Themed collection Metalloenzymes in Natural Product Biosynthetic Pathways

Front cover

Front cover

Metalloenzymes in natural product biosynthetic pathways
The Natural Product Reports themed issue on ‘Metalloenzymes in natural product biosynthetic pathways’ is introduced by the Guest Editors, Katherine Ryan and Catherine Drennan.

Nat. Prod. Rep., 2018,35, 612-614
https://doi.org/10.1039/C8NP90023J
Following the electrons: peculiarities in the catalytic cycles of radical SAM enzymes
Electrons do not always go where you expect them to in the catalytic cycles of radical SAM enzymes. Herein, a number of examples are considered along with potential avenues for future investigation.
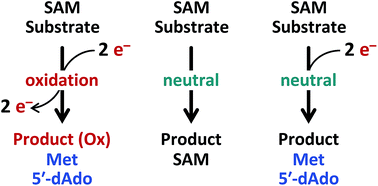
Nat. Prod. Rep., 2018,35, 615-621
https://doi.org/10.1039/C7NP00058H
Cobalamin-dependent radical S-adenosyl-L-methionine enzymes in natural product biosynthesis
This highlight examines the functions of cobalamin-dependent radical S-adenosyl-L-methionine enzymes that catalyse chemically-challenging reactions in several bacterial natural product biosynthetic pathways.
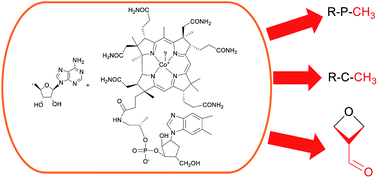
Nat. Prod. Rep., 2018,35, 707-720
https://doi.org/10.1039/C7NP00059F
Expanding the roles for 2-oxoglutarate-dependent oxygenases in plant metabolism
This review of 2-oxoglutarate-dependent oxygenases and other oxidases involved in plant metabolism provides a framework for continued enzyme discovery.
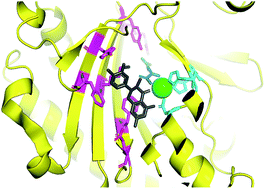
Nat. Prod. Rep., 2018,35, 721-734
https://doi.org/10.1039/C7NP00060J
Rieske non-heme iron-dependent oxygenases catalyse diverse reactions in natural product biosynthesis
The role played by Rieske non-heme iron-dependent oxygenases in natural product biosyntheses is reviewed, with particular focus on experimentally characterised examples.
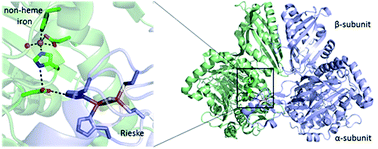
Nat. Prod. Rep., 2018,35, 622-632
https://doi.org/10.1039/C8NP00004B
Metabolic functions of the human gut microbiota: the role of metalloenzymes
Metalloenzymes play central roles in metabolic functions of the human gut microbiota that are associated with host health and disease.
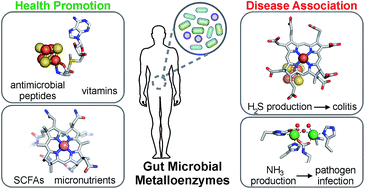
Nat. Prod. Rep., 2019,36, 593-625
https://doi.org/10.1039/C8NP00074C
Recent examples of α-ketoglutarate-dependent mononuclear non-haem iron enzymes in natural product biosyntheses
Proximal- and distal-type αKG binding to the Fe(II) centre might play a crucial role in fine-tuning the catalysis of αKG-dependent non-haem iron enzymes.
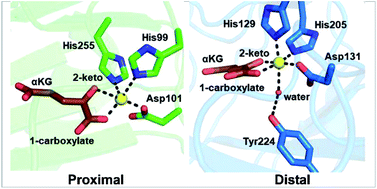
Nat. Prod. Rep., 2018,35, 792-837
https://doi.org/10.1039/C7NP00067G
Roles of 2-oxoglutarate oxygenases and isopenicillin N synthase in β-lactam biosynthesis
The 2OG oxygenases and IPNS contribute to the great structural diversity of β-lactam natural products, employing some remarkable mechanisms.
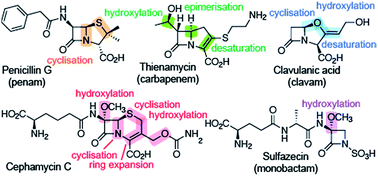
Nat. Prod. Rep., 2018,35, 735-756
https://doi.org/10.1039/C8NP00002F
Unrivalled diversity: the many roles and reactions of bacterial cytochromes P450 in secondary metabolism
This review highlights the scope of chemical transformations that cytochrome P450 enzymes catalyse within bacterial secondary metabolism.
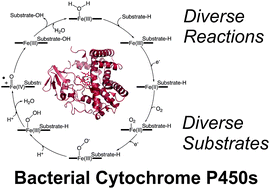
Nat. Prod. Rep., 2018,35, 757-791
https://doi.org/10.1039/C7NP00063D
C–C bond forming radical SAM enzymes involved in the construction of carbon skeletons of cofactors and natural products
An emerging group of radical SAM enzymes that catalyze C–C bond formations in natural product and cofactor biosynthesis are discussed.
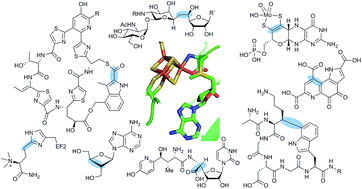
Nat. Prod. Rep., 2018,35, 660-694
https://doi.org/10.1039/C8NP00006A
Diiron monooxygenases in natural product biosynthesis
Two new families of diiron cluster-containing oxygenases serve as tailoring enzymes for NRPS and PKS biosynthetic systems.
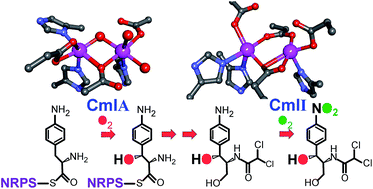
Nat. Prod. Rep., 2018,35, 646-659
https://doi.org/10.1039/C7NP00061H
Unique chemistry of non-heme iron enzymes in fungal biosynthetic pathways
Reactions by non-heme iron enzymes in structurally intriguing fungal natural products pathways are summarized and discussed.
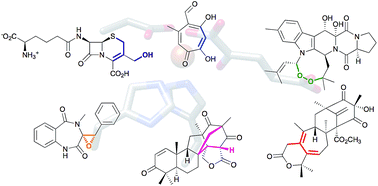
Nat. Prod. Rep., 2018,35, 633-645
https://doi.org/10.1039/C7NP00055C
Cytochromes P450 for natural product biosynthesis in Streptomyces: sequence, structure, and function
This review catalogues functionally characterized P450s from Streptomyces and discusses their sequences, structures, and functions in natural products biosynthesis.
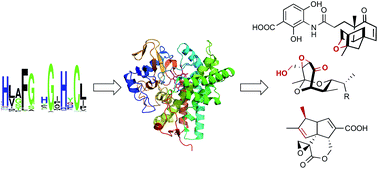
Nat. Prod. Rep., 2017,34, 1141-1172
https://doi.org/10.1039/C7NP00034K
About this collection
Natural Product Reports is delighted to present a themed issue dedicated to the role of metal-containing enzymes in natural product biosynthetic pathways, Guest Edited by Professors Catherine Drennan (MIT) and Katherine Ryan (The University of British Columbia).
Metal-containing enzymes are critical for catalysing challenging chemical transformations needed to build up structurally diverse natural products. In this themed issue, we will highlight the major classes of metalloenzymes that are used in natural products biosynthesis, including cytochrome P450s, radical SAM-dependent enzymes, and iron(II) α-keto-glutarate-dependent enzymes. These reviews discuss these enzymes in the context of pathways found in bacteria, fungi, and plants. The reviews also discuss the data key to unveiling the mechanisms of these enzymes and efforts to expand the chemistry of metalloenzymes from natural product biosynthesis using engineering approaches.