Themed collection Identification and Optimization of GPCR Ligands in the 21st Century

Expanding the horizons of G protein-coupled receptor structure-based ligand discovery and optimization using homology models
We show the key role of structural homology models in GPCR structure-based lead discovery and optimization, highlighting methodological aspects, recent progress and future directions.
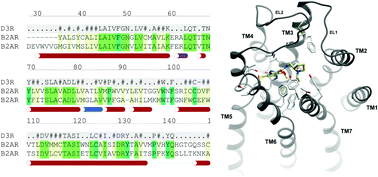
Chem. Commun., 2015,51, 13576-13594
https://doi.org/10.1039/C5CC05050B
Insights into a defined secondary binding region on β-adrenoceptors and putative roles in ligand binding and drug design
Putative roles of a secondary binding region shared among beta-adrenoceptors.
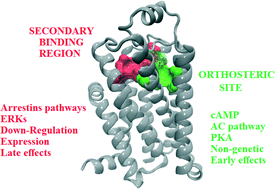
Med. Chem. Commun., 2015,6, 991-1002
https://doi.org/10.1039/C5MD00011D
Structural basis for Na+-sensitivity in dopamine D2 and D3 receptors
To understand the structural basis for the Na+-sensitivity of ligand binding to dopamine D2-like receptors, using computational analysis in combination with binding assays, we identified interactions critical in propagating the impact of Na+ on receptor conformations and on the ligand-binding site.
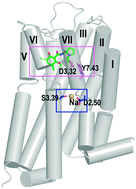
Chem. Commun., 2015,51, 8618-8621
https://doi.org/10.1039/C5CC02204E
Triblock peptide–linker–lipid molecular design improves potency of peptide ligands targeting family B G protein-coupled receptors
Design and characterization of triblock peptide–linker–lipid constructs for targeting family B G protein-couple receptors with improved bioactivity and biostability.
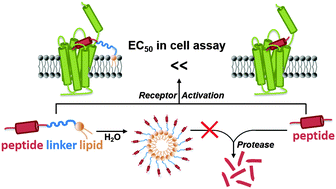
Chem. Commun., 2015,51, 6157-6160
https://doi.org/10.1039/C5CC00301F
Free energy calculations of A2A adenosine receptor mutation effects on agonist binding
A general computational scheme to evaluate the effects of single point mutations on ligand binding is reported.
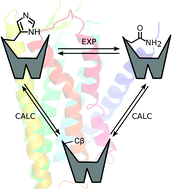
Chem. Commun., 2015,51, 3522-3525
https://doi.org/10.1039/C4CC09517K
Exploring the recognition pathway at the human A2A adenosine receptor of the endogenous agonist adenosine using supervised molecular dynamics simulations
The putative adenosine-hA2A AR recognition pathway is suggested by a series of Supervised Molecular Dynamics (SuMD) simulations.
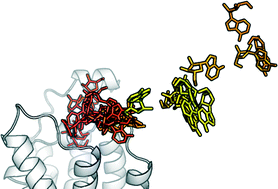
Med. Chem. Commun., 2015,6, 1081-1085
https://doi.org/10.1039/C5MD00016E
Structure-based virtual screening for fragment-like ligands of the G protein -coupled histamine H4 receptor
Structure-based virtual screening using H1R- and β2R-based histamine H4R homology models identified 9 fragments with an affinity ranging from 0.14 to 6.3 μm for H4R.

Med. Chem. Commun., 2015,6, 1003-1017
https://doi.org/10.1039/C5MD00022J
New substituted 9-propyladenine derivatives as A2Aadenosine receptor antagonists
New 9-propyladenines substituted at 2- or N6- and 8 positions behave as adenosine receptor antagonists with low nM affinity at the A2A subtype.
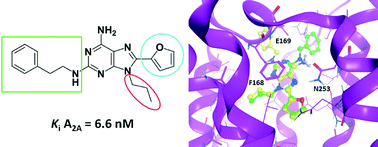
Med. Chem. Commun., 2015,6, 963-970
https://doi.org/10.1039/C5MD00034C
Discovery of HTL6641, a dual orexin receptor antagonist with differentiated pharmacodynamic properties
A novel series of potent, selective, and orally efficacious dual antagonists of the orexin receptors is described.

Med. Chem. Commun., 2015,6, 947-955
https://doi.org/10.1039/C5MD00027K
Structure-based design, synthesis by click chemistry and in vivo activity of highly selective A3 adenosine receptor agonists
C2-phenylethynyl (dark green) and C2-phenyl-triazolyl (light green) nucleosides in the A3AR.
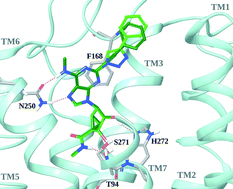
Med. Chem. Commun., 2015,6, 555-563
https://doi.org/10.1039/C4MD00571F
About this collection
MedChemComm and ChemComm are delighted to publish a collection of articles on the identification and optimization of GPCR ligands in the 21st century.
In the last few years the structural studies of G protein-coupled receptors (GPCR) have blossomed, shedding light on the structure and the functioning of the receptors and providing an increasingly more solid platform for the rational discovery and design of novel GPCR ligands.
This collection of articles, guest edited by Stefano Costanzi (American University), Miles Congreve (Heptares Therapeutics) and Christa Müller (University of Bonn) is a celebration of current achievements and future perspectives in this exciting field of research.
New articles will be added to this collection as they are published.
Please return to this page frequently to see the collection grow.