Porous aerogels prepared by crosslinking of cellulose with 1,4-butanediol diglycidyl ether in NaOH/urea solution
Abstract
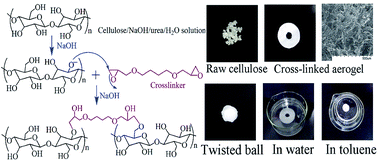
Cellulose aerogels based on crosslinking of cellulose with 1,4-butanediol diglycidyl ether (BDE) were homogeneously synthesized in NaOH/urea aqueous solution followed by freeze-drying. In the NaOH/urea aqueous solution, cellulose existed in a sodium alkoxide form that could react with the epoxide groups of BDE. The rheological behavior of the cellulose/NaOH/urea aqueous solution showed that the crosslinking reaction occurred immediately once the BDE was added to the cellulose/NaOH/urea aqueous solution at room temperature. The X-ray diffraction (XRD) characterization identified a transition from the crystalline structure of cellulose to an amorphous state of cellulose aerogels with increasing amount of BDE. Elemental analysis revealed the variation in carbon and oxygen elemental percentages in cellulose aerogels caused by the reaction between cellulose and BDE. The porous network of aerogels was observed by scanning electron microscopy (SEM) and the pore size of the aerogels increased as a function of BDE. The water adsorbent ability of aerogels was up to 41 g g−1—even after 30 squeezing–adsorption cycles, the water adsorbent ability was still 37 g g−1.

 Please wait while we load your content...
Please wait while we load your content...