 eV) observed for efficient photoinduced charge separation. The linker lengths and side-chain amphiphilicities critically influenced the folding (Fig. 7e) and as a result intermolecular interactions, self-assembly and eventual phase separation of these systems.113 Among all the dyads, amphiphilic dyad 13b (Lamphi) exhibited promising nano-organization behaviour that is ideal for (supra)molecular electronic applications, particularly for bulk heterojunction (BHJ) solar cells.
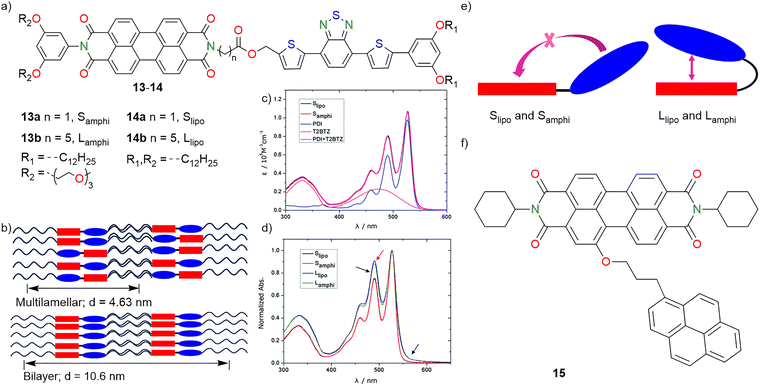 | ||
| Fig. 7 (a) Chemical structures of dyads 13a,b and 14a and b. (b) Scheme representing the phase separation diagram of Llipo with multilamellar structure (above) and of Lamphi with bilayer structure. (c) UV/vis absorption of dyads with short linkers and reference compounds. (d) Normalized absorption of dyads with short and long linkers. (e) Schematic diagram of intramolecular folding of dyads with short linkers and long linkers. (f) Molecular structure of compound 15. Fig. 7c and d are adapted with permission from ref. 113. | ||
Li and co-workers reported (2014) the synthesis, self-assembly, and electronic properties of a novel dyad molecule 15 consisting of PBI covalently linked to pyrene at one of the bay-positions via a flexible chain (Fig. 7f).114 The structural elucidation of 15 was performed using 1H NMR, mass-spectrometry and elemental analyses. Slow evaporation of chloroform (CHCl3) induced self-assembly of 15 into highly ordered nanobelts. X-ray diffraction (XRD) analysis showed long-range order with an edge-on molecular orientation and a π–π stacking distance of ∼0.31 nm, indicating H-type aggregation. Absorption spectra of films showed enhanced vibronic features, indicating strong molecular interactions. A bathochromic shift in pyrene absorption was observed, and the formation of a new emission band at 707 nm suggested excimer-like emission due to face-to-face stacking. Scanning electron microscopy (SEM) study revealed uniform nanobelt structures of ∼50 µm length and ∼230 nm width, with high aspect ratios.114 Films prepared by the quasi-Langmuir–Schäfer method exhibited a conductivity of 4.85 × 104 S cm−1 under ambient light, while conductivity decreased to 1.31 × 10−6 S cm−1 in the dark. The conductivity of 15 estimated using current–voltage (I–V) measurements in the presence of ambient light and dark conditions (an on/off ratio of ∼300) suggests the potential application of these assemblies in photodetectors and in supramolecular electronics.
Ribierre and co-workers (2016) investigated how molecular structure and self-assembly impact the CT properties of 16 (D–A dyad) and 17 (D–A–D triad) based on triphenylene (D) and PBI (A) units in thin films. PBI and triphenylene were covalently linked by flexible decyloxy alkyl chains and formed discotic liquid crystalline (LC) nanostructures (Fig. 8a).115 The dyad and triad self-organized into columnar mesophases and the dyad formed hexagonal columnar (Colhex) phases with alternating D and A stacks (Fig. 8b), while triads formed oblique columnar (Colobl) phases with distinct D and A columns, leading to two different D–A distances (1.4 nm and 2.5 nm) (Fig. 8c). These arrangements were characterized using XRD, microscopy, and grazing incidence X-ray scattering (GIXS) techniques. In dichloromethane (DCM) and apolar MCH solvents at lower concentrations, the dyad showed monomeric behaviour, while at higher concentrations in MCH, the dyad showed aggregation. Fluorescence studies of the dyad in MCH revealed aggregation-induced quenching upon increasing the concentration as evident from decreasing emission intensity. At low concentrations, the spectra show well-defined vibronic features typical of isolated PBI chromophores. Thin films showed distinct aggregation behaviour where the dyad formed H-aggregates while the triad formed J-aggregates. Solid films exhibited pronounced photoluminescence (PL) quenching attributed to photoinduced CT from the D to the A components. Compared to the solution, molecular packing in films promoted CT over energy transfer and the CT dynamics was probed by subpicosecond pump–probe spectroscopy. The formation of CT states occurred within ∼0.23 ± 0.05 ps and ∼0.27 ± 0.05 ps for dyad and triad films respectively where the CT recombination time was found to be ∼234 ± 5 ps for the dyad, while the triad showed biexponential decay (∼151 ± 11 ps for 82% and ∼358 ± 32 ps for 18%). The recombination kinetics correlated with the D–A spacing, where a longer D–A distance extended the CT state lifetime without significantly delaying its formation.115 Well-separated columnar organizations in triads favoured efficient CT and delayed recombination, features that are highly beneficial for supramolecular electronic applications.
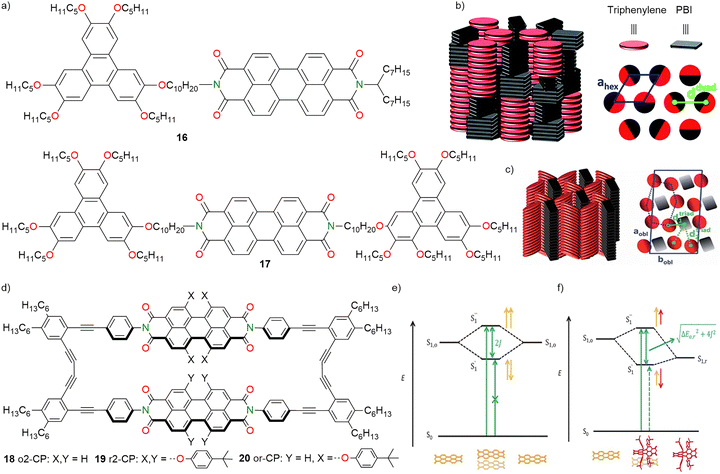 | ||
| Fig. 8 (a) Chemical structures of dyad 16 and triad 17. (b) Schematic representation of (left) the self-organization within the Colhex mesophases of the dyad 16 and (right) the hexagonal lattice formed by undifferentiated columns. (c) Schematic representation of (left) self-organization within the Colobl mesophases of the triad. (d) Chemical structures of 18–20. (e) Schematic representation of the exciton state diagram for the H-type PBI dimer stack constructed from identical and (f) different chromophores in a hypothetical collinear arrangement. Fig. 8b and c are adapted with permission from ref. 115. Copyright 2016, The Royal Society of Chemistry and Fig. 8e and f are adapted with permission from ref. 116. Copyright 2016, Wiley-VCH. | ||
Engel, Würthner, and co-workers (2016) investigated how the exciton-vibrational coupling affects the optical absorption properties of PBI dye stacks, particularly in homo-dimer (18, 19) and hetero-dimer (20) configurations (Fig. 8d).116 The PBI dimers formed H-type aggregates that led to blue-shifted absorption spectra with decreased 0–0 transition intensity. Coupling between electronic excitations (excitons) and intramolecular vibrations (mainly C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching) governed the absorption spectral shapes. Strong vibrational coupling enhanced 0–1 and 0–2 vibronic bands, especially when the 0–0 band was suppressed by H-aggregation (Fig. 8e). Homodimers showed symmetrical splitting and predictable vibronic features, while the heterodimers exhibited asymmetry in the excitonic coupling and more complex vibronic features attributed to the distinct energy levels of the two chromophores (Fig. 8f).116 Theoretical simulations corroborated well with experimental absorption spectra, confirming the role of both coulombic (excitonic) interactions and vibronic coupling. Understanding this interplay is therefore essential for the rational design of organic photonic and optoelectronic materials.
C stretching) governed the absorption spectral shapes. Strong vibrational coupling enhanced 0–1 and 0–2 vibronic bands, especially when the 0–0 band was suppressed by H-aggregation (Fig. 8e). Homodimers showed symmetrical splitting and predictable vibronic features, while the heterodimers exhibited asymmetry in the excitonic coupling and more complex vibronic features attributed to the distinct energy levels of the two chromophores (Fig. 8f).116 Theoretical simulations corroborated well with experimental absorption spectra, confirming the role of both coulombic (excitonic) interactions and vibronic coupling. Understanding this interplay is therefore essential for the rational design of organic photonic and optoelectronic materials.
Frauenrath and co-workers (2017) investigated the design, synthesis, and photophysical behaviour of A–D–A triad molecules, consisting of PBI and quaterthiophene, with polyisobutylene chains that promoted the formation of lamellar nanostructures (Fig. 9a).117 Two triad derivatives, 21a (PTP-9) and 21b (PTP-19), with different lengths of poly(isobutylene) side chains were synthesized. These molecules adopted a coil-rod-coil architecture, enabling microphase segregation and the formation of vertically oriented lamellae in thin films. The triads formed well-ordered lamellar phases aligned parallel to the substrate achieved by mechanical rubbing and thermal annealing. The longer side chains in 21b (PTP-19) assisted in maintaining an “edge-on” orientation even after rubbing and heating (Fig. 9b–d).117 Using TA spectroscopy, faster charge separation and slower charge recombination were observed, particularly in thin films.
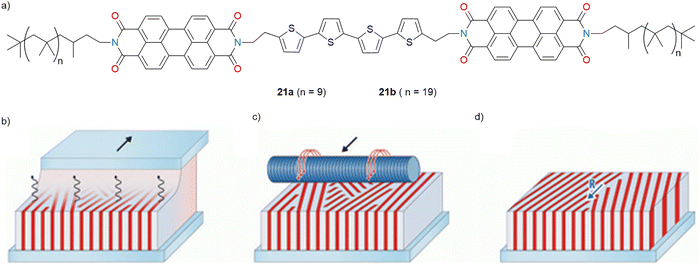 | ||
| Fig. 9 (a) Molecular structure of the polymer-substituted A–D–A triads 21a (PTP-9) and 21b (PTP-19). (b) Thin films of 21a or 21b exhibiting a lamellar phase with an ‘‘edge-on’’ orientation of the lamellae relative to the substrate. (c) Mechanical rubbing and annealing resulting in (d) a macroscopic alignment of the lamellar phase parallel to the rubbing direction. Fig. 9b–d are adapted with permission from ref. 117. Copyright 2017, The Royal Society of Chemistry. | ||
Those films exhibited long-lived charge-separated states for 21b (PTP-19) (∼150 ps) and showed slower recombination than 21a (PTP-9). The enhanced nano-structural order observed in 21b (PTP-19) contributed positively to charge delocalization and stabilization.
Würthner, Lehmann, and co-workers (2019) developed a photoconductive D–A dyad of PBI 22b (PBI2T) that self-assembled into LC core–shell nanostructures, enabling efficient photoinduced charge separation and transport. Compound 22b consisted of a tetraphenoxy-substituted PBI with four bithiophene donor side arms (Fig. 10a and b) and exhibited notable concentration-dependent aggregation behaviour in toluene. Comparison of the UV/vis absorption spectra of the monomeric solution in CHCl3 and the thin film highlighted the distinct coupling behaviour of the two chromophores in 22b.118 Compound 22b formed a highly ordered, helical LC phase composed of a coaxial D–A core–shell structure. Within this phase, six hydrogen-bonded PBI strands assembled into a sextuple-stranded 161 helix with strong π–π stacking and nanosegregated donor (bithiophene) and acceptor (PBI) domains. Structural and spectroscopic analyses (WAXS, middle-angle X-ray scattering, Fourier transform infrared (FTIR) spectroscopy and UV/vis spectroscopy) confirmed a hexagonal columnar arrangement where PBI cores aligned along the columnar axis and donor arms oriented perpendicularly (Fig. 10c, d, f and g). At low concentrations, PBI monomers absorbed maximally at 560 and 524 nm, while at higher concentrations they self-assembled into J-aggregates, characterized by a pronounced bathochromically shifted absorption maximum at 631 nm. The occurrence of isosbestic points at 578 and 476 nm confirmed the transition between two distinct species. The degree of aggregation (αagg) was modelled using the cooperative nucleation–elongation model. Global fitting yielded an elongation constant of Kelong = 8000 M−1 and a cooperativity constant of σ = 1.13 × 10−3 assuming a nucleus size of two molecules. Accordingly, the nucleation constant was determined as Knuc = 9 M−1. Temperature-dependent UV/vis absorption studies of 22b in MCH/toluene (v/v, 1/1, c0 = 2.09 × 10−4 M) revealed a transition from monomerically dissolved PBI obtained at high-temperature, characterized by absorption bands at 552, 516, and 381 nm, to self-assembled 22b at lower temperature, characterized by main transitions at 634 and 372 nm with isosbestic points at 474 and 569 nm (Fig. 10e).118 The calculated degree of aggregation could be accurately described by the cooperative nucleation–elongation model (Fig. 10h). To overcome trap sites and improve exciton mobility, 22b was investigated as a photoconductive material. Thin films (8–10 nm) prepared by spin-casting of 22b onto Si/SiO2 substrates with gold electrodes and annealed at 180 °C showed strongly enhanced photocurrents under white-light irradiation (38 mW cm−2). At +100 V, dark currents were only a few hundred femtoamperes, while photocurrents reached approximately 1 nA, with linear intensity dependence and instantaneous switching (<200 ms). Compared to the parent compound 22a (PBI 0T), which showed only about 50 pA photocurrent in the LC state, 22b exhibited a roughly 20-fold higher photoresponse. This improvement was attributed to its D–A design, enabling efficient PET, and to its sextuple-stranded helical self-assembly, which created robust, spatially separated electron- and hole-transport domains while suppressing recombination.
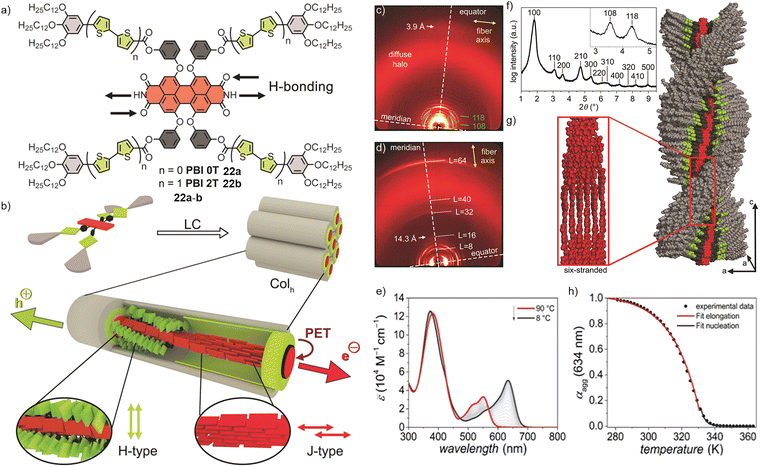 | ||
| Fig. 10 (a) Chemical structures of 22a and b. (b) Molecular arrangement of 22b in the columnar hexagonal LC phase with J-coupled PBIs (red) and H-coupled bithiophene units (green). WAXS diffraction pattern of (c) lying and (d) standing fibre of PBI 2T. (e) Temperature-dependent UV/vis spectra (90–8 °C) of 22b in MCH/toluene (v/v, 1/1). (f) Integrated intensities and assignment of the Miller indices of the Colh lattice of 22b with the inset showing integration of the off-meridional signals. (g) Schematic representation of the self-assembled structure of the sextuple-stranded 161 helix of 22b. (h) Plot of the degree of aggregation ∝agg at 634 nm over the temperature according to cooperative nucleation-elongation model. Fig. 10 is adapted with permission from ref. 118. Copyright 2019, Wiley-VCH. | ||
The same groups (2020) studied how trialkoxyphenyl-oligothiophene (TAPOT) units at the bay positions of PBIs affected their self-assembly behaviour by impacting directly their electronic response.74 They synthesized TAPOT-functionalized tetraphenoxy PBIs: 23a (PBI1T), 23b (PBI2T), and 23c (PBI3T), with mono-, bi-, and terthiophene units (Fig. 11a).74 In contrast to previous approaches, this design uniquely offered separate charge-transport channels for electrons and holes, along with the ability to adjust the number of strands in the coaxial cable core (Fig. 11b). In toluene, UV/vis spectroscopy revealed monomers at lower concentrations and aggregates at higher concentrations, evident from isosbestic points and an ∼80 nm bathochromic shift of the absorption maxima at high concentration. Aggregation followed a cooperative nucleation–elongation mechanism driven by hydrogen bonds and π–π interactions (Fig. 11d). The cooperativity parameters (σ) for 23a, 23b, and 23c are 3.0 × 10−3, 1.1 × 10−3, and 1.0 × 10−3, respectively, indicating increasing aggregation strength with longer side chains (Fig. 11f). Notably, 23c (PBI3T) aggregated even at lower concentrations (3 µM) due to extended π-conjugation and stacking. Spectroscopic analyses further revealed H-type exciton coupling (hypsochromic shifts) in the TAPOT side chains due to cofacial stacking and J-type exciton coupling (bathochromic shifts) in the PBI core, suggesting a slip-stacked arrangement. Solution-sheared films showed orientation-dependent photoconductivity where perpendicular alignment enhanced photocurrents (20, 45, and 14 nA) (Fig. 11c), while parallel alignment suppressed them (3, 0.5, and 2 nA) as schematically shown in Fig. 11e.74 Aligning charge-transport domains could boost photoresponse up to 90-fold in 23b (PBI2T). TA spectroscopy revealed a charge separation time constant of 1.6 ps for 23c (PBI3T), with a charge-separated state lasting for 768 ps, almost five times higher than that obtained for 23b (PBI2T) (161 ps), attributed to larger supramolecular domains in the former. Despite better exciton dynamics, 23c (PBI3T) underperformed in macroscopic photoconductivity, highlighting that molecular electronic properties alone do not determine device efficiency rather the film quality and processability also play decisive roles.
 | ||
| Fig. 11 (a) Chemical structures of 23a–c. (b) Illustration of the self-assembly of PBIs into D–A core–shell columnar structures. (c) Plot of the photoresponse of two-contact devices with the solution-sheared active layer of 23a (orange), 23b (green), and 23c (purple). The solid and dashed lines represent devices with the columnar long axis oriented perpendicular and parallel to the electrodes. The samples were illuminated with white light. (d) Concentration-dependent UV/vis absorption spectra of 23a in toluene at 298 K. (e) Schematic representation of a self-assembled seven-stranded 161 helix segment of 23c, illustrating the charge-separation and charge-transport processes upon light illumination. (f) Plot of ∝aggvs. concentration for 23a (orange squares), 23b (purple dots) and 23c (green triangles) along with the corresponding fits. Fig. 11 is adapted with permission from ref. 74. Copyright 2020, American Chemical Society. | ||
Valiyaveettil and co-workers (2020) synthesized various azo-functionalized PBI (azo-PBI) dyads to tune their optical, electronic, and self-assembly properties.119 They synthesized two types of dyads: conjugated dyads, 27, 28 and 29, where azo units were directly linked to the PBI, and non-conjugated dyads, 24, 25 and 26, where azo units were separated by spacers (Fig. 12a). The self-assembled morphologies, formed through slow solvent exchange, varied significantly: non-conjugated azo-PBI dyads 24–26 formed flexible fibrous structures (Fig. 12b), while conjugated dyads 27–29 formed more rigid assemblies such as whiskers, needles, and spherical particles (Fig. 12c).119 Optical studies of conjugated dyads in CHCl3 showed red-shifted, broadened absorption and emission spectra due to extended π-conjugation (Fig. 12e), while non-conjugated dyads exhibited more structured features (Fig. 12d).119 Dyad 26 displayed a large Stokes shift caused by aggregation while CT bands appeared for derivatives with amine substitution. The morphologies correlated with molecular planarity and steric effects and UV light-induced isomerization caused morphological changes, demonstrating their light-responsive behaviour. Additionally, amine-containing azo-PBIs (26 and 29) served as mild reductants for in situ gold nanoparticle formation.
 | ||
| Fig. 12 (a) Structures of azo-PBIs (24–29). (b) Self-assembled morphology of 24 and (c) 27 obtained from solvent-induced slow precipitation on a glass slide visualized by field-effect SEM. The scale bar is 1 µm. (d) UV/vis absorption spectra of 24–26 and (e) 27–29 in CHCl3. (f) Chemical structures of 30 and 31 (AzPBIs). (g) UV/vis spectra of 30 (1AzPBI) in different solvents. (h) Distinct aggregate colour in solution and solid states. Fig. 12b–e are adapted with permission from ref. 119. Copyright 2020, American Chemical Society. Fig. 12g and h are adapted with permission from ref. 120. Copyright 2022, Wiley-VCH GmbH. | ||
Chaudhuri and co-workers (2022) demonstrated reversible supramolecular polymorphism in PBI–trans-azobenzene dyads by tuning side-group conformations through solvent–solute hydrogen bonding rather than solvent exchange or pathway control.120 They synthesized two bay-substituted PBIs bearing trans-azobenzene units, 30 (1AzPBI) and 31 (2AzPBI), functionalized with one and two azobenzene groups, respectively (Fig. 12f). For 30 (1AzPBI), solvent polarity dictated three distinct aggregate forms: in dioxane, it remained molecularly dissolved with an S0–S1 absorption at 544 nm; in water, H-aggregates were formed; and in MeOH, a broad red-shifted absorption band at 668 nm appeared, with diminished S0–S1 intensity and a residual 427 nm band (Fig. 12g and h). In dimethylsulfoxide (DMSO), two new structured bands at 432 and 755 nm emerged, indicating strong interchromophoric coupling. The fluorescence decreased by 3–4 orders of magnitude in MeOH and DMSO aggregates and was completely quenched in water. All three aggregates were isolable in the solid state, exhibiting distinct morphologies: rice-grain-like hollow tubes in DMSO, flat blade-like sheets in water (15–60 nm thick), long filamentous structures in MeOH and aggregates (an average aggregate size of 70 nm in DMSO, 630 nm in MeOH, 115 nm in water) as confirmed by dynamic light scattering (DLS). Selected area electron diffraction (SAED) and XRD analyses revealed polymorph-specific diffraction patterns and d-spacings, confirming distinct molecular packing. Compound 31 (2AzPBI) displayed analogous solvent-dependent aggregation and solid-state isolability. Control dye 2BrPBI (without azobenzene) showed only H-aggregation, underscoring the critical role of azo units in directing polymorphism. Temperature-, solvent-, and pH-dependent studies revealed that reversible hydrogen bonding between protic solvents and azo-nitrogen atoms regulated π–π* transitions, aggregation cooperativity, and polymorph interconversion.120 This side-group conformation-driven approach provided a robust design principle to achieve reversible, stimuli-responsive supramolecular polymorphs starting from straightforward molecular design.
Hirsch and co-workers (2024) synthesized a series of highly water-soluble bola-amphiphilic zinc porphyrin–PBI triads 32, 33 and 34, differing in bay substitution in the PBI core and generation of the dendron units (Fig. 13a).80 Each triad featured a central zinc porphyrin linked to a PBI unit flanked by Newkome-type dendrons of either G1 (three carboxylic acids) or G2 (nine carboxylic acids) generations. Density functional theory (DFT) calculations provided insight into the electronic structure and geometry of the chromophore triads. Triads 32 and 33 consisted of twisted PBI cores where porphyrins were co-planar to the respective side of the PBI. While the PBI and porphyrins cores were both co-planar for triad 34. To investigate the aggregation behaviour of these triads in aqueous media, UV/vis absorption and fluorescence spectroscopy was performed in binary solvent mixtures of 0.1 M sodium hydroxide (NaOH) and tetrahydrofuran (THF). The gradual addition of THF altered the polarity of the medium and facilitated disaggregation of the assemblies. For triad 32, a progressive blue-shift and sharpening of the Soret-band to 426 nm was observed with increasing THF content (0–58 vol%), indicating disaggregation. The peaks at 561 nm and 602 nm corresponding to the Q-band and PBI respectively shifted to 558 nm and 598 nm, after addition of 32 vol% THF, with an increase in absorbance. After the addition of 50 vol% THF, the absorbance at 598 nm decreased, while that at 558 nm increased with a shoulder at 520 nm becoming more prominent. Triad 33 exhibited a slight bathochromic shift and sharpening of the Soret-band at 426 nm with increasing THF content (10–66 vol%), along with increased absorbance. At 33 vol% THF, a notable increase in absorbance was observed in the overlapping Q-band, PBI bands at 558 nm and 598 nm. At 66 vol% THF, the absorbance at 520 nm and 558 nm increased further, while the absorbance at 598 nm decreased. Triad 34 exhibited THF-dependent disaggregation behaviour in absorbance, where upon increasing THF content up to 50 vol%, a blue shift of the Soret-band (426 nm) of porphyrins was observed, accompanied by sharpening and significant changes in the PBI region. New bands appeared at 479 nm and 514 nm at 33 vol% THF, with further shifts and intensity variations at higher THF concentrations. Steady-state fluorescence studies of triads 32 and 34 (excited at 426 nm) revealed increased emission intensities upon addition of 30 vol% THF, showing characteristic peaks at 607 and 652 nm for triad 32 and 606 and 653 nm for triad 34. Similarly, triad 33 displayed re-emergent fluorescence signals at 606 and 652 nm up to 66 vol% THF.80 These observations indicate that THF addition improves solvation of the hydrophobic interior, leading to disaggregation of supramolecular assemblies (Fig. 13c).
 | ||
| Fig. 13 (a) Molecular structures of the amphiphilic triads (32–34). (b) Molecular structures of the amphiphilic dyads 35–37. (c) Possible aggregation model of amphiphilic triads (32–34). UV/vis absorption spectra of (d) 35, (e) 36, and (f) 37 in 10 mM aqueous NaOH. Fig. 13c is adapted with permission from ref. 80. Copyright 2025, Wiley-VCH. Fig. 13d–f are adapted with permission from ref. 79. Copyright 2024, Wiley-VCH. | ||
The same groups (2025) reported the design, synthesis, and characterization of a series of D–A dyads 35, 36 and 37 composed of a zinc-porphyrin donor and a PBI acceptor unit (Fig. 13b).79 These dyads were amphiphilic, incorporating Newkome-type dendritic hydrophilic groups on the porphyrin side and branched hydrophobic alkyl chains on the PBI, rendering them water-soluble and capable of self-assembling into supramolecular architectures in aqueous media. The aggregation behaviour and solvent-driven disassembly of the dyads were investigated through UV/vis absorption and fluorescence spectroscopy in mixtures of THF and 10 mM aqueous NaOH. In pure aqueous solution, all three dyads showed broad, red-shifted absorption with loss of vibronic signatures. Upon increasing THF content (up to 50 vol%), the spectra became sharper and blue-shifted, consistent with progressive disaggregation. Dyad 36 exhibited a non-linear disassembly profile, with a distinct intermediate state at 25% THF, suggesting reorganization between aggregated and monomeric forms (Fig. 13d). Dyad 36 showed moderate red shifts in aqueous media and gradual spectral sharpening upon THF addition, reflecting reduced aggregation (Fig. 13e). Dyad 37, lacking bulky mesityl groups, showed the strongest aggregation effects, including Soret band splitting and complete loss of the PBI (2,0) band in water while these features were recovered with increasing THF (Fig. 13f).79 The close porphyrin–porphyrin proximity in dyad 37 allowed strong electronic coupling, pointing to offset stacking and distinct aggregate geometries. Fluorescence spectroscopy supported these findings, revealing strong quenching in water and emission recovery upon THF addition, confirming the dynamic nature of aggregation and its solvent-dependent assembly–disassembly.
Scanning transmission electron microscopy (STEM) imaging revealed vesicle-like assemblies for dyad 35 and micelle-like particles for 36 and 37 dyads. DLS analysis supported the co-existence of small micelles and larger aggregates, depending on molecular structure and concentration. Overall, this study demonstrated how structural tuning in amphiphilic porphyrin-PBI dyads enabled control over their self-assembly and optical behaviour in water, with potential applications in LH and sensing and as supramolecular materials.
2.2. Foldameric and oligomeric heterochromophoric PBI assemblies
Donor–acceptor foldamers and oligomers serve as unique model systems to systematically understand the effect of intramolecular interactions and charge/energy transport behaviour on the overall (opto)electronic properties of supramolecular assemblies/polymers, thereby providing insights into the how these properties scale up from monomers to their assemblies.121A few representative examples of PBI heterochromophoric foldamers inherently possessing the p–n heterojunction type (D–A) structures and their aggregation are presented, which were designed and investigated for their potential roles in OSCs as well as in (supra)molecular electronics. Würthner, Kim and co-workers (2015) investigated foldamer systems composed of two PBI dyes covalently attached to conjugated backbones of 1,2-bis(phenylethynyl)benzene and phenylethynyl-bis(phenylene)indane (Fig. 14a and d).122 They synthesized three systems 38–40 and examined their solvent-dependent optical and photophysical properties. The UV/vis absorption spectra of the PBIs were recorded in CHCl3 and THF. Compound 38 exhibited monomeric behaviour in both solvents. In contrast, foldamers 39 and 40 remained monomeric in CHCl3, showing 0–0 to 0–1 vibronic band intensity ratios (A0–0/A0–1) of 1.28 and 1.16, respectively, compared to the monomeric value of ∼1.65, indicating weak excitonic coupling. In THF, however, 39 and 40 displayed reduced interchromophoric distances and stronger excitonic coupling, with A0–0/A0–1 ratios of 0.81 and 0.70, respectively. The steady-state fluorescence spectra of PBI foldamers 39 and 40 revealed distinct solvent-dependent conformations. In CHCl3, the fluorescence spectra mirrored the absorption profiles, indicating weak excitonic coupling and predominantly open conformations. In contrast, THF induced a red-shifted excimer emission, which was more pronounced for 40, reflecting stronger excited-state interactions due to co-facial π–π stacking. Notably, while 39 showed reduced emission efficiency in THF, 40 maintained similar fluorescence quantum yields in both solvents, attributed to an additional PET pathway between the conjugated backbone and the PBI chromophore. Time-resolved fluorescence and TA spectroscopy revealed two distinct solvent-dependent relaxation pathways observed for the PBI foldamers 39 and 40. In CHCl3, PET led to the formation of charge-separated states with time constants of 1.5 ps for 39 and 0.7 ps for 40 and their charge recombination times were 30–50 ps, respectively. In THF, the excited-state absorption band of 40 indicated the formation of excimer states, characterized by a gradual red-shifted emission centered around 700 nm.122 Redox data from CV and Rehm–Weller analysis could relate these photophysical processes to fundamental mechanisms in OSCs, emphasizing that suppressing excimer formation is crucial for efficient exciton diffusion and charge carrier generation.
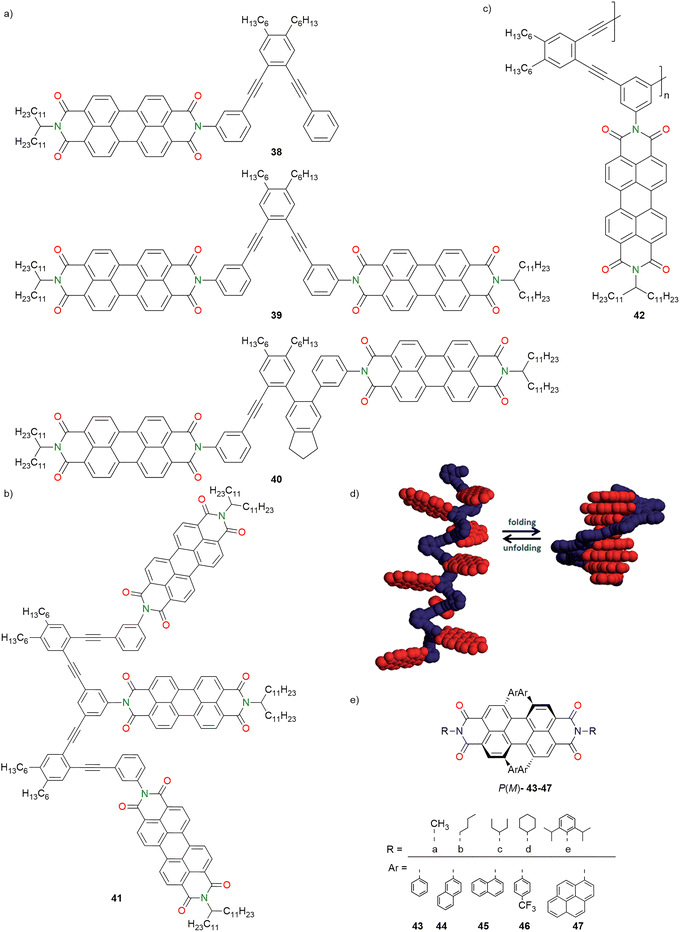 | ||
| Fig. 14 (a–c) Chemical structures of solvent-responsive PBI foldamers 38–42. (d) Schematic representation depicting folding-dependent equilibrium of PBI foldamers 38–40. (e) Chemical structures of PBI non-fullerene acceptors (NFAs) 43–47. Fig. 14d is adapted with permission from ref. 122. Copyright 2015, The Royal Society of Chemistry. | ||
Würthner, Kim, Engels and co-workers (2015) reported phenylene ethynylene tethered PBI foldamers with two (folda-dimer 39) and three (folda-trimer 41) PBI units (Fig. 14b).121 The folding behaviour was investigated using UV/vis absorption spectroscopy. The A0–0/A0–1 absorbance ratio of 39 was 1.3 in CHCl3 and 0.8 in THF, indicating an unfolded conformation in CHCl3 and a folded state stabilized by π–π interactions. Similar solvent-dependent trends were observed for folda-trimer 41. Temperature- and concentration-dependent UV/vis absorption studies showed negligible spectral variations for both foldamers 39 and 41, indicating the absence of further intermolecular self-assembly and confirming strong intramolecular π–π interactions stabilizing their folded structures. Temperature-dependent 1H NMR studies in deuterated THF further revealed intramolecular dynamic processes in both foldamers, attributed to restricted imide bond rotation and backbone flapping at elevated temperatures. Steady-state and time-resolved fluorescence along with TA studies revealed that optical excitation of PBI foldamers induced solvent-dependent conformational relaxation, leading to competing pathways of PET in the extended form and excimer formation in the folded state. The pronounced excimer emission, reduced quantum yields, and long excited-state lifetimes (20–26 ns) in THF confirmed efficient folding and strong π–π interactions, whereas faster PET and shorter lifetimes dominated in the more extended conformations in CHCl3.121
Followed by the studies of folda-dimer and trimer systems, the same groups developed similar solvent-responsive PBI foldamers incorporating a phenylene ethynylene backbone, wherein the octameric foldamer (42) (Fig. 14c) exhibited tunable folding behaviour analogous to the dimer (39).123 Solvent-induced transformation from an extended to a compact helical form was accompanied by a change in the A0–0/A0–1 absorbance ratio (1.65 for 38 (monomer); 0.78 for 39 in THF; 0.88 for 42 in MCH), indicative of H-type π–π stacking. Compounds 39 and 42 displayed broad excimer emission at 625 nm and decreased fluorescence quantum yields due to PET from the electron-rich oligo phenylene–ethynylene backbone to the PBI units. Excitation anisotropy and time-correlated single-photon counting studies revealed solvent-dependent rotational diffusion and ultrafast depolarization arising from intramolecular energy migration between PBI chromophores. Femtosecond TA and TA anisotropy (TAA) analyses further confirmed singlet–singlet annihilation and exciton hopping, while comparative visible-NIR TA spectra demonstrated efficient PET in the extended form but its suppression in the folded helical conformation.123 These findings highlight the importance of foldable oligomeric aggregates like 42 as model systems bridging isolated molecules and bulk materials for understanding exciton and CT processes in organic optoelectronics and supramolecular electronics.
Würthner and co-workers (2015) systematically investigated a series of twisted PBI dyes by varying different imide and aryl substituents in 1, 6, 7, and 12 at bay positions inducing conformationally stable axially chiral P(M) compounds 43–47 as NFAs in BHJ solar cells with a thiophene based donor polymer (Fig. 14e).124 By varying both imide (43a–43e) and aryl (43b–47b) substituents, they identified 2,6-diisopropylphenyl (imide) and 2-naphthyl (aryl) as the optimal combination, yielding derivative 44e, which achieved a record power conversion efficiency (PCE) of 4.3%, the highest reported efficiency in the BHJ solar cell for a single-core PBI lacking lateral π-extension or heteroatom incorporation. Structural analysis revealed that isolated chromophores with minimal π–π interactions (e.g., 43e and 44e) exhibited superior device performance. In contrast, PBIs with smaller, flexible imide substituents (e.g., n-butyl in 43b) exhibited enantiomer-dependent aggregation, resulting in the formation of slip-stacked structures that were detrimental to device efficiency. Comparison of solution (in DCM) and thin-film spectra revealed that both 43b and 43e retained their broad, featureless absorption profiles with minimal spectral shifts, indicating comparable molecular arrangements in solution and solid states. However, their CD spectra revealed notable differences: enantiopure P/M-43b showed a pronounced enhancement of the S0–S1 transition in thin films, attributed to helically twisted, π-stacked dimers observed in its crystal structure. In contrast, 43e exhibited only isolated chromophores without supramolecular interactions. Consequently, isolated and sterically shielded PBIs proved to be robust NFA candidates, irrespective of enantiopurity.124 This study also highlighted future molecular design directions conducive for supramolecular optoelectronics, including exploiting core twisting for multichromophoric NFAs.
2.3. Role of PBI heterochromophoric monomers, oligomers and assemblies modified for organic and supramolecular electronics
Fullerenes have served as conventional acceptors for OSCs for very long owing to their inherent electron deficiency and propensity to form well-defined domain separation, resulting in high performance in OSCs. However, their optical and electronic properties cannot be tuned easily by chemical derivatisation. On the other hand, PBIs, in addition to their unique photophysical, redox (long lived excited singlet state, high photostability, and stable radical dianions) and electronic properties, can be structurally tailored to achieve tunable optoelectronic properties.125 In fact, PBI dimers and oligomers have found profound applications in OSCs since the last decade.126 However, the PCE of conventional monomeric PBIs does not exceed 5% owing to their strong propensity of aggregate. To overcome this challenge, core annulation and core twisting of PBIs have been adopted as promising strategies to ensure optimal domain sizes and efficient charge transport behaviour in thin films, resulting in high performance organic photovoltaic and electronic devices.125Some of the best PBI candidates in this genre have been reported by Wang127,128 and Nuckolls129,130 groups, where they have shown that molecular rigidity is of paramount importance for high performance OSCs. Wang and co-workers pioneered the synthesis and characterization of bay-linked PBIs with heteroatom bridges containing S and Se atoms to achieve laterally extended PBIs, which led to higher LUMO levels and therefore increased open circuit voltages,127,131 PBI dimers and polymers132 and giant rylene diimides as electron acceptors for OSCs and n-channel semiconductors.128 They further reported twisted propeller PBIs, PBIs fused with central phenyl rings and ones connected by single bonds.128 The noteworthy aspect of the design was the rigidified structures that led to defined molecular ordering, high domain purity and eventually to high electron mobilities in the blends for OSCs.
Nuckolls and co-workers spearheaded the design of contorted polycyclic aromatics including linear oligomeric PBI ribbon like structures where PBI cores were fused with vinylene bridges. These atomically precise nanoribbons exhibited an exceptional PCE of up to 8% in solar cells without any optimization.133 Furthermore, they also reported intricate structures such as PBI helicenes,127,134 circulenes,135 twistacenes,26,99,100 and chiral conjugated and macrocyclic structures136,137 as highly efficient acceptors for organic photodetectors and helicene nanoribbons as exceptional acceptors. Over the last few years, this group developed solution processable and ambient-stable ultrahigh performance graphene nanoribbons138 leveraging upon the synthetic tunability of PBIs. Such nanoribbon architectures have shown tremendous potential towards advanced nanoelectronic applications.138
Furthermore, three-dimensional multichromophoric arrays139,140 by virtue of their reduced aggregation also led to appropriate domain sizes facile for efficient charge transport in devices. In recent years, further extended PBIs such as terylene diimides141 and quarterrylene diimides22 showed appreciable PCEs in OSCs. In many of these cases, the control of molecular packing arrangement was achieved by pre-programmed supramolecular engineering of individual molecules.
Table 1 summarizes all the examples of covalently connected heterochromophoric PBI compounds and the details of the optical and morphological properties of their self-assembly and applications and future potential for supramolecular electronics.
| Types of chromophores | λ abs (nm) | λ em (nm) | Types of aggregates (mode of interaction) | Morphology | Applications/future potential | Ref. |
|---|---|---|---|---|---|---|
| PBI and OPV 1, 2a-c (n = 0–2) and 3 | 300–650 for 1, 2a-c and 603 for 3 | 475–650 | J-type aggregates (π–π interactions and H-bonding) | 2b showed thicker and right-handed helical rod-like fibres | Photovoltaic device; an open circuit voltage of 0.74 V and a current density of 2.6 µA cm−2 | 110 |
| PBI and OPV (4–6) | 300–600 | — | 4 and 6 form J-aggregates and 5 forms H-aggregates (π–π interactions) | — | — | 111 |
| HBC and PBI (7–11) | HBC core (325–450) and PBI (450–600) | 520–620 | H-aggregates (π–π interactions) | — | Potential thin film electronic devices | 112 |
| Anthracene and PBI (12) | ∼502 and ∼545 (PBI) 355, 372, and 392 (anthracene) | — | π–π interactions | — | — | 81 |
| Benzothiodiazole and PBI (13, 14a–b) | ∼525, 490, and 460 | — | π–π interactions | 13a,14a-b formed fibres and 13b formed nanoparticles | Potential photovoltaic device performance | 113 |
| Pyrene and PBI (15) | 524 (solid film) | 707 (solid film) | H-aggregates (π–π interactions) | Nanobelt | I–V measurements (an Ion/Ioff ratio of ∼300) | 114 |
| Triphenylene and PBI (16, 17) | 16 (400–700) | 16 (500–700) | π–π interactions | — | Potential photovoltaics | 115 |
| Bay substituted and unsubstituted PBI (18–20) | 20 (460–586) | — | π–π interactions | — | — | 116 |
| Quaterthiophene and PBI (21a–b) | 558, 467 and 440 | — | H- and J-aggregates (π–π interactions) | — | Potential organic photovoltaics | 117 |
| Thiophene and PBI (22a–b) | 631 | — | J-aggregates (π–π interactions) | — | Photoconductivity measurements (22b showed 20-fold higher photoresponse than 22a) | 118 |
| Thiophene and PBI (23a-c) | 320–750 | 600–900 | J- aggregates (π–π interactions) | — | Photoconductivity measurements (23b showed 90-fold higher photoresponse) | 74 |
| Azobenzene and PBI (24–29) | 535, 538 and 548 (24–26); 558, 562 and 570 (27–29) | 565, 571 and 582 (24–26); 608, 622 and 628 (27–29) | — | 24–26 form fibres, 27–29 form nanoparticles | Potential photoelectronic devices | 119 |
| Azobenzene and PBI (30 and 31) | 300–900 | 400–900 (DMSO, MeOH); non-emissive (water) | H-aggregates | Rice-grain like structure with a tapered end (DMSO), long filamentous structures (MeOH), rigid and flat blade-like structures (water) | Potential stimuli-responsive materials | 120 |
| Zinc-porphyrin and PBI (32–34) | 400–650 | 500–750 | π–π interactions | — | — | 80 |
| Zinc-porphyrin and PBI (35–37) | 400–650 | 500–750 | — | Large spheres for 35 and small, large spherical particles for 36 and 37 | — | 79 |
| Phenylene ethynylene and PBI (38–40) | 400–600 | 533, 575, 615 (39); 533, 531, 577, 617 (40) | Folded state in THF and unfolded in CHCl3 (π–π interactions) | — | Potential organic photovoltaics | 122 |
| Phenylene ethynylene and PBI (39 and 41) | 493, 526 (39); 495, 534 (41) | 533, 575, 615 (39); 532, 617 (41) | Folded state in THF and unfolded in CHCl3 (π–π interactions | — | — | 121 |
| Phenylene ethynylene and PBI (39, 42) | 400–600 | 625 | Foldable oligomer aggregates (π–π interactions) | — | Potential organic solar cells | 123 |
| Naphthalene, pyrene and PBI (43–47) | 400–650 | — | — | — | Organic solar cells | 124 |
2.4. Non-covalent heterochromophoric PBI assemblies
Self-assembly of PBIs is predominantly governed by π–π stacking and furthermore the directionality of the aggregation is provided by other non-covalent interactions such as hydrogen bonding, solvophobic effects, electrostatic forces and CT interactions.36,142,143 These synergistic interactions result in formation of well-defined hierarchical superstructures such as homo- or hetero-assemblies as summarized in Fig. 15. Such interactions can be introduced through suitable substituents in the core or the sidechains of the PBI molecules and play a pivotal role in organization of molecular architectures via self-assembly or co-assembly. Furthermore, π–π stacking governs electronic communication, while hydrogen bonding directs spatial organization further dictating the aggregation mode. These interactions are advantageous as they enhance charge transport, long exciton migration and easy tunability of optoelectronic properties.36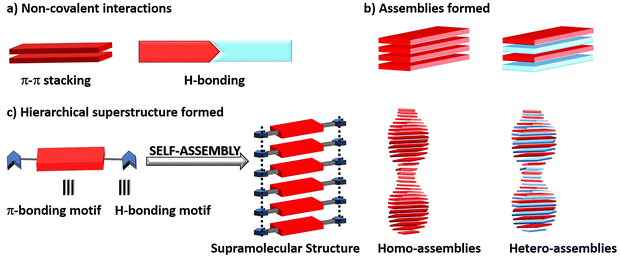 | ||
| Fig. 15 Schematic illustration of architectures formed through various (a) non-covalent interactions leading to (b) assemblies and eventually giving rise to (c) hierarchical superstructures. | ||
Schenning, Meijer and co-workers (2004) synthesized four PBI derivatives (PBI 1–PBI 4), some of which showed LC behaviour (Fig. 16a).144 Temperature- and concentration-dependent UV/vis absorption and CD spectroscopy in MCH revealed J-aggregation for PBI 1 and PBI 2, indicating ordered π–π stacking. PBI 1 exhibited complete fluorescence quenching at room temperature up to 80 °C, while PBI 2 showed fluorescence recovery and CD activity at elevated temperatures, suggesting hydrogen-bonded dimer formation for the latter. PBI 3 and PBI 4 displayed minimal CD signals, indicating weak stacking, likely due to poor solubility. Aggregation of PBI 1 followed an isodesmic model with a binding constant of (13.0 ± 0.5) × 103 M−1 which was lower than that for a more rigid analogue of PBI that lacked a methylene spacer. PBI 1, showing the most robust stacking, was further studied in the solid state and in orthogonal co-assembly with p-type OPV. AFM of MCH-cast films revealed highly entangled fibrous networks of PBI 1. In MCH, mixtures of PBI 1 and OPV (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (48) formed phase-separated stacks, confirmed by UV/vis (Fig. 16b), with a melting point (∼44 °C) similar to pure PBI 1 (∼43 °C).144 Films cast from MCH showed ordered morphology, while those from toluene did not. Although fluorescence quenching and TA confirmed efficient OPV to PBI 1 electron transfer, solar cell devices exhibited poor performance, likely due to unfavourable lateral alignment of the aggregates. Based on this example, it was inferred that vertical alignment is an important criterion to enhance the device performance.
1) (48) formed phase-separated stacks, confirmed by UV/vis (Fig. 16b), with a melting point (∼44 °C) similar to pure PBI 1 (∼43 °C).144 Films cast from MCH showed ordered morphology, while those from toluene did not. Although fluorescence quenching and TA confirmed efficient OPV to PBI 1 electron transfer, solar cell devices exhibited poor performance, likely due to unfavourable lateral alignment of the aggregates. Based on this example, it was inferred that vertical alignment is an important criterion to enhance the device performance.
 | ||
| Fig. 16 (a) Chemical structures of PBI 1–4 and OPV. (b) UV/vis spectral comparison of PBI and OPV in MCH and toluene at room temperature for both separate and mixed samples (48) at a concentration of 1 × 10−5 M. Fig. 16b is adapted and modified with permission from ref. 144. Copyright 2004, American Chemical Society. | ||
Schenning, Meijer, De Feyter and co-workers (2006) reported a supramolecular system 49 (OPV4UT-PBI) consisting of OPV and PBI covalently connected using ureido-s-triazine linkage (Fig. 17a).1451H NMR revealed the dimerization of 49 (OPV4UT-PBI) through quadruple hydrogen bonding in CHCl3. Self-assembly of 49 at the liquid/solid interface on highly oriented pyrolytic graphite (HOPG) revealed hydrogen-bonded OPV dimers with PBI units adsorbed in the inter-dimer gaps, as confirmed by STM imaging and bias-dependent contrast reversal (Fig. 17b).
 | ||
| Fig. 17 (a) Chemical structure of 49 (OPV4UT-PBI). (b) STM image depicting a monolayer of compound 49 (OPV4UT-PBI) physiosorbed on a HOPG surface from a concentrated 1,2,4-trichlorobenzene solution. The arrows highlight vacancy defects within the 2D lattice.145 (c) Proposed structures of 50 representing co-assemblies of (OPV4)2M and PBI. Fig. 17b is adapted with permission from ref. 145. Copyright 2006, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
The study highlighted the distinct adsorption affinities of OPV and PBI moieties, with PBI exhibiting weaker surface binding and higher mobility, which limited the monolayer stability. Optical studies in CHCl3 showed that excitation of 49 (OPV4UT-PBI) at 440 nm (OPV absorption) quenched OPV fluorescence at 536 nm with strong PBI emission at 580 nm indicative of energy transfer and excluding the possibility of electron transfer due to the large distance between the two chromophores. In apolar dodecane, PBIs self-assembled into H-type aggregates, evident by hypsochromically shifted absorption (λmax = 508 nm) and quenched fluorescence (λem = 566 nm). Surprisingly, CD spectroscopy showed no Cotton effect at any concentration, indicating achiral self-assembly, attributed to the steric hindrance of bulky PBI units and conformational flexibility of the pentyl spacer. Temperature-dependent fluorescence studies revealed reversible disassembly of aggregates into molecularly dissolved monomers and hydrogen-bonded dimers above ∼45 °C, restoring PBI emission. Femtosecond photoinduced absorption measurements confirmed the absence of electron transfer, with quenching attributed solely to PBI aggregation. In mixed assemblies upon heterodimer formation of 49 (OPV4UT-PBI) with OPVnUT where n = 0 and 1, strong OPV fluorescence quenching and PBI emission occurred in CHCl3via energy transfer, whereas in dodecane, incorporation into OPV aggregates enhanced the quenching efficiency but reduced the PBI luminescence due to aggregation.145
The same groups (2008) reported a bifunctional system 50 comprised of melamine-functionalized OPV units ((OPV4)2M) (Fig. 17c), capable of binding two PBI moieties via complementary hydrogen bonding.146 STM imaging on graphite revealed well-organized cyclic and linear donor/acceptor (D/A) architectures, with ring-like assemblies representing densely packed chromophore ensembles. In CHCl3, ((OPV4)2M) exhibited absorption at 436 nm and strong fluorescence at 502 nm. In contrast, in apolar MCH at higher concentrations (7.5 × 10−5 M), blue-shifted absorption at 419 nm and quenched, red-shifted emission at 522 and 547 nm indicated OPV aggregation into H-type assemblies.
UV/vis absorption and CD studies revealed aggregation above a concentration of 10−5 M, with Cotton effects in CD suggesting chirality-driven self-assembly, though exciton coupling remained weak. Temperature-dependent UV/vis absorption, PL, and CD measurements confirmed reversible thermal disassembly, with spectra above ∼24 °C resembling those in CHCl3, indicative of weak interactions and an intermediate dissociation state. Mixing with PBI yielded J-type aggregates, supported by UV/Vis data while fluorescence titration revealed strong 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complexation with near-complete emission quenching via photoinduced electron transfer, while emission recovery at non-stoichiometric ratios reflected partial disassembly.146 The observed structural and compositional disorders were attributed to inherent flexibility and lack of pre-organization in OPV and PBI units, highlighting the necessity of pre-organized building blocks for achieving well-defined multicomponent supramolecular architectures.
1 complexation with near-complete emission quenching via photoinduced electron transfer, while emission recovery at non-stoichiometric ratios reflected partial disassembly.146 The observed structural and compositional disorders were attributed to inherent flexibility and lack of pre-organization in OPV and PBI units, highlighting the necessity of pre-organized building blocks for achieving well-defined multicomponent supramolecular architectures.
Shinkai and co-workers (2008) investigated the self-sorting behaviour of a supramolecular network 51 (4Thio/Pery) formed by two structurally similar self-assembling components—Thio (oligothiophene-based, n = 4) and PBI (perylene-based)—in chlorobenzene (CB) (Fig. 18a and b).147 Despite their resemblance, Thio possessed four hydrogen-bonding sites and showed much stronger aggregation (Kagg > 105 M−1) compared to PBI (Kagg = 1.3 × 103 M−1). UV/vis absorption spectra of the gels showed blue-shifted absorption bands versus monomers, indicating H-aggregation in tetrachloroethane solution. Upon heating, PBI dissociated at ∼25 °C, while Thio remained stable up to ∼95 °C. When both gelators were mixed and heated in CB, a mixed organogel was formed. The absorption spectra appeared as a linear sum of the individual components, with no new CT bands, suggesting electronically independent, self-sorted assembly. Temperature-dependent UV/vis absorption further confirmed this behaviour where PBI exhibited peak sharpening upon cooling, while Thio showed a red shift. The dissociation temperatures in the mixed system closely matched those observed in the individual gels, reinforcing the self-sorting nature of the assembly. This behaviour was also retained during the cooling process. Self-sorting was further evident from CD spectroscopy and SEM revealed fibrous morphologies for both pure and mixed systems (Fig. 18c).147 A longer oligothiophene chain (Thio, n = 6) was later used with PBI to improve D/A interactions, and the resulting film exhibited visible-light-induced photoelectrical conversion.
 | ||
| Fig. 18 (a) Chemical structures of Thio and Pery. (b) Schematic representation of the self-sorting organogel formation, leading to the generation of p–n heterojunction sites. (c) SEM images of (i) Pery gel, and ii) 51 (4Thio/Pery) self-sorting gel. (d) Molecular structures of T-OPV and C-PBI. (e) Schematic representation of the non-covalent CT amphiphile 52 (T-OPV/C-PBI) and its self-assembly into 1D supramolecular architectures. (f) AFM height images of the fibres of 52 (T-OPV/C-PBI) co-assembly. (g) Transmission electron microscopy (TEM) images revealing the formation of nanotubular structures. Fig. 18c is reproduced with permission from ref. 147. Copyright 2008, American Chemical Society. Fig. 18e–g are reproduced with permission from ref. 148. Copyright 2012, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
George and co-workers (2012) reported a supramolecular alternate D/A co-assembly 52 (T-OPV/C-PBI) in water using amphiphilic T-OPV and C-PBI (Fig. 18d and e).148 T-OPV, bearing a T-shaped amphiphilic structure, formed a non-covalent amphiphilic complex with PBI through synergistic π–π stacking, electrostatic and CT interactions. Upon increasing the equimolar concentration of T-OPV and C-PBI, the solution became increasingly viscous, and at a total concentration of 3.3 mM, a dark wine-coloured gel was formed, indicative of a 1D co-assembled structure. The gel showed a CT absorption band at 700 nm and was non-fluorescent, confirming efficient CT complexation. FE-SEM, AFM, and TEM analyses revealed highly interconnected fibrous networks, while rheology demonstrated excellent mechanical strength. To investigate the molecular organization, a UV/vis titration experiment was performed by gradually adding T-OPV to a fixed concentration of C-PBI (10−4 M) and UV/vis titration showed a red-shift (14 nm) in the absorption band of OPV accompanied by a CT band formation at 700 nm, with vibronic ratio reversal (I0–1/I0–0 > 1 to <1) indicating transition from PBI homo-assembly to D/A stacks. Photophysical studies revealed complete quenching of T-OPV and C-PBI emission due to CT interactions, confirmed by Job's plot and NMR titration (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry). Morphological studies (TEM and AFM) revealed dense, cross-linked fibrous networks with tubular morphologies, indicative of well-ordered supramolecular nanostructures (Fig. 18f and g).148 The nanotubular assemblies exhibited a high conductivity of 0.02 S cm−1, the value being the highest for undoped supramolecular systems at that time, highlighting their promise in supramolecular electronics. Therefore, this study exemplifies efficient co-facial assembly of larger π-conjugated systems by a non-covalent amphiphilic design, thereby effectively avoiding phase-segregation.
1 stoichiometry). Morphological studies (TEM and AFM) revealed dense, cross-linked fibrous networks with tubular morphologies, indicative of well-ordered supramolecular nanostructures (Fig. 18f and g).148 The nanotubular assemblies exhibited a high conductivity of 0.02 S cm−1, the value being the highest for undoped supramolecular systems at that time, highlighting their promise in supramolecular electronics. Therefore, this study exemplifies efficient co-facial assembly of larger π-conjugated systems by a non-covalent amphiphilic design, thereby effectively avoiding phase-segregation.
Fukuzumi and co-workers (2012) reported the aqueous self-assembly of a D/A system, 53 (TAIPBI–ZnTPPSK4), composed of N,N′-di(2-(trimethylammonium iodide)ethyl)PBI (TAIPBI) and meso-tetrakis(4-sulfonatophenyl)porphyrin zinc tetrapotassium (ZnTPPSK4) salt (Fig. 19a).149 In MeOH, TAIPBI exhibited monomeric vibronic transitions at 429, 457, 486 and 522, whereas in water it formed π-stacked aggregates with red-shifted absorption bands at 501 and 537 nm, indicative of co-facial stacking. Aggregation was enhanced at higher concentration and lower temperature, leading to stronger excitonic coupling and quenched fluorescence. Radical anion species of TAIPBI stacks exhibited broad, red-shifted absorption compared to monomeric PBI radical anions, suggesting extensive electronic delocalization. DLS revealed aggregates of size 105 nm to 460 nm (an average hydrodynamic diameter of 188 nm) while SEM confirmed 1D columnar morphologies.
 | ||
| Fig. 19 (a) Chemical structures of TAIPBI and ZnTPPSK4. (b) Schematic representation of charge recombination and charge separation between TAIPBI stacks and ZnTPPSK4. (c) Spectral changes during the UV/vis titration of TAIPBI with ZnTPPSK4 in water. Fig. 19c is reproduced and modified with permission from ref. 149. Copyright 2012, American Chemical Society. | ||
The excited-state dynamics of TAIPBI aggregates were studied using TA spectroscopy that revealed markedly shorter and concentration-dependent singlet lifetimes (220–303 ps) for aqueous aggregates relative to monomers in MeOH (4.4 ns). Furthermore, mixing TAIPBI aggregates with ZnTPPSK4 yielded supramolecular complexes (Fig. 19b), evident from UV/vis absorption, fluorescence, and DLS measurements. Spectral changes include the red-shift of TAIPBI absorption (Fig. 19c) and emergence of new bands corresponding to ZnTPPSK4, indicating strong π–π and electrostatic interactions.149 Titration studies revealed distinct stoichiometries (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 2
1 and 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 for TAIPBI: ZnTPPSK4) with high formation constants (K ∼105–106 M−1). Complexation led to large aggregate sizes and enabled long-range charge transport by photoinduced charge injection from ZnTPPSK4 into TAIPBI stacks, affording efficient charge separation (high kCS/kCR = 3000). These observations established that aqueous supramolecular assemblies of PBI and porphyrins can mimic natural photosynthetic architectures by integrating LH, charge separation, and charge transport within a single nanostructure, thereby offering key insights into the design of bioinspired supramolecular materials for electronic applications.
3 for TAIPBI: ZnTPPSK4) with high formation constants (K ∼105–106 M−1). Complexation led to large aggregate sizes and enabled long-range charge transport by photoinduced charge injection from ZnTPPSK4 into TAIPBI stacks, affording efficient charge separation (high kCS/kCR = 3000). These observations established that aqueous supramolecular assemblies of PBI and porphyrins can mimic natural photosynthetic architectures by integrating LH, charge separation, and charge transport within a single nanostructure, thereby offering key insights into the design of bioinspired supramolecular materials for electronic applications.
Braunschweig and co-workers (2013) reported the co-assembled helical supramolecular polymers 54 and 55 (diketopyrrolopyrrole (DPP)-PBI) based on (DPP 1,2) derivatives and PBI, stabilized by triple hydrogen bonding and π–π interactions. DPP1 consisted of homochiral (S)-2-methylbutyl side chains, while DPP 2 consisted of racemic 2-ethyloctyl groups (Fig. 20a).150 UV/vis absorption spectroscopy of DPP 1 revealed absorption at 538 and 580 nm at 25 °C and bathochromic shift of the bands upon cooling, consistent with J-type aggregation. Despite chiral side chains, variable-temperature CD (VT-CD) displayed no Cotton effect, suggesting isodesmic aggregation that was primarily governed by π–π stacking, further supported by slip-stacked geometry observed from X-ray crystallographic analysis. For PBI, cooling diminished the 520 nm monomer band while producing a broad band at 592 nm, indicative of rotationally displaced stacking facilitated by hydrogen bonding. Upon mixing DPP 1 and PBI in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (54), the absorption spectra at 40 °C resembled those of individual components, but cooling led to the appearance of two sharp absorption bands at 563 and 615 nm, suggesting formation of J-type hetero-assemblies that suppressed PBI homoaggregation. VT-CD revealed bisignate Cotton effects, confirming chiral hetero-assembly formation, with the CD transition temperature being 5 °C lower than that in the absorption study, implying the initial association of PBI into disordered DPP 1 aggregates followed by reorganization into heteroaggregate helices. DPP 2–PBI (55) mixtures exhibited similar spectral changes but lacked chirality.150 DFT calculations revealed a head-to-tail alignment of PBI units in the hetero-assemblies, consistent with J-aggregation. A quantitative model was developed to describe heteroaggregate formation via H-bonding and π–π stacking, and thermodynamic parameters (ΔH° and ΔS°) for each interaction were derived from temperature-dependent UV/vis and CD data. The model revealed that PBI was bound to disordered DPP stacks to form chiral superstructures (Fig. 20b), where initial H-bonding was enthalpically driven and subsequent π-stacking overcame the entropic penalties to promote heterosuperstructure formation upon cooling.150
1 ratio (54), the absorption spectra at 40 °C resembled those of individual components, but cooling led to the appearance of two sharp absorption bands at 563 and 615 nm, suggesting formation of J-type hetero-assemblies that suppressed PBI homoaggregation. VT-CD revealed bisignate Cotton effects, confirming chiral hetero-assembly formation, with the CD transition temperature being 5 °C lower than that in the absorption study, implying the initial association of PBI into disordered DPP 1 aggregates followed by reorganization into heteroaggregate helices. DPP 2–PBI (55) mixtures exhibited similar spectral changes but lacked chirality.150 DFT calculations revealed a head-to-tail alignment of PBI units in the hetero-assemblies, consistent with J-aggregation. A quantitative model was developed to describe heteroaggregate formation via H-bonding and π–π stacking, and thermodynamic parameters (ΔH° and ΔS°) for each interaction were derived from temperature-dependent UV/vis and CD data. The model revealed that PBI was bound to disordered DPP stacks to form chiral superstructures (Fig. 20b), where initial H-bonding was enthalpically driven and subsequent π-stacking overcame the entropic penalties to promote heterosuperstructure formation upon cooling.150
 | ||
| Fig. 20 (a) Chemical structures of co-assembled helical supramolecular polymers 54 and 55. (b) Proposed model illustrating the heteroaggregation of DPP (red tiles) and PBI (blue tiles) units leading to the formation of chiral supramolecular assemblies in compounds 54 and 55. (c) Scheme representing the formation of superstructures 54 upon cooling driven by H-bonding and orthogonal π–π stacking interactions and frontier molecular orbital (FMO) alignment illustrating possible photoinduced electron and hole transfer pathways by donor or acceptor excitation. Fig. 20b and c are reproduced with permission from ref. 150 and 151. Copyright 2013 and 2014, American Chemical Society. | ||
In a subsequent study, the same group (2014) explored the optical and electronic properties of 54 (DPP 1–PBI) co-assembled supramolecular polymers, focusing on their CT behaviour.151 Owing to the electronic complementarity of their FMO (Fig. 20c), electron transfer occurred from the photoexcited DPP 1 singly occupied molecular orbital (SOMO) (−3.24 eV) to the PBI LUMO (−3.63 eV) or hole transfer from the photoexcited PBI SOMO (−5.35 eV) to the DPP 1 HOMO (−5.98 eV), with respective driving forces of −0.39 eV and −0.63 eV respectively (Fig. 16d).151 VT fluorescence quenching studies on a (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) DPP 1
1) DPP 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PBI mixture revealed aggregation-induced static quenching upon cooling from 70 °C to 20 °C, which was corroborated by TA spectroscopy. At elevated temperatures, dynamic quenching led to enhanced DPP 1 fluorescence, which peaked at 40 °C and diminished upon further cooling due to the onset of static quenching. No change in absorption or DPP 1 fluorescence was observed in the absence of PBI, indicating no aggregation-induced quenching. These results indicated that fluorescence quenching arose from non-covalent heteroaggregation, corroborated by a DLS study indicating the formation of superstructures of length 1–2 µm. TA studies revealed a CT band at 622 nm, highlighting subtle D/A electronic interactions upon self-assembly.151 These findings underline the potential of such supramolecular systems in solar energy harvesting and supramolecular electronics.
PBI mixture revealed aggregation-induced static quenching upon cooling from 70 °C to 20 °C, which was corroborated by TA spectroscopy. At elevated temperatures, dynamic quenching led to enhanced DPP 1 fluorescence, which peaked at 40 °C and diminished upon further cooling due to the onset of static quenching. No change in absorption or DPP 1 fluorescence was observed in the absence of PBI, indicating no aggregation-induced quenching. These results indicated that fluorescence quenching arose from non-covalent heteroaggregation, corroborated by a DLS study indicating the formation of superstructures of length 1–2 µm. TA studies revealed a CT band at 622 nm, highlighting subtle D/A electronic interactions upon self-assembly.151 These findings underline the potential of such supramolecular systems in solar energy harvesting and supramolecular electronics.
Ajayaghosh and co-workers (2015) reported self-sorted 1D supramolecular nanostructures 56 (TTV/PBI) comprised of trithienylenevinylene (TTV, p-type) and PBI (n-type), forming coaxial p–n heterojunctions (Fig. 21a and b).76 TTV displayed an absorption maximum at 501 nm in CHCl3, which shifted hypsochromically to 464 nm in n-decane, indicative of strong H-type 1D aggregation. PBI showed sharp absorption bands at 458, 491, and 526 nm in CHCl3, with an additional shoulder at 580 nm indicative of J-type aggregation in n-decane. Upon mixing TTV and PBI in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, no spectral shifts were observed, ruling out CT complexation and suggesting independent self-assembly. The distinct self-assembly behaviour was attributed to directional hydrogen bonding in TTV and extended π–π stacking in PBI. Morphological characterization via AFM and TEM revealed different architectures: elongated fibres for TTV, sheet-like structures for PBI, and rod-like morphologies for the 56 (TTV/PBI) mixture, supporting hierarchical self-sorting (Fig. 21c and d).76 Powder XRD (PXRD) confirmed crystalline stacking for PBI and amorphous character for TTV, while the mixture showed features of both. Kelvin probe force microscopy indicated interfacial electronic interactions through a surface potential shift to −100 mV in the blend, suggesting nanoscale phase separation and interfacial interactions between oppositely charged donor and acceptor stacks. Additionally, flash photolysis time-resolved microwave conductivity (FP-TRMC) showed 12-fold enhanced photoconductivity, attributed to coaxial p–n alignment as compared with their individual assemblies. Thus, while the self-sorting driven co-assembly strategy enabled the formation of well-defined 1D p–n heterojunctions with excellent photoconductivity, the inability to achieve controlled vertical fibre alignment between electrodes remained a key challenge impeding their practical integration into devices.
1 ratio, no spectral shifts were observed, ruling out CT complexation and suggesting independent self-assembly. The distinct self-assembly behaviour was attributed to directional hydrogen bonding in TTV and extended π–π stacking in PBI. Morphological characterization via AFM and TEM revealed different architectures: elongated fibres for TTV, sheet-like structures for PBI, and rod-like morphologies for the 56 (TTV/PBI) mixture, supporting hierarchical self-sorting (Fig. 21c and d).76 Powder XRD (PXRD) confirmed crystalline stacking for PBI and amorphous character for TTV, while the mixture showed features of both. Kelvin probe force microscopy indicated interfacial electronic interactions through a surface potential shift to −100 mV in the blend, suggesting nanoscale phase separation and interfacial interactions between oppositely charged donor and acceptor stacks. Additionally, flash photolysis time-resolved microwave conductivity (FP-TRMC) showed 12-fold enhanced photoconductivity, attributed to coaxial p–n alignment as compared with their individual assemblies. Thus, while the self-sorting driven co-assembly strategy enabled the formation of well-defined 1D p–n heterojunctions with excellent photoconductivity, the inability to achieve controlled vertical fibre alignment between electrodes remained a key challenge impeding their practical integration into devices.
 | ||
Fig. 21 (a) Chemical structures of (a) TTV and PBI. (b) Schematic representation of different possible hierarchical structures in 56 (TTV/PBI). (c) AFM images of 57 (TTV/PBI) at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio, drop cast from n-decane solutions onto a silicon wafer at room temperature. (d) TEM images of 56 (TTV/PBI) at a 1 1 molar ratio, drop cast from n-decane solutions onto a silicon wafer at room temperature. (d) TEM images of 56 (TTV/PBI) at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio, drop-cast from n-decane solutions onto carbon-coated copper grids at room temperature. Fig. 21b–d are adapted with permission from ref. 76. Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. 1 molar ratio, drop-cast from n-decane solutions onto carbon-coated copper grids at room temperature. Fig. 21b–d are adapted with permission from ref. 76. Copyright 2015, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Calzaferri, Häner and co-workers (2015) demonstrated the coexistence of two independent exciton states within alternating chromophore stacks of PBI and 1,8-dibutynylpyrene (Fig. 22a and b) organized on a DNA scaffold where hybridization of complementary strands gave rise to duplex structures 57.152 Two hybrid series were designed: Series A (hybrids 1–8) to probe nearest-neighbour (NN) interactions and Series B (hybrids 9–16) for next-nearest-neighbour (NNN) coupling. Duplexes 9 and 13 showed monomer-like absorption with slight spectral shifts of 5 nm and 30 nm, indicating weak excitonic interactions. An exception was observed for duplex 4 wherein the deviation of the absorption spectrum at 482 nm was seen, attributed to strong H-type excitonic coupling between closely packed PBIs. Similar analyses for duplexes 5–8, which featured pyrene-pyrene NN arrangements, showed comparable behaviour supporting H-aggregate formation via π–π stacking of pyrene units. Notably, even NNN-arranged PBIs or pyrenes in Series B showed strong excitonic interactions, suggesting long-range coupling. In duplexes 10–12, absorption bands were hypsochromically shifted when two or more PBIs were present in alternating positions, even when separated by intervening pyrene units, indicating that significant excitonic coupling persists beyond direct adjacency. Likewise, duplexes 14–16 showed that pyrene units also formed H-aggregates when arranged as next-nearest neighbours.152 Titration experiments further confirmed a shift from strong NN coupling to weaker NNN interactions and these studies led to the conclusion that NN and NNN excitonic couplings can coexist and were tunable by chromophore sequence and spacing along the DNA scaffold. In this work, a classical exciton theory was utilized quantitatively to understand the co-existence of two independent exciton states of PBI and 1,8-dibutynylpyrene in hybrids.
 | ||
| Fig. 22 (a) Top: Schematic of the PBI (blue) and 1,8-dibutynylpyrene (green) building blocks, along with modified oligonucleotides, illustrating the formation of hybrids 1–16. Bottom: Hybrids organized into Series A and B, corresponding to nearest- and next-nearest-neighbour interactions. (b) Illustration of the two coexisting exciton states (H-aggregates) within an alternating PBI–pyrene stack (pyrene: white/green; PBI: grey/blue). Fig. 22a is adapted with permission from ref. 152. Copyright 2015 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Carrascosa, Martin and co-workers (2015) reported the first example of highly ordered n/p-type functional materials formed via electrostatic co-assembly. In this study, two PBI derivatives (PBI 5 and PBI 6), differing in their terminal polar substituents (guanidinium or quaternary ammonium), were employed as n-type components, while previously reported tetrathiafulvalene (TTF) nanofibres served as the p-type counterparts (Fig. 23a).153 UV/vis absorption spectroscopy of PBIs in water revealed a blue-shift (∼20 nm) compared to MeOH, indicative of H-aggregate formation while AFM revealed fibrous morphologies of the assemblies. Due to the intrinsic self-assembly behaviour of both PBI and TTF, a hybrid material was prepared by mixing equal volumes of their aqueous solutions, resulting in a red precipitate, driven by electrostatic interactions. UV/vis absorption spectra of dispersions of 58 (TTF/PBI 5) and 59 (TTF/PBI 6) showed additive spectral features consistent with a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry, suggesting successful and reversible co-assembly through electrostatic association. CD spectroscopy further confirmed the formation of chiral PBI aggregates templated by the TTF assemblies. SEM, TEM, and AFM revealed homogeneously fibrous morphologies (Fig. 23b–d).153 SAXS displayed two distinct peaks corresponding to d-spacings of 9.3 and 5.8 nm, indicating long-range supramolecular order. PXRD further supported the presence of well-defined β-sheet-like structures, showing broad reflections at 2θ = 9.7° and 19.3°. FP-TRMC was utilized to study the charge transport behaviour of the assemblies, whereby upon 355 nm laser excitation, films of the co-assembled hybrid exhibited pronounced photoconductivity transients, demonstrating efficient charge carrier generation superior to that obtained for the individual pristine films. This study represents the first example of an electrostatically driven n/p-type ionic co-assembly with a high degree of structural organization and remarkable photoconductivity with photoconductivity transients of 5.1 × 10−4 and 1.2 × 10−4 cm2 V−1 s−1 for 58 (TTF/PBI 5) and 59 (TTF/PBI 6) respectively. Controlling self-assembly and morphology was a significant challenge; however, electrostatic co-assembly of two complementary self-assembling nanofibres offered a straightforward and accessible strategy to obtain materials with excellent photoconductive properties.
1 stoichiometry, suggesting successful and reversible co-assembly through electrostatic association. CD spectroscopy further confirmed the formation of chiral PBI aggregates templated by the TTF assemblies. SEM, TEM, and AFM revealed homogeneously fibrous morphologies (Fig. 23b–d).153 SAXS displayed two distinct peaks corresponding to d-spacings of 9.3 and 5.8 nm, indicating long-range supramolecular order. PXRD further supported the presence of well-defined β-sheet-like structures, showing broad reflections at 2θ = 9.7° and 19.3°. FP-TRMC was utilized to study the charge transport behaviour of the assemblies, whereby upon 355 nm laser excitation, films of the co-assembled hybrid exhibited pronounced photoconductivity transients, demonstrating efficient charge carrier generation superior to that obtained for the individual pristine films. This study represents the first example of an electrostatically driven n/p-type ionic co-assembly with a high degree of structural organization and remarkable photoconductivity with photoconductivity transients of 5.1 × 10−4 and 1.2 × 10−4 cm2 V−1 s−1 for 58 (TTF/PBI 5) and 59 (TTF/PBI 6) respectively. Controlling self-assembly and morphology was a significant challenge; however, electrostatic co-assembly of two complementary self-assembling nanofibres offered a straightforward and accessible strategy to obtain materials with excellent photoconductive properties.
 | ||
| Fig. 23 (a) (Left) Molecular structures and (right) illustration of the n/p-nanodomains. (b) Microscopic images of the co-assembled 58 (TTF/PBI 5). (c) TEM micrographs showing the fibre-like structures. (d) Fluorescence confocal micrograph of the filament. Fig. 23 is adapted with permission from ref. 153. Copyright 2015, American Chemical Society. | ||
Würthner and co-workers (2019) reported the first synthesis of supramolecular triblock copolymers 60 and 61 using kinetically controlled two-component seeded living polymerization (Fig. 24a and b).37 They explored three bay-substituted PBI derivatives, Cl-PBI, MeS-PBI, and previously studied MeO-PBI, and investigated their self-assembly behaviour using temperature-dependent UV/vis spectroscopy. Rapid cooling produced kinetically trapped H-aggregates, while slow cooling led to thermodynamically stable J-aggregates. Cl-PBI and MeS-PBI displayed faster transformation kinetics than MeO-PBI due to greater steric hindrance and a more twisted π-system, confirmed by DFT-calculated twist angles (∼20° for Cl-/MeS-PBI, ∼11.4° for MeO-PBI). Cooperative nucleation–elongation analysis yielded elongation enthalpies (ΔHe) of −83.3 kJ mol−1 for Cl-PBI and MeS-PBI while ΔG° values around −43.4 kJ mol−1 for Cl–PBI and −43.7 kJ mol−1 for MeS–PBI. AFM imaging revealed distinct helical morphologies, with Cl–PBI and MeS–PBI forming nanofibres of 10 nm and 5 nm pitch, respectively. Using ultrasonicated seeds of MeO-PBI, the authors demonstrated the formation of both A–B–A (60) and B–A–B (61) block copolymers by combining kinetically trapped and seedable PBI derivatives. Seeded polymerization with MeO-PBI as the central block (A) and Cl-PBI or MeS-PBI as terminal blocks (B) led to B–A–B triblock structures (61).37 The UV/vis absorption study confirmed rapid transformation of trapped Cl-PBI and trapped MeS-PBI to their aggregated forms upon MeO-PBI seed addition, with polymerization kinetics dependent on seed-to-monomer ratios. AFM analysis provided morphologies of the aggregates where central segments with a 5 nm pitch (MeO-PBI) were flanked by segments of 10 nm pitch (Cl-PBI).
The B–A–B triblocks (61) remained stable in solution for up to 20 hours.37 This example established a versatile strategy for precise supramolecular polymer design based on kinetic control and unimolecular building blocks, offering advantages over traditional crystallization-driven self-assembly in terms of sequence control. Table 2 summarizes the optical and morphological properties of the above examples of heterochromophoric PBI assemblies and co-assemblies formed via various non-covalent interactions and their applications and future potential for supramolecular electronics.
| Types of chromophores | λ abs (nm) | λ em (nm) | Types of aggregates (mode of interaction) | Morphology | Applications/future potential | Ref. |
|---|---|---|---|---|---|---|
| PBI and OPV (48) | 300–700 | Highly quenched | H-bonding | Ordered network of fibres | — | 144 |
| PBI and OPV (49) | 443, 512 and 552 | 580 | Quadruple H-bonding | Dimers | — | 145 |
| PBI and OPV (50) | 350, 443, 591 | 450–700 | J-aggregates (H- bonding, π−π stacking and solvophobic effects) | Highly interconnected fibrous network morphology | — | 146 |
| Thiophene and PBI (51) | 300–700 | 600 | H-bonding | Fibrous aggregates | Photocurrent generation | 147 |
| OPV and PBI (52) | 300–700 | Non-fluorescent | Individual aggregation in mixed stacks (π–π and hydrophobic interactions) | Highly interconnected fibrous network morphology | I–V measurements (conductivity = 0.02 S cm−1) | 148 |
| N,N′-di(2-(trimethylammoniumiodide)ethylene) perylenediimide (TAIPBI) and tetrakis(4-sulfonatophenyl)porphyrin zinc tetrapotassium salt (ZnTPPSK4) (53) | 434, 442, 450, 515, 556, 559, 595, 601 | 560–700 | Strong π−π and ionic interactions in an aqueous medium | 1D stacks | Potential for organic photovoltaics | 149 |
| Diketopyrrolopyrrole (DPP) and PBI (54 and 55) | 350–650 | — | J-aggregates (H-bonding and π–π stacking) | Head-to-tail alignment of PBI units in heteroaggregates | Potential for photovoltaics | 150 |
| DPP and PBI (54) | 356 (DPP), 622 | 550–750 | H-bonding and orthogonal π-stacking | Superstructure formation | — | 151 |
| Trithienylenevinylene derivative (TTV) and PBI (56) | 300–700 | — | H-bonding and π–π interaction | Rod-like structures | Photoconductivity measurements (12-fold enhancement compared to individual assemblies) | 76 |
| PBI and 1,8-dibutynylpyrene (57) | 450–700 | — | H-aggregates (aromatic stacking interactions) | — | — | 152 |
| Tetrathiafulvalene (TTF) and PBI (58 and 59) | 300–650 | — | Individual aggregation in a co-assembled state (electrostatic interactions) | Fibrous morphology | Photoconductivity measurements (up to 0.8 cm2 V−1 S−1) | 153 |
| PBI derivatives (60 and 61) | 400–700 | — | J-aggregates (H-bonding) | Supramolecular block copolymers | — | 37 |
2.5. Charge transfer based heterochromophoric D/A PBI self-assemblies
Charge transfer (CT) based heterochromophoric D/A self-assemblies based on PBI derivatives154–157 have exhibited promising features in the context of their applications in supramolecular electronics as discussed above144,148,151 and a few representative examples are discussed in this section (Fig. 25a). In bisthiophene–PBI co-oligomer aggregates reported by Burghardt and co-workers, efficient intermolecular CT occurred from the molecular packing in the LC phase. However, long-range charge separation was hindered due to large coulombic barriers and weak charge-transfer integrals, highlighting molecular packing as the decisive factor governing charge separation and competing pathways in aggregated D/A systems.158 CT interactions were also vital in solution-processed D/A co-crystals formed through alternating π–π stacking between PBI acceptors and polycyclic aromatic hydrocarbon donors such as coronene as reported by Zang and co-workers.159 Such co-crystals exhibited bathochromically shifted characteristic CT absorption and emission features, while cofacial stacking led to their self-assembly into 1D, microscale fibrillar architectures. These findings demonstrated a simple solution-processed strategy for the direct fabrication of D/A co-crystals.159 Likewise, another study conducted by Wasielewski, Marks and co-workers showed supramolecular ribbon-like architectures of PBI–porphyrin dyads that provided further insight into charge separation in multichromophoric assemblies. In these systems, spatial segregation of PBI and porphyrin units into distinct columns enabled efficient charge carrier generation and rapid charge delocalization following initial charge separation, establishing an important design principle for OPV materials.105 Furthermore, long-range charge separation driven by symmetry breaking and non-nearest-neighbour charge pair formation was studied in intercalated PBI–polyfluorene blends by time-resolved spectroscopic techniques by Lüer, Toolan and co-workers. These studies demonstrated the generation of weakly bound coulombic charge pairs, emphasizing the importance of long-range transfer and morphological control in achieving near-unity charge separation yields in BHJ OPVs.160 Another example reported by Wasielewski and co-workers demonstrated that single-crystal D/A co-crystals composed of PBI and extended aromatic donors such as peri-xanthenoxanthene exhibited distinct CT absorption and emission features.161 The perpendicular orientation of transition dipole moments and selective photoexcitation of PBI resulted in ultrafast formation of CT exciton evident from femtosecond studies. Likewise, related mixed-stack co-crystals comprised of pyrene donors and various PBI acceptors reported by the same group revealed that subtle changes in co-crystal morphology and acceptor substitution strongly influenced the CT exciton diffusion. Enhanced charge delocalization in one of the PBI derivatives substituted with 2,6-diisopropylphenyl led to increased CT exciton diffusion, underscoring the sensitivity of excited-state dynamics to supramolecular packing.162Beyond co-crystallization,163 studies comparing self-assembly versus complexation behaviour of PBI derivatives by Würthner and co-workers revealed a strong propensity of PBIs to form dimeric aggregates. In the presence of electron-rich polycyclic aromatic hydrocarbons, these electron-deficient PBIs formed well-defined social self-sorted D/A supramolecular complexes.164 The resulting assemblies exhibited guest-dependent excited-state behaviour, wherein either CT interactions or exciplex formation dominated the photophysical response, reinforcing the central role of supramolecular organization in governing CT behaviour.164 Thin-film studies employing organic molecular beam deposition further revealed that molecular intermixing and CT characteristics in D/A systems are strongly modulated by PBI core functionalization and imide side-chain engineering as reported by Schreiber, Brütting, Hinderhofer and co-workers.165 Short alkyl chains at the imide position and electron-withdrawing groups such as cyano groups at bay-substituents promoted well-ordered co-crystal formation with the donor molecule diindenoperylene, giving rise to pronounced CT states.165 These observations highlight how core functionalization and rational side-chain engineering enable precise control over molecular mixing and CT properties. Another strategy to obtain heterochromophoric CT PBI based assemblies was adopted by Lei, Chen and co-workers where host–guest microcrystals were formed from non-macrocyclic PBI derivatives and polyaromatic hydrocarbons such as substituted anthracene guests.166 The guest incorporation in one- and 2D architectures generated either locally excited or CT excited states, leading to highly efficient solid-state emissions spanning from the orange to the near-infrared region.166 Such systems have been further exploited for low-threshold microscale lasing, demonstrating their promise for optoelectronic applications.166
Despite improved understanding of CT dynamics in heterochromophoric PBI assemblies over the years, only a few examples have exhibited the possibility of leveraging charge separation167 and its dynamics for electronic applications, further resulting in functional optoelectronic devices.168–173 Early reports demonstrated the pivotal role of vertical segregation and large interfacial area between CT donor and acceptor units in the generation of efficient photovoltaic devices with external quantum efficiencies (EQE) of >34% and power efficiencies of 2%.174 In this example, Müllen, Friend, MacKenzie and co-workers prepared films of PBI and HBC by a solution processed method, wherein the CT behaviour was confirmed by quenching of emission of PBI. The strong quenching in near equimolar blend solution of PBI and HBC confirmed efficient photoinduced charge separation, while illumination-dependent EQE action spectra further validated effective interfacial exciton dissociation and charge collection governed by the internal vertical morphology.174 Another similar report by Thelakkat, Würthner and co-workers discussed hydrogen bonding mediated self-assembly of D/A CT systems consisting of a PBI organogelator (p-type) and a polymer, poly(vinyl-dimethoxytetraphenylbenzidine) (pvDMTPD, n-type), into organogelator nanowires (Fig. 25c).175 These nanostructured networks were fabricated either by stepwise xerogel preparation followed by solvent removal, pores and groove filling by polymer or by a single-step blend method forming a 3D-PBI-polymer network with varying compositions. SEM analysis indicated twisted entangled strands of the PBI-xerogel superstructure and clear distinction of filling of xerogel vacancies by the amorphous polymer in the blend method.175 Furthermore, LH in multilayer devices (Fig. 25b) was studied using comparison of absorption spectra of the individual layers, indicating broad absorption in the 400–650 nm range. Photoinduced charge separation was also studied by changing the blend ratio in PBI:polymer and revealed a strong composition–device performance relationship. For the fabricated devices, the short circuit current density of 0.28 mA cm−2, an open circuit voltage of 390 mV, a fill factor of 38% and a PCE of 0.04% were obtained, indicating the highest photocurrent and efficiency achieved in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 blend (PBI
1 blend (PBI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) polymer) due to better LH.175 This example underscores the utility of a low molecular weight organogelator and a hole conducting polymer to achieve appreciable charge carrier mobility and better performance in a D/A BHJ. The design and studies of D/A CT systems based on PBI therefore offered promising applications in supramolecular electronics.
polymer) due to better LH.175 This example underscores the utility of a low molecular weight organogelator and a hole conducting polymer to achieve appreciable charge carrier mobility and better performance in a D/A BHJ. The design and studies of D/A CT systems based on PBI therefore offered promising applications in supramolecular electronics.
3. Overview of supramolecular electronics based on PBI homo-assemblies
In molecular electronics, the characteristic molecular dimensions are typically below 5 nm, and the single molecular architectures are typically involved in electronic conduction. In contrast, supramolecular electronics involve electronic conduction through larger assemblies with dimensions in the range of approximately 5–100 nm, where chromophore–chromophore interactions are non-covalent, predominantly π–π stacking and in some cases also hydrogen bonding and van der Waals interactions.20,176,177 Depending on the molecular size and the nature of intermolecular interactions as well as interaction with electrodes, PBI-based systems have been utilized for molecular electronics178,179 as well as their homo-assemblies for supramolecular electronics extensively.180In PBI-based (supra)molecular electronics, charge-carrier mobility is the highest along cofacial π–π stacking directions.52,101,181 Although single crystals can achieve such optimized packing, their large-scale fabrication and controlled device integration remain challenging, particularly in directing charge transport through controlled molecular orientation.182 On the other hand, self-assembled 1D nanomaterials, such as nanowires and nanobelts with π–π stacking aligned along the long axis, offer attractive platforms for both fundamental studies and practical applications by enabling a direct link between molecular packing and charge-transport behaviour.57,70,182–184 Along these lines, various assembly strategies, including poor-solvent addition185 and seeded growth,186 have been developed to obtain nano-belt/fibrous assemblies, some of which are briefly summarized below.
In 2007, amphiphilic PBI molecules were assembled into millimetre-long nanobelts using a seeded self-assembly approach, which displayed enhanced photoconductivity in the presence of hydrazine vapor. The extended nanobelts enabled the fabrication of two-electrode devices, while long-range π–π stacking allowed for efficient conductivity modulation through surface doping.186 Similar nanobelts derived from symmetric PBI showed increased π-electron delocalization upon hydrazine doping, with electron transport confirmed by electron spin resonance (ESR) and I–V measurements.187 In 2010, PBI D–A systems were demonstrated to form millimetre-long nanoribbons via a poor-solvent addition method, exhibiting high photoconductivity under white-light illumination (a photocurrent on/off ratio of ≈103).185 Likewise, alkyl side-chain interfacial engineering produced organic nanofibril heterojunctions with extended D–A interfaces and efficient long-range charge transport, leading to enhanced photocurrent generation and reduced charge recombination in supramolecular photoconductive materials (photocurrent Ion/off ≈ 104).188 In 2016, the same group prepared D–A perylene composite nanostructures that exhibited charge transport and conductivity with a high photocurrent on/off ratio.189 Furthermore, 1-methylpiperidine–substituted PBI was reported to self-assemble into highly conductive 1D nanoribbons due to an intrinsic self-doping effect.190 Interactions between the substituents and PBI cores generated anionic radicals that increased charge-carrier density, while ordered π–π stacking enabled efficient charge transport.190 In 2016, 1-methylpiperidine-PBI (MP-PBI) nanofibres exhibited persistent photoconductivity due to defect-assisted charge separation within π–π stacked structures where the effect was thermally activated, with an activation energy of approximately 137 meV.191 In the following year, charge transport in D–A morpholine substituted PBI (BNO-PBI) nanofibres was shown to be governed by a thermally activated hopping process.192 Packing-dependent photoconductivity was also observed in PBI-functionalized cyanuric acid-melamine assemblies, where a columnar-to-lamellar transition enhanced photoinduced charge transport.193 Overall, controlled molecular design and self-assembly in PBI systems dictate charge transport and optoelectronic performance, as evident from correlations between crystalline order, morphology, and device behaviour in OFETs.194 Single-crystalline PBI nanowire phototransistors195 and peptide-programmed assemblies further highlight how defect minimization and packing control enable exceptional photo-response and efficient performance in devices.196
Since supramolecular systems spontaneously form ordered structures through non-covalent interactions, they severely reduce the reliance on expensive top-down methods of device fabrication, thereby enabling large-area, low-cost manufacturing.58 Furthermore, homo- and heterochromophoric PBI assemblies have played crucial roles in improving our understanding of utilizing these systems for supramolecular electronic applications.118 While homo-assemblies involve simpler chemical synthesis,185 predictable self-assembly behaviour and better control on superstructure formation ensuring long range order, hetero-assemblies involve elaborate synthesis and complex self-assembly processes often requiring a combination of structural, optical and electronic characterization.112 However, the latter provide several advantages such as built-in electric field generation, directional charge separation and transport,109 broadband LH, tunable device performance due to synergistic chromophore interactions, and overall, more promise towards achieving functional supramolecular electronic devices.175 Homo-assemblies are ideal systems for studying fundamental charge transport behaviour,182 while their hetero-counterparts albeit the challenges in device integration and fabrication are better suited for advanced functional supramolecular electronic applications.
4. Conclusion and future perspectives
Over the past few decades, significant progress has been achieved in the design and fundamental understanding of PBI-based D–A systems. Through systematic variations in molecular architecture, linker length, side-chain polarity, and substituent symmetry, researchers have established robust structure–property correlations governing self-assembly, exciton coupling, charge-transport behaviour, and eventual applications and potential towards supramolecular electronics.8,20 Self-assembly provides a powerful strategy to construct well-organized π-conjugated materials with tunable structural and functional properties. As highlighted in this review, self-assembly not only leads to emergent material properties at the supramolecular level but also the translation of these properties at the macroscopic bulk level.7,181,197–199 Supramolecular organization of functional chromophores offers emerging opportunities in materials science, while also bridging the gap between natural and artificial systems by emulating the hierarchical design principles found in natural self-assembled systems. Self-assembly of chromophoric systems continues to attract immense attention due to their central role in organic electronics, supramolecular electronics, and energy-harvesting devices.19Non-covalent interactions such as π–π stacking, hydrogen bonding, and hydrophobic interactions have played a pivotal role in governing aggregate morphology. Examples of earlier developed hydrogen-bonded OPV–PBI assemblies demonstrated the cooperative nature of π–π and hydrogen-bond interactions, while covalently linked more recent systems such as HBC–PBI and thiophene–PBI dyads revealed how controlled D–A organization and nano-segregation promoted efficient charge separation and transport.74,112 In examples of supramolecular systems, hydrogen-bonded systems require precise geometrical complementarity between hydrogen-bond donors and acceptors, where subtle mismatches often result in altered packing or reduced stability. Despite considerable progress, challenges remain in constructing robust self-organized π-systems due to the inherent sensitivity of non-covalent interactions to external conditions such as temperature, solvent polarity, and concentration. Furthermore, precise nanoscale positioning of chromophores, control over dimensionality, and the vertical alignment of supramolecular structures remain non-trivial. Another key challenge is gaining mechanistic insights into the multistep and competitive pathways involved in supramolecular self-assembly. Since the photophysical properties of different aggregates (e.g., H- vs. J-type or mixed states) can differ significantly, a thorough understanding of these pathways is critical. Recent advances in orthogonal self-assembly, particularly the co-organization of p- and n-type fibres, have provided promising approaches that resulted in assemblies with emergent photophysical and charge-transport properties distinct from those of the individual building blocks.144
Although significant advancements have been achieved in the design strategy of supramolecular heterochromophoric systems, their translation into supramolecular electronic applications is still in its infancy. While heterochromophoric assemblies offer several advantages as discussed in Section 3, limited research has been performed on the integration of such systems in electronic device configurations.19 In the context of device applications, self-assembled systems offer unique properties such as self-healing ability that can be exploited to develop self-healing electrodes, miniaturization that can lead to ultra-small but efficient circuits, and ability to efficiently transport holes and electrons that can lead to efficient charge conduction.200 Furthermore, some allied and emerging research areas in this context are stretchable electronic applications by utilizing supramolecular ionic gels,201 utilizing self-assembled host–guest systems for charge transport and electrochemical storage202 and chiral self-assemblies for chiral supramolecular electronics.203 In the last few years, significant efforts have been devoted towards improving the translational potential of homo-and hetero-assemblies, particularly in supramolecular electronics and in several other domains.200
Recent advances in heterochromophoric PBI self-assembly have also been extended to amphiphilic, photoresponsive, and aqueous systems, enabling tunable supramolecular architectures ranging from helical stacks to micellar79,80 and vesicular structures.77,78 The development of self-sorted, compartmentalized assemblies in aqueous media represents a promising avenue, offering insights into biological processes while enabling design of advanced functional materials with adaptive, stimuli-responsive behaviour and cutting-edge applications such as (supra)molecular photocatalysis,204,205 optoelectronics, photofunctional materials, organic photovoltaics, photodetectors and artificial photosynthetic systems.42 In the context of applications of PBI assemblies in (supra)molecular electronics, newer heterochromophoric PBI core lateral extension and annulation,127,128 nanoribbon like structure formation,129,138 strategies for core twisting206 and the study of their self-assemblies could be worth pursuing.
Yet another new frontier in this research domain could be the redox-mediated transient self-assembly in PBI based heterochromophoric systems that remains unexplored. Dissipative self-assembly (DSA) enables the formation of transient, dynamic supramolecular structures through fuel-driven activation and spontaneous deactivation cycles, offering spatiotemporal control over assembly processes.207–210 With advancements made in chemically, pH, and light-controlled DSA systems based on PBIs,211–213 these examples are limited to isolated chromophoric systems in the literature and not explored for D–A type PBI compounds. Owing to their excellent redox and photophysical properties, PBIs are promising candidates for redox-driven DSA due to their ability to undergo reversible reduction to radical anion and dianion states. Their assemblies can be disrupted by inducing redox responsiveness through chemical reductants to form radical anion/dianion monomers, thus leading to oscillatory assembly–disassembly behaviour.210,212 Such strategies could therefore open up avenues for designing new D–A supramolecular materials with emergent tunable and dynamic behaviour and potential applications in electronics.
Looking forward, further progress will depend on integrating precise supramolecular control with dynamic and stimuli-responsive design to achieve adaptive photophysical behaviour and their integration in real device configurations.200 Computational modeling and machine learning can aid design as well as predicting the self-assembly outcomes, while (ultrafast) spectroscopy and advanced nanoscale characterization will be key to unravel the charge dynamics in complex self-assembled architectures. The continued evolution of PBI-based donor–acceptor systems thus promises not only to provide deeper insights into self-assembly mechanisms but also the realization of next-generation functional materials for energy conversion and (supra)molecular electronics in the coming years.
Author contributions
V. Gupta and S. Samanta: literature search, data collection and analysis, writing, preparation of graphics; S. Sengupta: conceiving the topic, literature search, data collection, discussion, writing, editing and overall supervision.Conflicts of interest
There are no conflicts of interest to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors sincerely thank MoE-Scheme for Transformational and Advanced Research in Sciences (STARS) with project no. MoE-STARS/STARS-2/2023–0418 for financial support of the work. S. S. thanks IISER Mohali for a postdoctoral fellowship and V. G. thanks the Prime Minister's Research Fellowship (PMRF) for a PhD fellowship. Dedicated to Prof. Dr. Frank Würthner on the occasion of his 61st Birthday.Notes and references
- T. Brixner, R. Hildner, J. Köhler, C. Lambert and F. Würthner, Adv. Energy Mater., 2017, 7, 1700236 CrossRef.
- G. McDermott, S. M. Prince, A. A. Freer, A. M. Hawthornthwaite-Lawless, M. Z. Papiz, R. J. Cogdell and N. W. Isaacs, Nature, 1995, 374, 517–521 CrossRef CAS.
- J. E. Bullock, R. Carmieli, S. M. Mickley, J. Vura-Weis and M. R. Wasielewski, J. Am. Chem. Soc., 2009, 131, 11919–11929 CrossRef CAS PubMed.
- S. M. Mickley Conron, L. E. Shoer, A. L. Smeigh, A. B. Ricks and M. R. Wasielewski, J. Phys. Chem. B, 2013, 117, 2195–2204 CrossRef CAS PubMed.
- T. S. Balaban, H. Tamiaki and A. R. Holzwarth, Supramolecular Dye Chemistry, Springer-Verlag, Berlin/Heidelberg, 2005, vol. 258, pp. 1–38 Search PubMed.
- T. S. Balaban, Acc. Chem. Res., 2005, 38, 612–623 CrossRef CAS PubMed.
- S. Sengupta and F. Würthner, Acc. Chem. Res., 2013, 46, 2498–2512 CrossRef CAS PubMed.
- J. M. Lim, P. Kim, M.-C. Yoon, J. Sung, V. Dehm, Z. Chen, F. Würthner and D. Kim, Chem. Sci., 2013, 4, 388–397 RSC.
- H. Lin, R. Camacho, Y. Tian, T. E. Kaiser, F. Würthner and I. G. Scheblykin, Nano Lett., 2010, 10, 620–626 CrossRef CAS PubMed.
- Y. Yamamoto, T. Fukushima, Y. Suna, N. Ishii, A. Saeki, S. Seki, S. Tagawa, M. Taniguchi, T. Kawai and T. Aida, Science, 2006, 314, 1761–1764 CrossRef CAS PubMed.
- L. Brunsveld, B. J. B. Folmer, E. W. Meijer and R. P. Sijbesma, Chem. Rev., 2001, 101, 4071–4098 CrossRef CAS PubMed.
- F. J. M. Hoeben, P. Jonkheijm, E. W. Meijer and A. P. H. J. Schenning, Chem. Rev., 2005, 105, 1491–1546 CrossRef CAS PubMed.
- M. M. Safont-Sempere, G. Fernández and F. Würthner, Chem. Rev., 2011, 111, 5784–5814 CrossRef CAS PubMed.
- B. E. Dial and K. D. Shimizu, Appl. Supramol. Chem., 2012, 301–320 CAS.
- F. Würthner, C. R. Saha-Möller, B. Fimmel, S. Ogi, P. Leowanawat and D. Schmidt, Chem. Rev., 2016, 116, 962–1052 CrossRef PubMed.
- R. Dobrawa and F. Würthner, Chem. Commun., 2002, 1878–1879 RSC.
- N. Gospodinova and E. Tomšík, Prog. Polym. Sci., 2015, 43, 33–47 CrossRef CAS.
- V. K. Praveen, C. Ranjith, E. Bandini, A. Ajayaghosh and N. Armaroli, Chem. Soc. Rev., 2014, 43, 4222–4242 RSC.
- A. Jain and S. J. George, Mater. Today, 2015, 18, 206–214 CrossRef CAS.
- A. P. H. J. Schenning and E. W. Meijer, Chem. Commun., 2005, 3245 RSC.
- A. Nowak-Król and F. Würthner, Org. Chem. Front., 2019, 6, 1272–1318 RSC.
- L. Chen, C. Li and K. Müllen, J. Mater. Chem. C, 2014, 2, 1938–1956 RSC.
- M. Son, K. H. Park, C. Shao, F. Würthner and D. Kim, J. Phys. Chem. Lett., 2014, 5, 3601–3607 CrossRef CAS PubMed.
- R. F. Fink, J. Seibt, V. Engel, M. Renz, M. Kaupp, S. Lochbrunner, H.-M. Zhao, J. Pfister, F. Würthner and B. Engels, J. Am. Chem. Soc., 2008, 130, 12858–12859 CrossRef CAS PubMed.
- F. Würthner, Chem. Commun., 2004, 1564–1579 RSC.
- M. A. Martínez, E. E. Greciano and L. Sánchez, Chem. – Eur. J., 2019, 25, 16012–16016 CrossRef PubMed.
- F. Würthner, Pure Appl. Chem., 2006, 78, 2341–2349 CrossRef.
- F. Würthner, T. E. Kaiser and C. R. Saha-Möller, Angew. Chem., Int. Ed., 2011, 50, 3376–3410 CrossRef PubMed.
- M. Hecht and F. Würthner, Acc. Chem. Res., 2021, 54, 642–653 CrossRef CAS PubMed.
- S. Yagai, T. Seki, T. Karatsu, A. Kitamura and F. Würthner, Angew. Chem., Int. Ed., 2008, 47, 3367–3371 CrossRef CAS PubMed.
- S. Ghosh, X. Li, V. Stepanenko and F. Würthner, Chem. – Eur. J., 2008, 14, 11343–11357 CrossRef CAS PubMed.
- F. Würthner, C. Bauer, V. Stepanenko and S. Yagai, Adv. Mater., 2008, 20, 1695–1698 CrossRef.
- C. Rest, M. J. Mayoral and G. Fernández, Int. J. Mol. Sci., 2013, 14, 1541–1565 CrossRef CAS PubMed.
- R. S. Wilson-Kovacs, X. Fang, M. J. L. Hagemann, H. E. Symons and C. F. J. Faul, Chem. – Eur. J., 2022, 28, e202103443 CrossRef CAS PubMed.
- M. Wehner and F. Würthner, Nat. Rev. Chem., 2020, 4, 38–53 CrossRef CAS.
- P. Ximenis, D. Martínez, L. Rubert and B. Soberats, Chem. Soc. Rev., 2025, 54, 11659–11698 RSC.
- W. Wagner, M. Wehner, V. Stepanenko and F. Würthner, J. Am. Chem. Soc., 2019, 141, 12044–12054 CrossRef CAS PubMed.
- D. Cappelletti, M. Barbieri, A. Aliprandi, M. Maggini and L. Đorđević, Nanoscale, 2024, 16, 9153–9168 RSC.
- M. Wehner, M. I. S. Röhr, M. Bühler, V. Stepanenko, W. Wagner and F. Würthner, J. Am. Chem. Soc., 2019, 141, 6092–6107 CrossRef CAS PubMed.
- S. Garain and F. Würthner, Chem. Commun., 2025, 61, 3081–3092 RSC.
- C. You, C. Hippius, M. Grüne and F. Würthner, Chem. – Eur. J., 2006, 12, 7510–7519 CrossRef CAS PubMed.
- P. D. Frischmann, K. Mahata and F. Würthner, Chem. Soc. Rev., 2013, 42, 1847–1870 RSC.
- P. Spenst and F. Würthner, J. Photochem. Photobiol. C, 2017, 31, 114–138 CrossRef CAS.
- V. Gupta and S. Sengupta, Chem. - Asian J., 2025, 20, e00363 CrossRef CAS PubMed.
- D. Zhao, Q. Wu, Z. Cai, T. Zheng, W. Chen, J. Lu and L. Yu, Chem. Mater., 2016, 28, 1139–1146 CrossRef CAS.
- J. H. Kim, T. Schembri, D. Bialas, M. Stolte and F. Würthner, Adv. Mater., 2022, 34, 2104678 CrossRef CAS PubMed.
- H. Sun and Q. Zhang, ChemPhotoChem, 2024, 8, e202300213 CrossRef CAS.
- Y. Shan, X. Xu, X. Jin, X. Ding, S. Wang and H. Chen, Energy Fuels, 2025, 39, 18376–18405 CrossRef CAS.
- C. Rest, R. Kandanelli and G. Fernández, Chem. Soc. Rev., 2015, 44, 2543–2572 RSC.
- F. Würthner, C. Thalacker and A. Sautter, Adv. Mater., 1999, 11, 754–758 CrossRef.
- F. Würthner, C. Thalacker, A. Sautter, W. Schärtl, W. Ibach and O. Hollricher, Chem. – Eur. J., 2000, 6, 3871–3886 CrossRef.
- M. R. Wasielewski, Acc. Chem. Res., 2009, 42, 1910–1921 CrossRef CAS PubMed.
- R. Bhosale, J. Míśek, N. Sakai and S. Matile, Chem. Soc. Rev., 2010, 39, 138–149 RSC.
- J. H. Oh, S. Liu, Z. Bao, R. Schmidt and F. Würthner, Appl. Phys. Lett., 2007, 91, 212107 CrossRef.
- B. A. Jones, A. Facchetti, M. R. Wasielewski and T. J. Marks, Adv. Funct. Mater., 2008, 18, 1329–1339 CrossRef CAS.
- R. Schmidt, J. H. Oh, Y. Sun, M. Deppisch, A.-M. Krause, K. Radacki, H. Braunschweig, M. Könemann, P. Erk, Z. Bao and F. Würthner, J. Am. Chem. Soc., 2009, 131, 6215–6228 CrossRef CAS PubMed.
- L. Zang, Acc. Chem. Res., 2015, 48, 2705–2714 CrossRef CAS PubMed.
- H. Usta, A. Facchetti and T. J. Marks, Acc. Chem. Res., 2011, 44, 501–510 CrossRef CAS PubMed.
- L. Nian, W. Zhang, N. Zhu, L. Liu, Z. Xie, H. Wu, F. Würthner and Y. Ma, J. Am. Chem. Soc., 2015, 137, 6995–6998 CrossRef CAS PubMed.
- Z. Xie, B. Xiao, Z. He, W. Zhang, X. Wu, H. Wu, F. Würthner, C. Wang, F. Xie, L. Liu, Y. Ma, W. Y. Wong and Y. Cao, Mater. Horiz., 2015, 2, 514–518 RSC.
- L. Nian, Z. Chen, S. Herbst, Q. Li, C. Yu, X. Jiang, H. Dong, F. Li, L. Liu, F. Würthner, J. Chen, Z. Xie and Y. Ma, Adv. Mater., 2016, 28, 7521–7526 CrossRef CAS PubMed.
- Z. Xie and F. Würthner, Adv. Energy Mater., 2017, 7, 1602573 CrossRef.
- F. Galeotti, W. Mróz, M. Catellani, B. Kutrzeba-Kotowska and E. Kozma, J. Mater. Chem. C, 2016, 4, 5407–5415 RSC.
- J. H. Oh, Y. Sen Sun, R. Schmidt, M. F. Toney, D. Nordlund, M. Könemann, F. Würthner and Z. Bao, Chem. Mater., 2009, 21, 5508–5518 CrossRef CAS.
- P. Alessio, M. L. Braunger, R. F. Aroca, C. De Almeida Olivati and C. J. L. Constantino, J. Phys. Chem. C, 2015, 119, 12055–12064 CrossRef CAS.
- B. A. Jones, A. Facchetti, M. R. Wasielewski and T. J. Marks, J. Am. Chem. Soc., 2007, 129, 15259–15278 CrossRef CAS PubMed.
- R. Schmidt, M. M. Ling, J. H. Oh, M. Winkler, M. Könemann, Z. Bao and F. Würthner, Adv. Mater., 2007, 19, 3692–3695 CrossRef CAS.
- B. A. Jones, M. J. Ahrens, M. H. Yoon, A. Facchetti, T. J. Marks and M. R. Wasielewski, Angew. Chem., Int. Ed., 2004, 43, 6363–6366 CrossRef CAS PubMed.
- R. J. Chesterfield, J. C. McKeen, C. R. Newman, P. C. Ewbank, D. A. da Silva Filho, J.-L. Brédas, L. L. Miller, K. R. Mann and C. D. Frisbie, J. Phys. Chem. B, 2004, 108, 19281–19292 CrossRef CAS.
- M. Gsänger, J. H. Oh, M. Könemann, H. W. Höffken, A. Krause, Z. Bao and F. Würthner, Angew. Chem., Int. Ed., 2010, 49, 740 CrossRef PubMed.
- I. Song, X. Shang, J. Ahn, J. H. Lee, W. Choi, H. Ohtsu, J. C. Kim, S. K. Kwak and J. H. Oh, Chem. Mater., 2022, 34, 8675–8683 CrossRef CAS.
- N. J. Hestand and F. C. Spano, J. Chem. Phys., 2015, 143, 244707 CrossRef PubMed.
- N. J. Hestand and F. C. Spano, Chem. Rev., 2018, 118, 7069–7163 CrossRef CAS PubMed.
- M. Hecht, T. Schlossarek, S. Ghosh, Y. Tsutsui, A. Schmiedel, M. Holzapfel, M. Stolte, C. Lambert, S. Seki, M. Lehmann and F. Würthner, ACS Appl. Nano Mater., 2020, 3, 10234–10245 CrossRef CAS.
- S. Chen, P. Slattum, C. Wang and L. Zang, Chem. Rev., 2015, 115, 11967–11998 CrossRef CAS PubMed.
- S. Prasanthkumar, S. Ghosh, V. C. Nair, A. Saeki, S. Seki and A. Ajayaghosh, Angew. Chem., Int. Ed., 2015, 54, 946–950 CrossRef CAS PubMed.
- D. Görl, X. Zhang and F. Würthner, Angew. Chem., Int. Ed., 2012, 51, 6328–6348 CrossRef PubMed.
- X. Zhang, S. Rehm, M. M. Safont-Sempere and F. Würthner, Nat. Chem., 2009, 1, 623–629 CrossRef CAS PubMed.
- E. J. Schulze, M. Wu, C. D. Methfessel, E. Spiecker and A. Hirsch, Chem. – Eur. J., 2025, 31, e202500279 CrossRef CAS PubMed.
- E. J. Schulze, C. L. Ritterhoff, E. Franz, O. Tavlui, O. Brummel, B. Meyer and A. Hirsch, Chem. – Eur. J., 2024, 30, e202303515 CrossRef CAS PubMed.
- M. Supur, Y. M. Sung, D. Kim and S. Fukuzumi, J. Phys. Chem. C, 2013, 117, 12438–12445 CrossRef CAS.
- X. Feng, Y. An, Z. Yao, C. Li and G. Shi, ACS Appl. Mater. Interfaces, 2012, 4, 614–618 CrossRef CAS PubMed.
- M. Xu, J. M. Han, Y. Zhang, X. Yang and L. Zang, Chem. Commun., 2013, 49, 11779–11781 RSC.
- V. K. Praveen, B. Vedhanarayanan, A. Mal, R. K. Mishra and A. Ajayaghosh, Acc. Chem. Res., 2020, 53, 496–507 CrossRef CAS PubMed.
- Y. Xiao, Z. Chen, X. Liu, X. Wang, G. Ding, Z. Wang, P. Wang and G. Liao, Mater. Horiz., 2025, 12, 9922–9951 RSC.
- A. Kumari and S. Sengupta, ChemCatChem, 2024, 16, e202301033 CrossRef CAS.
- H. Xu, Z. Wang, S. Feng, X. Liu, X. Gong and J. Hua, Int. J. Hydrogen Energy, 2023, 48, 8071–8081 CrossRef CAS.
- K. Kong, S. Zhang, Y. Chu, Y. Hu, F. Yu, H. Ye, H. Ding and J. Hua, Chem. Commun., 2019, 55, 8090–8093 RSC.
- J. Daniel, A. P. Satheesh and K. Kartha Kalathil, Chem. – Eur. J., 2024, 30, e202401278 CrossRef CAS PubMed.
- H. Kar and S. Ghosh, Isr. J. Chem., 2019, 59, 881–891 CrossRef CAS.
- A. Mukherjee and S. Ghosh, Org. Mater., 2021, 3, 405–416 CrossRef.
- X. Peng, L. Wang and S. Chen, J. Incl. Phenom. Macrocycl. Chem., 2021, 99, 131–154 CrossRef CAS.
- J. Li, P. Li, M. Fan, X. Zheng, J. Guan and M. Yin, Angew. Chem., Int. Ed., 2022, 61, e202202532 CrossRef CAS PubMed.
- S. Xu, X. Wang, L. Zeng and X. Bu, Adv. Mater., 2025, 37, e08888 CrossRef CAS PubMed.
- X. Shang, I. Song, J. H. Lee, W. Choi, J. Ahn, H. Ohtsu, J. C. Kim, J. Y. Koo, S. K. Kwak and J. H. Oh, ACS Nano, 2020, 14, 14146–14156 CrossRef CAS PubMed.
- L. Zhang, I. Song, J. Ahn, M. Han, M. Linares, M. Surin, H.-J. Zhang, J. H. Oh and J. Lin, Nat. Commun., 2021, 12, 142 CrossRef CAS PubMed.
- X. Shang, J. Ahn, J. H. Lee, J. C. Kim, H. Ohtsu, W. Choi, I. Song, S. K. Kwak and J. H. Oh, ACS Appl. Mater. Interfaces, 2021, 13, 12278–12285 CrossRef CAS PubMed.
- X. Shang, I. Song, H. Ohtsu, Y. H. Lee, T. Zhao, T. Kojima, J. H. Jung, M. Kawano and J. H. Oh, Adv. Mater., 2017, 29, 1605828 CrossRef PubMed.
- K. Gao, S. H. Lee, W. Zhao, J. Ahn, T. W. Kim, Z. Li, H. Zhuo, Z. Wang, X. Zheng, Y. Yan, G. Chang, W. Ma, M. Zhang, G. Long, J. H. Oh and X. Shang, Mater. Horiz., 2025, 12, 1903–1912 RSC.
- S. T. Bao, H. Jiang, C. Schaack, S. Louie, M. L. Steigerwald, C. Nuckolls and Z. Jin, J. Am. Chem. Soc., 2022, 144, 18772–18777 CrossRef CAS PubMed.
- L. Zang, Y. Che and J. S. Moore, Acc. Chem. Res., 2008, 41, 1596–1608 CrossRef CAS PubMed.
- A. G. Slater, L. M. A. Perdigão, P. H. Beton and N. R. Champness, Acc. Chem. Res., 2014, 47, 3417–3427 CrossRef CAS PubMed.
- K. Zhao, L. An, X. Zhang, W. Yu, P. Hu, B. Wang, J. Xu, Q. Zeng, H. Monobe, Y. Shimizu, B. Heinrich and B. Donnio, Chem. – Eur. J., 2015, 21, 10379–10390 CrossRef CAS PubMed.
- P. E. Hartnett, S. M. Dyar, E. A. Margulies, L. E. Shoer, A. W. Cook, S. W. Eaton, T. J. Marks and M. R. Wasielewski, Chem. Sci., 2015, 6, 402–411 RSC.
- J. L. Logsdon, P. E. Hartnett, J. N. Nelson, M. A. Harris, T. J. Marks and M. R. Wasielewski, ACS Appl. Mater. Interfaces, 2017, 9, 33493–33503 CrossRef CAS PubMed.
- T. Van der Boom, R. T. Hayes, Y. Zhao, P. J. Bushard, E. A. Weiss and M. R. Wasielewski, J. Am. Chem. Soc., 2002, 124, 9582–9590 CrossRef CAS PubMed.
- V. V. Roznyatovskiy, R. Carmieli, S. M. Dyar, K. E. Brown and M. R. Wasielewski, Angew. Chem., Int. Ed., 2014, 53, 3457–3461 CrossRef CAS PubMed.
- B. Rybtchinski, L. E. Sinks and M. R. Wasielewski, J. Am. Chem. Soc., 2004, 126, 12268–12269 CrossRef CAS PubMed.
- M. Madhu, R. Ramakrishnan, V. Vijay and M. Hariharan, Chem. Rev., 2021, 121, 8234–8284 CrossRef CAS PubMed.
- F. Würthner, Z. Chen, F. J. M. Hoeben, P. Osswald, C. C. You, P. Jonkheijm, J. V. Herrikhuyzen, A. P. H. J. Schenning, P. P. A. M. Van Der Schoot, E. W. Meijer, E. H. A. Beckers, S. C. J. Meskers and R. A. J. Janssen, J. Am. Chem. Soc., 2004, 126, 10611–10618 CrossRef PubMed.
- E. H. A. Beckers, S. C. J. Meskers, A. P. H. J. Schenning, Z. Chen, F. Würthner, P. Marsal, D. Beljonne, J. Cornil and R. A. J. Janssen, J. Am. Chem. Soc., 2006, 128, 649–657 CrossRef CAS PubMed.
- L. F. Dössel, V. Kamm, I. A. Howard, F. Laquai, W. Pisula, X. Feng, C. Li, M. Takase, T. Kudernac, S. De Feyter and K. Müllen, J. Am. Chem. Soc., 2012, 134, 5876–5886 CrossRef PubMed.
- J. Peng, F. Zhai, X. Guo, X. Jiang and Y. Ma, RSC Adv., 2014, 4, 13078–13084 RSC.
- Y. Shi and X. Y. Li, Adv. Mater. Res., 2014, 936, 911–914 Search PubMed.
- K. J. Lee, J. H. Woo, Y. Xiao, E. Kim, L. M. Mazur, D. Kreher, A. J. Attias, K. Matczyszyn, M. Samoc, B. Heinrich, S. Méry, F. Fages, L. Mager, A. D’Aléo, J. W. Wu, F. Mathevet, P. André and J. C. Ribierre, RSC Adv., 2016, 6, 57811–57819 RSC.
- D. Bialas, C. Brüning, F. Schlosser, B. Fimmel, J. Thein, V. Engel and F. Würthner, Chem. – Eur. J., 2016, 22, 15011–15018 CrossRef CAS PubMed.
- D. Rolland, J. C. Brauer, L. Hartmann, L. Biniek, M. Brinkmann, N. Banerji and H. Frauenrath, J. Mater. Chem. C, 2017, 5, 1383–1393 RSC.
- M. Hecht, T. Schlossarek, M. Stolte, M. Lehmann and F. Würthner, Angew. Chem., Int. Ed., 2019, 58, 12979–12983 CrossRef CAS PubMed.
- T. Türel and S. Valiyaveettil, J. Org. Chem., 2020, 85, 10593–10602 CrossRef PubMed.
- S. Datta and D. Chaudhuri, Angew. Chem., Int. Ed., 2022, 61, e202201956 CrossRef CAS PubMed.
- B. Fimmel, M. Son, Y. M. Sung, M. Grüne, B. Engels, D. Kim and F. Würthner, Chem. – Eur. J., 2015, 21, 615–630 CrossRef CAS PubMed.
- A. Nowak-Król, B. Fimmel, M. Son, D. Kim and F. Würthner, Faraday Discuss., 2015, 185, 507–527 RSC.
- M. Son, B. Fimmel, V. Dehm, F. Würthner and D. Kim, ChemPhysChem, 2015, 16, 1757–1767 CrossRef CAS PubMed.
- B. Mahlmeister, R. Renner, O. Anhalt, M. Stolte and F. Würthner, J. Mater. Chem. C, 2022, 10, 2581–2591 RSC.
- A. Nowak-Król, K. Shoyama, M. Stolte and F. Würthner, Chem. Commun., 2018, 54, 13763–13772 RSC.
- X. Zhang, Z. Lu, L. Ye, C. Zhan, J. Hou, S. Zhang, B. Jiang, Y. Zhao, J. Huang, S. Zhang, Y. Liu, Q. Shi, Y. Liu and J. Yao, Adv. Mater., 2013, 25, 5791–5797 CrossRef CAS PubMed.
- W. Jiang, Y. Li and Z. Wang, Acc. Chem. Res., 2014, 47, 3135–3147 CrossRef CAS PubMed.
- N. Liang, D. Meng and Z. Wang, Acc. Chem. Res., 2021, 54, 961–975 CrossRef CAS PubMed.
- S. R. Peurifoy, T. J. Sisto, F. Ng, M. L. Steigerwald, R. Chen and C. Nuckolls, Chem. Rec., 2019, 19, 1050–1061 CrossRef CAS PubMed.
- M. Ball, Y. Zhong, Y. Wu, C. Schenck, F. Ng, M. Steigerwald, S. Xiao and C. Nuckolls, Acc. Chem. Res., 2015, 48, 267–276 CrossRef CAS PubMed.
- W. Jiang, Y. Li and Z. Wang, Chem. Soc. Rev., 2013, 42, 6113–6127 RSC.
- C. H. Wu, C. C. Chueh, Y. Y. Xi, H. L. Zhong, G. P. Gao, Z. H. Wang, L. D. Pozzo, T. C. Wen and A. K. Y. Jen, Adv. Funct. Mater., 2015, 25, 5326–5332 CrossRef CAS.
- T. J. Sisto, Y. Zhong, B. Zhang, M. T. Trinh, K. Miyata, X. Zhong, X. Y. Zhu, M. L. Steigerwald, F. Ng and C. Nuckolls, J. Am. Chem. Soc., 2017, 139, 5648–5651 CrossRef CAS PubMed.
- N. J. Schuster, R. Hernández Sánchez, D. Bukharina, N. A. Kotov, N. Berova, F. Ng, M. L. Steigerwald and C. Nuckolls, J. Am. Chem. Soc., 2018, 140, 6235–6239 CrossRef CAS PubMed.
- V. B. R. Pedersen, S. K. Pedersen, Z. Jin, N. Kofod, B. W. Laursen, G. V. Baryshnikov, C. Nuckolls and M. Pittelkow, Angew. Chem., Int. Ed., 2022, 61, e202212293 CrossRef CAS PubMed.
- M. Ball, B. Fowler, P. Li, L. A. Joyce, F. Li, T. Liu, D. Paley, Y. Zhong, H. Li, S. Xiao, F. Ng, M. L. Steigerwald and C. Nuckolls, J. Am. Chem. Soc., 2015, 137, 9982–9987 CrossRef CAS PubMed.
- B. Zhang, M. T. Trinh, B. Fowler, M. Ball, Q. Xu, F. Ng, M. L. Steigerwald, X.-Y. Zhu, C. Nuckolls and Y. Zhong, J. Am. Chem. Soc., 2016, 138, 16426–16431 CrossRef CAS PubMed.
- S. R. Peurifoy, Q. Xu, R. May, N. A. Gadjieva, T. J. Sisto, Z. Jin, L. E. Marbella and C. Nuckolls, Chem. Sci., 2020, 11, 9978–9982 RSC.
- J. Zhang, Y. Li, J. Huang, H. Hu, G. Zhang, T. Ma, P. C. Y. Chow, H. Ade, D. Pan and H. Yan, J. Am. Chem. Soc., 2017, 139, 16092–16095 CrossRef CAS PubMed.
- H. Hu, Y. Li, J. Zhang, Z. Peng, L. Kuen Ma, J. Xin, J. Huang, T. Ma, K. Jiang, G. Zhang, W. Ma, H. Ade and H. Yan, Adv. Energy Mater., 2018, 8, 1800234 CrossRef.
- J. Feng, N. Liang, W. Jiang, D. Meng, R. Xin, B. Xu, J. Zhang, Z. Wei, J. Hou and Z. Wang, Org. Chem. Front., 2017, 4, 811–816 RSC.
- T. Seki, X. Lin and S. Yagai, Asian J. Org. Chem., 2013, 2, 708–724 CrossRef CAS.
- D. González-Rodríguez and A. P. H. J. Schenning, Chem. Mater., 2011, 23, 310–325 CrossRef.
- J. Van Herrikhuyzen, A. Syamakumari, A. P. H. J. Schenning and E. W. Meijer, J. Am. Chem. Soc., 2004, 126, 10021–10027 CrossRef CAS PubMed.
- J. Zhang, F. J. M. Hoeben, M. J. Pouderoijen, A. P. H. J. Schenning, E. W. Meijer, F. C. De Schryver and S. De Feyter, Chem. – Eur. J., 2006, 12, 9046–9055 CrossRef CAS PubMed.
- F. J. M. Hoeben, J. Zhang, C. C. Lee, M. J. Pouderoijen, M. Wolffs, F. Würthner, A. P. H. J. Schenning, E. W. Meijer and S. De Feyter, Chem. – Eur. J., 2008, 14, 8579–8589 CrossRef CAS PubMed.
- K. Sugiyasu, S. Kawano, N. Fujita and S. Shinkai, Chem. Mater., 2008, 20, 2863–2865 CrossRef CAS.
- K. V. Rao and S. J. George, Chem. – Eur. J., 2012, 18, 14286–14291 CrossRef CAS PubMed.
- M. Supur and S. Fukuzumi, J. Phys. Chem. C, 2012, 116, 23274–23282 CrossRef CAS.
- S. Rieth, Z. Li, C. E. Hinkle, C. X. Guzman, J. J. Lee, S. I. Nehme and A. B. Braunschweig, J. Phys. Chem. C, 2013, 117, 11347–11356 CrossRef CAS.
- D. Ley, C. X. Guzman, K. H. Adolfsson, A. M. Scott and A. B. Braunschweig, J. Am. Chem. Soc., 2014, 136, 7809–7812 CrossRef CAS PubMed.
- C. B. Winiger, S. M. Langenegger, G. Calzaferri and R. Häner, Angew. Chem., Int. Ed., 2015, 127, 3714–3718 CrossRef.
- J. López-Andarias, M. J. Rodriguez, C. Atienza, J. L. López, T. Mikie, S. Casado, S. Seki, J. L. Carrascosa and N. Martín, J. Am. Chem. Soc., 2015, 137, 893–897 CrossRef PubMed.
- Y. Hong, J. Kim, W. Kim, C. Kaufmann, H. Kim, F. Würthner and D. Kim, J. Am. Chem. Soc., 2020, 142, 7845–7857 CrossRef CAS PubMed.
- M. L. Williams, I. Schlesinger, R. M. Jacobberger and M. R. Wasielewski, J. Am. Chem. Soc., 2022, 144, 18607–18618 CrossRef CAS PubMed.
- J. R. Palmer, S. B. Tyndall, G. C. Mantel, O. J. Buras, R. M. Young, M. D. Krzyaniak and M. R. Wasielewski, J. Am. Chem. Soc., 2025, 147, 17394–17403 CrossRef CAS PubMed.
- H. Loong, J. Zhou, N. Jiang, Y. Feng, G. Xie, L. Liu and Z. Xie, J. Phys. Chem. B, 2022, 126, 2441–2448 CrossRef PubMed.
- M. Polkehn, H. Tamura, P. Eisenbrandt, S. Haacke, S. Méry and I. Burghardt, J. Phys. Chem. Lett., 2016, 7, 1327–1334 CrossRef CAS PubMed.
- C. Wang, J. Wang, N. Wu, M. Xu, X. Yang, Y. Lu and L. Zang, RSC Adv., 2017, 7, 2382–2387 RSC.
- A. Isakova, S. Karuthedath, T. Arnold, J. R. Howse, P. D. Topham, D. T. W. Toolan, F. Laquai and L. Lüer, Nanoscale, 2018, 10, 10934–10944 RSC.
- I. Schlesinger, N. E. Powers-Riggs, J. L. Logsdon, Y. Qi, S. A. Miller, R. Tempelaar, R. M. Young and M. R. Wasielewski, Chem. Sci., 2020, 11, 9532–9541 RSC.
- M. S. Myong, Y. Qi, C. Stern and M. R. Wasielewski, J. Mater. Chem. C, 2021, 9, 16911–16917 RSC.
- V. Belova, A. Hinderhofer, C. Zeiser, T. Storzer, J. Rozbořil, J. Hagenlocher, J. Novák, A. Gerlach, R. Scholz and F. Schreiber, J. Phys. Chem. C, 2020, 124, 11639–11651 CrossRef CAS.
- S. Soldner, O. Anhalt, M. B. Sárosi, M. Stolte and F. Würthner, Chem. Commun., 2023, 59, 11656–11659 RSC.
- N. Rußegger, H. Kim, J. Hagara, O. Vladimirov, T. Eberle, I. Zaluzhnyy, A. Gerlach, A. Hinderhofer, W. Brütting and F. Schreiber, J. Phys. Chem. C, 2023, 127, 11128–11137 CrossRef.
- H. Gao, H. He, L. Zhang, Z. Feng, X. Chen and Y. Lei, Adv. Opt. Mater., 2024, 12, 2400619 CrossRef CAS.
- L. Ernst, H. Song, D. Kim and F. Würthner, Nat. Chem., 2025, 17, 767–776 CrossRef CAS PubMed.
- A. A. Mohapatra, M. Pranav, S. Yadav, C. Gangadharappa, J. Wu, C. Labanti, J. Wolansky, J. Benduhn, J.-S. Kim, J. Durrant and S. Patil, ACS Appl. Mater. Interfaces, 2023, 15, 25224–25231 CrossRef CAS PubMed.
- P. Yu, Y. Li, H. Zhao, L. Zhu, Y. Wang, W. Xu, Y. Zhen, X. Wang, H. Dong, D. Zhu and W. Hu, Small, 2021, 17, 2006574 CrossRef CAS PubMed.
- R. Sato, D. Yoo and T. Mori, CrystEngComm, 2019, 21, 3218–3222 RSC.
- Y. Su, Y. Li, J. Liu, R. Xing and Y. Han, Nanoscale, 2015, 7, 1944–1955 RSC.
- E. H. A. Beckers, Z. Chen, S. C. J. Meskers, P. Jonkheijm, A. P. H. J. Schenning, X.-Q. Li, P. Osswald, F. Würthner and R. A. J. Janssen, J. Phys. Chem. B, 2006, 110, 16967–16978 CrossRef CAS PubMed.
- M. Wolffs, N. Delsuc, D. Veldman, N. V. Anh, R. M. Williams, S. C. J. Meskers, R. A. J. Janssen, I. Huc and A. P. H. J. Schenning, J. Am. Chem. Soc., 2009, 131, 4819–4829 CrossRef CAS PubMed.
- L. Schmidt-Mende, A. Fechtenkötter, K. Müllen, E. Moons, R. H. Friend and J. D. MacKenzie, Science, 2001, 293, 1119–1122 CrossRef CAS PubMed.
- A. Wicklein, S. Ghosh, M. Sommer, F. Würthner and M. Thelakkat, ACS Nano, 2009, 3, 1107–1114 CrossRef CAS PubMed.
- S. Herbst, B. Soberats, P. Leowanawat, M. Stolte, M. Lehmann and F. Würthner, Nat. Commun., 2018, 9, 2646 CrossRef PubMed.
- E. W. Meijer and A. P. H. J. Schenning, Nature, 2002, 419, 353–354 CrossRef CAS PubMed.
- I. Díez-Pérez, Z. Li, S. Guo, C. Madden, H. Huang, Y. Che, X. Yang, L. Zang and N. Tao, ACS Nano, 2012, 6, 7044–7052 CrossRef PubMed.
- C. Li, A. Mishchenko, Z. Li, I. Pobelov, T. Wandlowski, X. Q. Li, F. Würthner, A. Bagrets and F. Evers, J. Phys.: Condens. Matter, 2008, 20, 374122 CrossRef CAS PubMed.
- H. Huang, C. E. Chou, Y. Che, L. Li, C. Wang, X. Yang, Z. Peng and L. Zang, J. Am. Chem. Soc., 2013, 135, 16490–16496 CrossRef CAS PubMed.
- Z. Chen, V. Stepanenko, V. Dehm, P. Prins, L. D. A. Siebbeles, J. Seibt, P. Marquetand, V. Engel and F. Würthner, Chem. – Eur. J., 2007, 13, 436–449 CrossRef CAS PubMed.
- F. C. Grozema and L. D. A. Siebbeles, Int. Rev. Phys. Chem., 2008, 27, 87–138 Search PubMed.
- A. Tolkki, E. Vuorimaa, V. Chukharev, H. Lemmetyinen, P. Ihalainen, J. Peltonen, V. Dehm and F. Würthner, Langmuir, 2010, 26, 6630–6637 CrossRef CAS PubMed.
- Z. Chen, M. G. Debije, T. Debaerdemaeker, P. Osswald and F. Würthner, ChemPhysChem, 2004, 5, 137–140 CrossRef CAS PubMed.
- Y. Che, X. Yang, G. Liu, C. Yu, H. Ji, J. Zuo, J. Zhao and L. Zang, J. Am. Chem. Soc., 2010, 132, 5743–5750 CrossRef CAS PubMed.
- Y. Che, A. Datar, K. Balakrishnan and L. Zang, J. Am. Chem. Soc., 2007, 129, 7234–7235 CrossRef CAS PubMed.
- Y. Che, A. Datar, X. Yang, T. Naddo, J. Zhao and L. Zang, J. Am. Chem. Soc., 2007, 129, 6354–6355 CrossRef CAS PubMed.
- Y. Che, H. Huang, M. Xu, C. Zhang, B. R. Bunes, X. Yang and L. Zang, J. Am. Chem. Soc., 2011, 133, 1087–1091 CrossRef CAS PubMed.
- C. Wang, B. R. Bunes, M. Xu, N. Wu, X. Yang, D. E. Gross and L. Zang, ACS Sens., 2016, 1, 552–559 CrossRef CAS.
- N. Wu, C. Wang, B. R. Bunes, Y. Zhang, P. M. Slattum, X. Yang and L. Zang, ACS Appl. Mater. Interfaces, 2016, 8, 12360–12368 CrossRef CAS PubMed.
- N. Wu, C. Wang, P. M. Slattum, Y. Zhang, X. Yang and L. Zang, ACS Energy Lett., 2016, 1, 906–912 CrossRef CAS.
- N. Wu, Y. Zhang, C. Wang, P. M. Slattum, X. Yang and L. Zang, J. Phys. Chem. Lett., 2017, 8, 292–298 CrossRef CAS PubMed.
- S. Yagai, M. Usui, T. Seki, H. Murayama, Y. Kikkawa, S. Uemura, T. Karatsu, A. Kitamura, A. Asano and S. Seki, J. Am. Chem. Soc., 2012, 134, 7983–7994 CrossRef CAS PubMed.
- R. C. Savage, E. Orgiu, J. M. Mativetsky, W. Pisula, T. Schnitzler, C. L. Eversloh, C. Li, K. Müllen and P. Samorì, Nanoscale, 2012, 4, 2387–2393 RSC.
- H. Yu, Z. Bao and J. H. Oh, Adv. Funct. Mater., 2013, 23, 629–639 CrossRef CAS.
- R. E. Ginesi, F. J. Angus, M. Giza, G. N. M. K. Macleod, J. Doutch, P. Docampo and E. R. Draper, Adv. Mater. Interfaces, 2025, 12, 2500151 CrossRef CAS.
- C. Li and H. Wonneberger, Adv. Mater., 2012, 24, 613–636 CrossRef CAS PubMed.
- Y. Zhao, Y. Guo and Y. Liu, Adv. Mater., 2013, 25, 5372–5391 CrossRef CAS PubMed.
- H. Marciniak, X.-Q. Li, F. Würthner and S. Lochbrunner, J. Phys. Chem. A, 2011, 115, 648–654 CrossRef CAS PubMed.
- D. E. Balderston, E. Feo, A. Leonescu, M. Stevens, A. M. Wilmshurst, P. A. Gale, C. J. E. Haynes, G. T. Williams and J. R. Hiscock, Chem. Soc. Rev., 2025, 54, 8888–8924 RSC.
- S. Yamada and T. Honda, ACS Mater. Au, 2025, 5, 35–44 CrossRef CAS PubMed.
- A. H. Abdelhay and A. D. Bani-Yaseen, Mater. Today Chem., 2024, 40, 102259 CrossRef CAS.
- A. M. Garcia, G. Martínez and A. Ruiz-Carretero, Front. Chem., 2021, 9, 722727 CrossRef CAS PubMed.
- Z. Li, F. Liu, Y. Lu, J. Hu, J. Feng, H. Shang, B. Sun and W. Jiang, ACS Catal., 2025, 15, 1829–1840 CrossRef CAS.
- D. McDowall, B. J. Greeves, R. Clowes, K. McAulay, A. M. Fuentes-Caparrós, L. Thomson, N. Khunti, N. Cowieson, M. C. Nolan, M. Wallace, A. I. Cooper, E. R. Draper, A. J. Cowan and D. J. Adams, Adv. Energy Mater., 2020, 10, 2002469 CrossRef CAS.
- C. Schaack, A. M. Evans, F. Ng, M. L. Steigerwald and C. Nuckolls, J. Am. Chem. Soc., 2022, 144, 42–51 CrossRef CAS PubMed.
- A. Sharko, D. Livitz, S. De Piccoli, K. J. M. Bishop and T. M. Hermans, Chem. Rev., 2022, 122, 11759–11777 CrossRef CAS PubMed.
- S. A. P. Van Rossum, M. Tena-Solsona, J. H. Van Esch, R. Eelkema and J. Boekhoven, Chem. Soc. Rev., 2017, 46, 5519–5535 RSC.
- S. De and R. Klajn, Adv. Mater., 2018, 30, 1706750 CrossRef PubMed.
- M. Weißenfels, J. Gemen and R. Klajn, Chem, 2021, 7, 23–37 Search PubMed.
- A. Sorrenti, J. Leira-Iglesias, A. J. Markvoort, T. F. A. De Greef and T. M. Hermans, Chem. Soc. Rev., 2017, 46, 5476–5490 RSC.
- J. Leira-Iglesias, A. Sorrenti, A. Sato, P. A. Dunne and T. M. Hermans, Chem. Commun., 2016, 52, 9009–9012 RSC.
- C. Chen, J. S. Valera, T. B. M. Adachi and T. M. Hermans, Chem. – Eur. J., 2023, 29, e202202849 CrossRef CAS PubMed.
Footnote |
| † Contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |