Themed collection Microphysiological systems in pharmaceutical safety and ADME applications

Introduction to a manuscript series on the characterization and use of microphysiological systems (MPS) in pharmaceutical safety and ADME applications
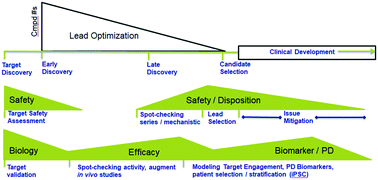
Opportunities in the drug discovery/development process for potential MPS incorporation.
Lab Chip, 2020,20, 1049-1057
https://doi.org/10.1039/C9LC01168D
Cardiovascular microphysiological systems (CVMPS) for safety studies – a pharma perspective
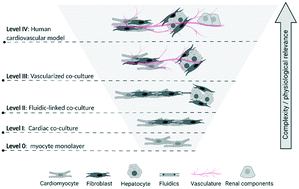
The integrative responses of the cardiovascular (CV) system are essential for maintaining blood flow to provide oxygenation, nutrients, and waste removal for the entire body.
Lab Chip, 2021,21, 458-472
https://doi.org/10.1039/D0LC01040E
Application of microphysiological systems in biopharmaceutical research and development
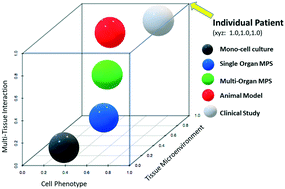
In this review, we describe some of the advances made for tissue-specific microphysiological systems and outline the advantages and challenges of applying and further developing MPS technology in preclinical biopharmaceutical research.
Lab Chip, 2020,20, 697-708
https://doi.org/10.1039/C9LC00962K
Microphysiological systems for ADME-related applications: current status and recommendations for system development and characterization
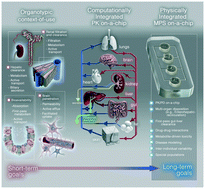
Potential applications of MPS in the ADME discipline.
Lab Chip, 2020,20, 446-467
https://doi.org/10.1039/C9LC00857H
Drug-induced skin toxicity: gaps in preclinical testing cascade as opportunities for complex in vitro models and assays
Selected skin MPS features desired to advance further adoption within the pharmaceutical industry.

Lab Chip, 2020,20, 199-214
https://doi.org/10.1039/C9LC00519F
Microphysiological lung models to evaluate the safety of new pharmaceutical modalities: a biopharmaceutical perspective
Drug developers seek specific advancements in the development and qualification of microphysiological lung models for the evaluation of drug safety; here these essential elements are discussed from the perspective of the biopharmaceutical industry.

Lab Chip, 2019,19, 3152-3161
https://doi.org/10.1039/C9LC00492K
Developing in vitro assays to transform gastrointestinal safety assessment: potential for microphysiological systems
Drug-induced gastrointestinal toxicities (DI-GITs) are among the most common adverse events in clinical trials.
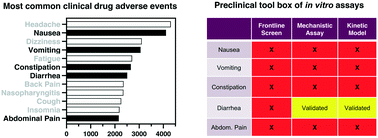
Lab Chip, 2020,20, 1177-1190
https://doi.org/10.1039/C9LC01107B
A pharmaceutical industry perspective on microphysiological kidney systems for evaluation of safety for new therapies
The human kidney contains approximately one million nephrons.
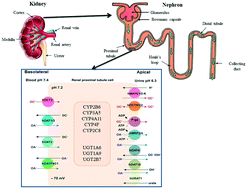
Lab Chip, 2020,20, 468-476
https://doi.org/10.1039/C9LC00925F
Liver microphysiological systems development guidelines for safety risk assessment in the pharmaceutical industry
This pharmaceutical industry guidance based on a 3-staged benchmarking strategy aims to help MPS developers and end users identify what could be the most valuable models for safety risk assessment, as well as provide an overview of contexts of use.
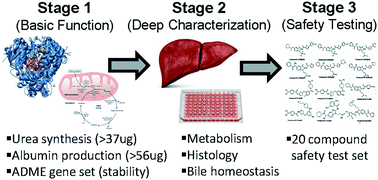
Lab Chip, 2020,20, 215-225
https://doi.org/10.1039/C9LC00768G
About this collection
The International Consortium for Innovation and Quality in Pharmaceutical Development (IQ) MPS Affiliate is a group of pharmaceutical industry scientists who seek to expedite appropriate characterization and incorporation of MPS to potentially improve drug safety assessment and provide safer and more effective medicines to patients. In keeping with this mission, the IQ MPS Affiliate scientists have prepared a series of organotypic manuscripts for several key drug safety and disposition target tissues (lung, liver, kidney, skin, gastrointestinal, cardiovascular, and blood brain barrier/central nervous system). The goal of these manuscripts is to provide key information related to likely initial contexts of use (CoU) and key characterization data needed for incorporation of MPS in pharmaceutical safety screening including a list of characteristic functions, cell types, toxicities, and test agents (representing major mechanisms of toxicity) that can be used by MPS developers. Additional manuscripts focusing on testing biologically based therapeutics and ADME considerations have been prepared as part of this effort. These manuscripts focus on general needs for assessing biologics and ADME endpoints and include similar information to the tissue specific manuscripts where appropriate. This collection has been coordinated by Kristin Fabre and Terry Van Vleet.