Ionic liquid-based fluorescence sensing paper: rapid, ultrasensitive, and in-site detection of methamphetamine in human urine†
Abstract
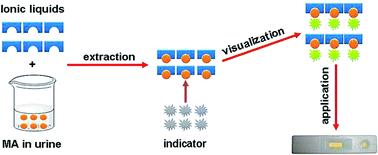
A low cost sensing paper chip supporting an ionic liquid and a fluorescent indicator agent was developed to perform a rapid and ultrasensitive in-site methamphetamine test in human urine. The extraction ability of ionic liquids for methamphetamine, fluorescence behaviour of the system, and performance of the sensing paper were emphasized.

 Please wait while we load your content...
Please wait while we load your content...