Application of chiral cationic iridium(III) complexes for triplet–triplet annihilation up-conversion of photon energy in R-limonene
Akihiko
Yamagishi
a,
Jun
Yoshida
 b and
Hisako
Sato
b and
Hisako
Sato
 *c
*c
aFaculty of Medicine, Toho University, 5-21-16 Oomori-nishi, Ota-ku, Tokyo, 143-8540, Japan
bCollege of Humanities & Sciences, Nihon University, Department of Chemistry, Setagaya-ku, Tokyo 156-8550, Japan
cFaculty of Science Ehime University, 2-5, Bunkyo-cho, Matsuyama, 790-8577, Japan. E-mail: sato.hisako.yq@ehime-u.ac.jp
First published on 7th January 2026
Abstract
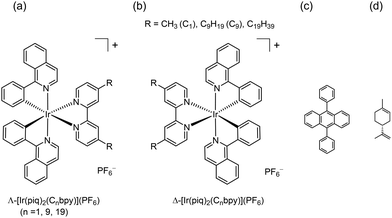
Converting photon energy in a green solvent is a challenge for developing environmentally friendly technologies. Triplet–triplet annihilation up-conversion (TTA-UC) in R-limonene is one such attempt. Since R-limonene is a nonpolar medium, conventional organic compounds or neutral metal complexes are used as a donor. In this study, cationic chiral iridium(III) complexes are used for the first time. The studies were based on the finding that a cyclometalated cationic iridium(III) complex in a chiral form, Δ- or Λ-[Ir(piq)2(dmbpy)]+ (piqH = 1-phenyisoquinoline, dmbpy = 4,4′-dimethyl-2,2′-bipyridine), was soluble enough in R-limonene to perform TTA-UC. Conversely, its racemic form was barely soluble due to its high crystallinity. When an R-limonene solution containing chiral [Ir(piq)2(dmbpy)]PF6 (ca. 2 × 10−5 M) and DPA (0.3–2.6 mM) was irradiated with laser light at 445 nm, the up-conversion of photon energy to 430 nm was achieved with a quantum yield as high as 4.5% under air. Despite the slight difference in solubility between the enantiomers, no stereoselectivity was observed in TTA-UC. The effects of long alkyl chains attached to a donor molecule, or Δ- or Λ- [Ir(piq)2(Cnbpy)]+ (Cnbpy = 4,4′-nonyl (C9)- or 4,4′-nonyldecyl (C19)-2,2′-bipyridine), were investigated on the solubility and efficiency of TTA-UC.
Introduction
Recently there has been wide interest in the pursuit of an elucidation of the mechanism by which molecular systems convert photon energy in green solvents.1–4 This is expected to solve energy supply problems without damaging environments. Among these investigations, triplet–triplet annihilation up-conversion (TTA-UC) has been studied using R-limonene as a representative green solvent.5–11R-limonene is a non-polar organic solvent that is reported to reduce the quenching effects of oxygen molecules in air due to the presence of a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond.2 Conventionally, emitting metal complexes with no charge, such as Pt-porphyrin complexes, have been used since they dissolve in R-limonene. These complexes attain a quantum yield of a few percent.2
C bond.2 Conventionally, emitting metal complexes with no charge, such as Pt-porphyrin complexes, have been used since they dissolve in R-limonene. These complexes attain a quantum yield of a few percent.2
Cyclometalated iridium(III) complexes (denoted as Ir(III) complexes) are extensively used as a donor in TTA-UC because they are highly emissive in the UV- visible region.12–25 In fact, our group established the systems of triplet–triplet annihilation up-conversion (TTA-UC) of photon energy by use of various types of Ir(III) complexes.25 They are positively charged and are hybridized with inorganic polymers such as clay minerals. The systems achieve reasonable quantum yield of a few percentages even under air.25–28
Using these complexes in non-polar green solvents would expand the range of donors in TTA-UC systems. Motivated by this expectation, the present study attempted to use positively charged iridium(III) complexes. To overcome the issue of low solubility, we found that the chiral form of a positively charged Ir(III) complex was soluble enough in R-limonene. The used complex was Δ- or Λ-[Ir(piq)2(dmbpy)]+ (piqH = 1-phenyisoquinoline, dmbpy = 4,4′-dimethyl-2,2′-bipyridine). The racemic form of the same complex was hardly soluble probably due to high crystallinity. An R-limonene solution containing the chiral cationic complex as a donor and 9, 10-diphenylanthracene (DPA) as an acceptor, the quantum yield for TTA-UC attained as high as 4.5% under air. In this study, R-limonene was used instead of its antipodal enantiomer, because the formed was available in a purer form commercially.
Results and discussion
Solubility of cationic iridium(III) complexes in R-limonene
In the present study, a cationic chiral Ir(III) complex, Δ- or Λ-[Ir(piq)2(dmbpy)]+, was used as a donor in TTA-UC. The complex (Chart 1) was selected because it possessed a broad absorption band with the peak position at 430 nm and absorbed a laser light at 445 nm.26 In order to see the effects of long alkyl chains, the complexes, Δ- or Λ- [Ir(piq)2(Cnbpy)]+ (Cn-bpy = 4,4′-nonyl (C9)- or 4,4′-nonyldecyl (C19)-2,2′-bipyridine), were also studied. The preparation of enantiomeric [Ir(piq)2(dmbpy)]+ and [Ir(piq)2(C9bpy)]+ was reported previously.26–28 The chromatographic resolution of [Ir(piq)2(C19-bpy)]+ was achieved in the present work as shown in Fig. S1 (SI). The electronic circular dichroism (ECD) spectra of these enantiomers are shown in Fig. S1 and S2 (SI). The assignment of Δ- or Λ-configuration was performed on the basis of their ECD spectra.26–31Firstly chirality effects were studied on the solubility of [Ir(piq)2(dmbpy)]PF6 in R-limonene. Fig. S3 (SI) shows that the temperature dependence of solubility of the racemic (ΔΛ-), Δ- and Λ-enantiomers of the complex. As a result, chirality influenced solubility in this solvent remarkably. At 273 K, for example, ΔΛ-, Δ- and Λ-enantiomers were soluble to 0.8 × 10−5 M, 2.0 × 10−4, and 2.5 × 10−4 M, respectively. Usually, a racemate takes a stable antipodal pair in a crystalline state so that it is less soluble than an enantiomer.32,33 Therefore, the higher solubility of the enantiomeric form compared to the racemic one was attributed to low crystallinity in the solid states. Slight difference was observed between the opposite enantiomers. It reflected stereoselective effects when chiral R-limonene molecules solvated Δ- or Λ-[Ir(piq)2(dmbpy)]PF6, respectively.
The effects of long alkyl chains were investigated for the complexes of Δ- or Λ- [Ir(piq)2(Cnbpy)]+ (Cnbpy = 4,4′-nonyl (C9)- or 4,4′-nonyldecyl (C19)-2,2′-bipyridine). These complexes were soluble to more than 10−4 M at room temperature even in the form of racemates. Reasons might lie in poor crystallinity since the long alkyl chains were possible to take various conformations in solid states with nearly the same free energy. The enantiomers were even more soluble to more than 10−3 M at room temperature.
Energy transfer in R-limonene
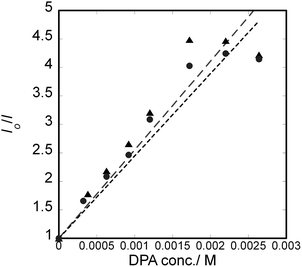
The enantiomeric forms of [Ir(piq)2(dmbpy)]PF6 were used for studying the photochemical processes in R-limonene. The racemate was hardly soluble so that it was unable to provide sufficient emission under the irradiation of a laser light. When a solution of an enantiomeric Ir(III) complex in the solvent was irradiated at 445 nm, emission was observed with the dual peaks around 590 nm and 630 nm. The duality suggested the emissions from two excited triplet state with different vibrational levels. The intensity of the emission decreased upon the addition of DPA. It indicated that the photon energy was transferred from excited Δ-or Λ-[Ir(piq)2(dmbpy)]+ to DPA. The same experiments were performed using Δ- or Λ-[Ir(piq)2(Cnbpy)]+ (n = 9 or 19). Their emission spectra are given in Fig. S4–S6 (SI).Fig. 1 shows the plots of the I0/I ratios against the DPA concentration (Stern–Volmer plots) at 300 K under air, where I0 and I denote the intensities of emission at 590 nm in the absence and presence of DPA, respectively. The plots were constructed according to the following equation:
| I0/I = 1 + KSV[DPA]. | (1) |
| KSV = kq/{kr + kqox[O2]}, | (2) |
According to the results in Table 1, it was concluded that energy transfer occurred from excited Ir(III) complexes to DPA. A small difference in kq/kr was observed between the Δ- and Λ-enantiomers. It might be caused by the stereoselectivity effects in solvating a chiral complex in R-limonene. Besides, It was noted that the slope of a plot (KSV) was more than two times higher than that observed previously for the similar Ir(III) complex and DPA as a donor/acceptor pair in the medium of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) dichloromethane/methanol/water (or KSV = 600 M−1).24 The results reflected that R-limonene reduced substantially the competitive quenching by oxygen molecules.
1 (v/v) dichloromethane/methanol/water (or KSV = 600 M−1).24 The results reflected that R-limonene reduced substantially the competitive quenching by oxygen molecules.
| Ir(III) complexes | K SV or kq/kr (M−1) for Δ-isomer | K SV or kq/kr (M−1) for Λ-isomer | τ (μs) for Δ-isomerb | k q (M−1 s−1) for Δ-isomer |
|---|---|---|---|---|
| a The experimental conditions are described in the text. b The lifetime under air. | ||||
| [Ir(piq)2(dmbpy)]+ | 1554 | 1446 | 0.22 | 7.06 × 109 |
| [Ir(piq)2(Cnbpy)]+ (n = 9) | 2550 | 3276 | 0.27 | 9.44 × 109 |
| [Ir(piq)2(Cnbpy)]+ (n = 19) | 1457 | 1191 | 0.28 | 5.20 × 109 |
With a purpose of examining the effects of long alkyl chains, the same experiments were performed for a pair of [Ir(piq)2(Cnbpy)]+/DPA (n = 9 or 19) (Fig. S7 in SI). As summarized in Table 1, the presence of alkyl chains in donors affected little the efficiency of energy transfer. The UV-vis spectra were shown in Fig. S8 in SI.
The lifetime of a used complex (τ) was measured in the same medium (Fig. S9 in SI). Under air, τ is expressed by:
| τ = {kr + kqox[O2]}−1. | (3) |
| kq = KSV/τ. | (4) |
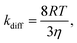 | (5) |
| kox[O2] = 1/τ − 1/τN. | (6) |
TTA-UC of photon energy by Ir(III) complexes in R-limonene
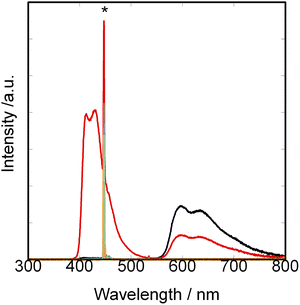
TTA-UC of photon energy in R-limonene was investigated for the pair of Δ- (or Λ)-[Ir(piq)2(dmbpy)]+ as a donor and DPA as an acceptor. Firstly, a CH2Cl2 solution containing the standard pair, [Ru(dmbpy)3]2+(dmbpy = 4,4′-dimethylbypyridine)/DPA, was irradiated at 445 nm under air using a laser pulse. The results of laser irradiation on the system are shown in SI. Next, an R-limonene solution of Δ- or Λ-[Ir(piq)2(dmbpy)]PF6 was irradiated under the same conditions. The results are shown in Fig. 2 and 3.In case of the pair of Δ- (or Λ)-[Ir(piq)2(dmbpy)]+/DPA, dual peaks appeared around 430 nm on adding DPA. The concomitant decrease of intensity was observed at the dual peaks around 590 nm (black and red curves in Fig. 2). The results indicated that TTA-UC of photon energy occurred for this pair. A reason for the dual nature of the induced UC band remained to be clarified. It might imply that the up-converted emission took place from two different singlet states in DPA. A reason was unclear, either, why the ratio of peak intensity of two dual peaks varied with the increase of concentration of added DPA as shown in Fig. S4 (SI).
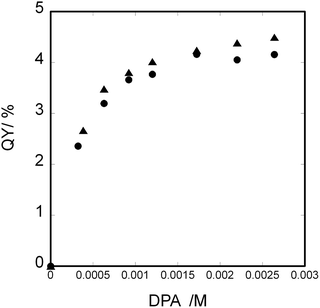
The quantum yield of up-conversion was calculated using the following equation;35
| Φ(uc,ref) = 2Φstd(Astd/Asam)(Istd/Isam)(Esam/Estd)(nsam/nstd)2 | (7) |
| Ir(III) complexes | Φ (uc,ref) (QY) for Δ-isomer (%) | Φ (uc,ref) (QY) for Λ-isomer (%) |
|---|---|---|
| [Ir(piq)2(dmbpy)]+ | 4.5 | 4.2 |
| [Ir(piq)2(Cnbpy)]+ (n = 9) | 3.6 | 3.3 |
| [Ir(piq)2(Cnbpy)]+ (n = 19) | 3.3 | 2.6 |
When the power of the laser light was varied, the quantum yield increased quadratically in the initial region, followed by linear increase in the high-laser-power region (Fig. S11 in SI). These results are in agreement with the known dependence of TTA-UC efficiency on laser power.15
The same experiments were performed for a pair of [Ir(piq)2(Cnbpy)]+/DPA (n = 9 or 19) as shown in Fig. S5 and S6 (SI). The results are also included in Table 2. It was concluded that the presence of long alkyl chains in a donor lowered the efficiency of TTA-UC. The presence of alkyl chains might interfere the proximity of the donor/acceptor pair in R-limonene.
Experimental section
Materials
A cationic cyclometalated Ir(III) complex, Δ-or Λ-[Ir(piq)2(Cnbpy)]+ (piqH = 1-phenyisoquinoline, Cnbpy = 4,4′-dialkyl-2,2′-bipyridine; dialkyl = dimethyl (C1), dinonyl (C9), and dinonyldecyl (C19) were prepared and optically resolved into Δ- and Λ-enantiomers, as described previously.26–28 The chromatogram in resolving racemic [Ir(piq)2(C19-bpy)]PF6 is shown in Fig. S1 (SI). The circular dichroism spectra of resolved [Ir(piq)2(dmbpy)]+, [Ir(piq)2(C9bpy)]+ and [Ir(piq)2(C19bpy)]+ are shown in Fig. S1 and S2 (SI). DPA was purchased from Tokyo Kasei Ind. Co. (Japan). R-limonene was purchased from FUJIFILM Wako Pure Chemical Corporation without purification.Instruments
UV-vis spectra were collected using a U-3810 spectrometer (Hitachi, Japan) or UV-vis spectrophotometer (V-730, JASCO Co., Japan). Electronic circular dichroism spectra were measured using a polarimeter J-720 (JAPSCO Co., Japan). An LSP-1000 instrument (Unisoku Co., Japan) was used to measure TTA-UC. A CW laser with an emission power of 0.0–51 mW was used for continuous irradiation at the excitation wavelength of 445 nm. Each sample (0.4 mL) was mounted into a glass tube with a diameter of 5 mm. Signals were accumulated by averaging five scans. Emission decay profile was monitored on irradiating a sample solution by a laser pulse at 355 nm. The emission signals at 600 nm were averaged for 128 pulses. The obtained curves were analyzed under the assumption of a single-exponential decay.Solubility measurements
1–5 mg of an enantiomeric or racemic Ir(III) complex as a PF6 salt was placed in a 5 mL vial. 1 mL of R-limonene was added and stirred. The sample was placed for one day at given temperature. The mixture was centrifuged. The residue of an unresolved complex was ascertained to be present at the bottom. The UV-vis spectrum of a filtrate was measured using a quartz cell of 2 mm optical path. The concentration of a solubilized iridium(III) complex was determined from the absorbance at 430 nm (ε = 4800).Conclusions
Triplet–triplet annihilation up-conversion (TTA-UC) was performed in R-limonene for the donor/acceptor pair of Δ- (or Λ)-[Ir(piq)2(dmbpy)]+/DPA. The chiral forms of the complex were soluble enough to detect emission. As a result, the UC quantum efficiency of 4.5% was achieved under air. No difference in efficiency was observed between the opposite enantiomers. Attaching two long alkyl chains to the iridium(III) complex increased solubility while decreasing the efficiency of TTA-UC by about 3%.Author contributions
Akihiko Yamagishi: writing – original draft, conceptualization, validation, methodology, investigation, and data curation. Jun Yoshida: writing – reviewing, and editing, conceptualization, validation, visualization, investigation, data curation, funding acquisition, and formal analysis. Hisako Sato: writing – original draft, conceptualization, data curation, methodology, investigation, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI) and the digital data of this study are available from the corresponding authors upon reasonable request. Supplementary information: chromatogram for optical resolution; electronic circular dichroism spectra; solubility; emission spectra of R-limonene solution containing Ir(III) complex and DPA under air; S–V plots of R-limonene solution; UV-vis spectra containing Ir(III) complex and DPA; lifetime of Ir(III) complexes; standard emission spectra; dependence of up-conversion efficiency on the intensity of laser light. See DOI: https://doi.org/10.1039/d5cp03773e.Acknowledgements
This work was financially supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI Grants (JP17H03044, JP20K21090, JP22H02033, JP23K13766, and JP23K04783).Notes and references
- G. Quaglia, F. Campana, L. Latterini and L. Vaccaro, ACS Sustainable Chem. Eng., 2022, 10, 9123 CrossRef CAS.
- J. Ma, S. Chen, C. Ye, M. Li, T. Liu, X. Wang and Y. Song, Phys. Chem. Chem. Phys., 2019, 21, 14516 RSC.
- M. R. Schreier, X. Guo, B. Pfund, Y. Okamoto, T. R. Ward, C. Kerzig and O. S. Wenger, Acc. Chem. Res., 2022, 55, 1290 CrossRef CAS PubMed.
- Y. Murakami, A. Motooka, R. Enomoto, K. Niimi, A. Kaiho and N. Kiyoyanagi, Phys. Chem. Chem. Phys., 2020, 22, 27134 RSC.
- N. Yanai and N. Kimizuka, Acc. Chem. Res., 2017, 50, 2487 CrossRef CAS PubMed.
- S. H. C. Askes and S. Bonnet, Nat. Rev. Chem., 2018, 2, 437 CrossRef.
- M. Uji, T. J. B. Záhringer, C. Kerzig and N. Yanai, Angew. Chem., Int. Ed., 2023, 62, e202301506 CrossRef CAS PubMed.
- T. Zhao and P. Duan, Angew. Chem., Int. Ed., 2024, 63, e202406524 Search PubMed.
- L. Huang and G. Han, Nat. Rev. Chem., 2024, 8, 238 Search PubMed.
- T. Zhao, J. Han, X. Qin, M. Zhou and P. Duan, J. Phys. Chem. Lett., 2024, 11, 311 CrossRef PubMed.
- W. Yao, X. Song, L. Xue, S. Liu, L. Tang, Y. Chen, H. Liu and X. Li, ChemPhotoChem, 2024, 8, e202400184 Search PubMed.
- L. Wei, C. Yang and W. Wu, Chem. Commun., 2025, 61, 7221 RSC.
- L. Ma, H. Guo, Q. Li, S. Guo and J. Zhao, Dalton Trans., 2012, 41, 10680 Search PubMed.
- Y. Nakadai, S. Tsuchiya, M. Uehara, S. Umezawa, R. Motoki, H. Umezawa, T. Ikoma and T. Yui, J. Phys. Chem. B, 2022, 126, 8245 Search PubMed.
- W. Zhao and F. N. Castellano, J. Phys. Chem. A, 2006, 110, 11440 CrossRef CAS PubMed.
- Y. Lu, J. Wang, N. McGoldrick, X. Cui, J. Zhao, C. Caverly, B. Twamley, G. M. Ó. Máille, B. Irwin, R. S. Conway-Kenny and M. Draper, Angew. Chem., Int. Ed., 2016, 55, 14688 Search PubMed.
- I. S. Alkhaibari, X. Zhang, J. Zhao, T. M. Stonelake, R. C. Knighton, P. N. Horton, S. J. Coles, N. J. Buurma, E. Richards and S. J. A. Pope, Inorg. Chem., 2024, 63, 9931 Search PubMed.
- P. Duan, N. Yanai and N. Kimizuka, Chem. Commun., 2014, 50, 13111 RSC.
- C. E. Elgar, H. Y. Otaif, X. Zhang, J. Zhao, P. N. Horton, S. J. Coles, J. M. Beames and S. J. A. Pope, Eur. J. Chem., 2021, 27, 3427 Search PubMed.
- X. Jiang, S. Guo, J. Peng, D. Zhao and Y. Ma, ACS Appl. Mater. Interfaces, 2016, 8, 11441 Search PubMed.
- X. Guo, Q. Chen, Y. Tong, Y. Li, Y. Liu, D. Zhao and Y. Ma, J. Phys. Chem. A, 2018, 122, 6963 CrossRef CAS PubMed.
- T. Zhao, J. Han, X. Qin, M. Zhou and P. Duan, J. Phys. Chem. Lett., 2020, 11, 311 CrossRef CAS PubMed.
- W. Wu, W. Wu, S. Ji, H. Guo and J. Zhao, Dalton Trans., 2011, 40, 5953 Search PubMed.
- A. Yamagishi, K. Takimoto, R. Ito, J. Yoshida and H. Sato, Appl. Clay Sci., 2025, 272, 107828 CrossRef CAS.
- A. Yamagishi, K. Tamura, S. Yamamoto, F. Sato, J. Yoshida and H. Sato, Appl. Clay Sci., 2024, 255, 107397 Search PubMed.
- H. Sato, K. Tamura, M. Taniguchi and A. Yamagishi, Appl. Clay Sci., 2014, 97–98, 84 Search PubMed.
- H. Sato, K. Tamura, K. Ohara and S.-I. Nagaoka, New J. Chem., 2014, 38, 132 RSC.
- K. Tamura, A. Yamagishi, T. Kitazawa and H. Sato, Phys. Chem. Chem. Phys., 2015, 17, 18288 RSC.
- X. Chen, Y. Okamoto, T. Yano and J. Otsuki, J. Sep. Sci., 2007, 30, 713 CrossRef CAS PubMed.
- D. R. Martir, C. Momblona, A. Pertegás, D. B. Cordes, A. M. Z. Slawin, H. J. Bolink and E. Zysman-Colman, ACS Appl. Mater. Interfaces, 2016, 8, 33907 CrossRef CAS PubMed.
- A. Kanbe, K. Yokoi, Y. Yamada, M. Tsurui, Y. Kitagawa, Y. Hasegawa, D. Ogata, J. Yuasa and S. Aoki, Inorg. Chem., 2023, 62, 11325 Search PubMed.
- I. Kalf, B. Calmuschi and U. Englert, CrystEngComm, 2002, 4, 548–551 RSC.
- C. P. Broch, W. B. Schweizer and J. D. Dunitz, J. Am. Chem. Soc., 1991, 113, 9811 Search PubMed.
- R. A. Clará, A. C. G. Marigliano and H. N. Sólimo, J. Chem. Eng. Data, 2009, 54, 1087 CrossRef.
- T. N. Singh-Rachford and F. N. Castellano, Coord. Chem. Rev., 2010, 254, 2560 CrossRef CAS.
| This journal is © the Owner Societies 2026 |