Themed collection Nanostructures in Ionic Liquids

Plasma electrochemistry in ionic liquids : an alternative route to generate nanoparticles
In this perspective we report on the application of plasma electrochemistry for the generation of nanoparticles in ionic liquids.

Phys. Chem. Chem. Phys., 2011,13, 13472-13478
https://doi.org/10.1039/C1CP20501C
Shape-controlled synthesis of silver crystals mediated by imidazolium-based ionic liquids
A shape-controlled synthetic route to Ag crystals is developed by using ionic liquids (ILs) as a shape-regulating agent.
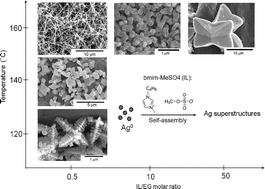
Phys. Chem. Chem. Phys., 2011,13, 16138-16141
https://doi.org/10.1039/C1CP20582J
Preparation of polymer -supported gold nanoparticles based on resins containing ionic liquid -like fragments: easy control of size and stability
Gold nanoparticles (AuNP–ILLPs) immobilised onto Supported Ionic Liquid-like Phases (SILLPs) are easily synthesised. Their size and morphology can be easily tuned by the structural elements of the SILLPs
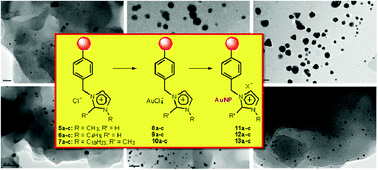
Phys. Chem. Chem. Phys., 2011,13, 14831-14838
https://doi.org/10.1039/C1CP20970A
Size and shape of Au nanoparticles formed in ionic liquids by electron beam irradiation
The electron beam conditions (acceleration energy, beam current and irradiation time) and the combination of cations and anions of ionic liquid affect the size and shape of Au particles.
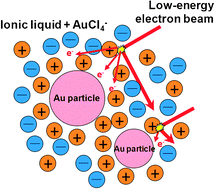
Phys. Chem. Chem. Phys., 2011,13, 14823-14830
https://doi.org/10.1039/C1CP20643E
Anion effect on the shape evolution of gold nanoparticles during seed-induced growth in imidazolium-based ionic liquids
The efficiency of imidazolium-based cations to induce shape-anisotropic growth of gold nanocrystals in weak reducing conditions is found to depend sensitively on anion characteristics.
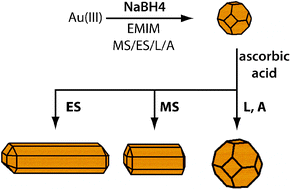
Phys. Chem. Chem. Phys., 2011,13, 13572-13578
https://doi.org/10.1039/C1CP20518H
A smart palladium catalyst in ionic liquid for tandem processes
Palladium species in IL containing MeHPO3− as anion becomes a multi-function catalyst for giving fragrance precursors (II) in one-pot synthesis by means of a coupling/reduction tandem process.
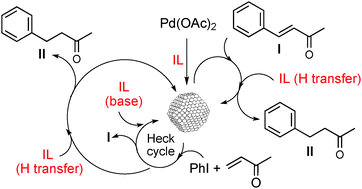
Phys. Chem. Chem. Phys., 2011,13, 13579-13584
https://doi.org/10.1039/C1CP20619B
Ionic liquids as recycling solvents for the synthesis of magnetic nanoparticles
MFe2O4 (M = Fe, Co, Mn, and Ni) nanoparticles were synthesized by thermal decomposition of Fe(Acac)3/M(Acac)2 in imidazolium ionic liquids.

Phys. Chem. Chem. Phys., 2011,13, 13558-13564
https://doi.org/10.1039/C1CP21518C
Modulating the immobilization process of Au nanoparticles on TiO2(110) by electrostatic interaction between the surface and ionic liquids
We found that the adsorption states of Au NPs on TiO2(110) varied with the ILs that encapsulate the NPs during the entire process from synthesis of NPs to immobilization of the NPs on the surface at 323 K.
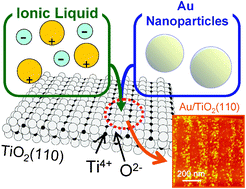
Phys. Chem. Chem. Phys., 2011,13, 13585-13593
https://doi.org/10.1039/C1CP20814D
On the formation of anisotropic gold nanoparticles by sputtering onto a nitrile functionalised ionic liquid
Sputtering deposition of gold onto the 1-(butyronitrile)-3-methylimidazolium bis(trifluoromethylsulfonyl)imide (BCN)MI·N(Tf)2 ionic liquid (IL) has generated colloidal and stable gold nanospheres (AuNS) and gold nanodisks (AuND).
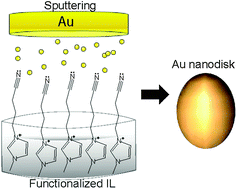
Phys. Chem. Chem. Phys., 2011,13, 13552-13557
https://doi.org/10.1039/C1CP21406C
Growth of sputter-deposited gold nanoparticles in ionic liquids
In situ growth of gold nanoparticles prepared by sputter deposition in the bulk phase of ionic liquids.
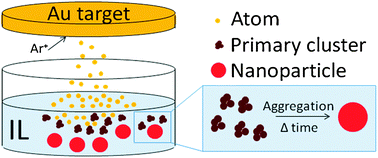
Phys. Chem. Chem. Phys., 2011,13, 13565-13571
https://doi.org/10.1039/C1CP20552H
Pronounced sponge-like nanostructure in propylammonium nitrate
The bulk structure of the ionic liquid propylammonium nitrate (PAN) has been determined using neutron diffraction experiments and empirical potential structure refinement fits to the data.

Phys. Chem. Chem. Phys., 2011,13, 13544-13551
https://doi.org/10.1039/C1CP21080G
N-Donor ligands based on bipyridine and ionic liquids: an efficient partnership to stabilize rhodium colloids. Focus on oxygen -containing compounds hydrogenation
Various poly-N-donor ligands have proved to be efficient extra protective agents for the stabilization of Rh(0) nanospecies in [BMI][PF6]. A significant ratio of ketone derivatives was observed in the hydrogenation of anisole and cresols as model lignin compounds.
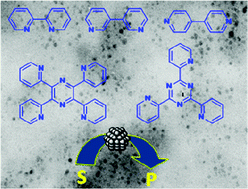
Phys. Chem. Chem. Phys., 2011,13, 13510-13517
https://doi.org/10.1039/C1CP20602H
Nanostructure changes in protic ionic liquids (PILs) through adding solutes and mixing PILs
The effect of adding primary n-alcohols and hexane on the nanostructure of protic ionic liquids (PILs), and mixing PILs, was explored.
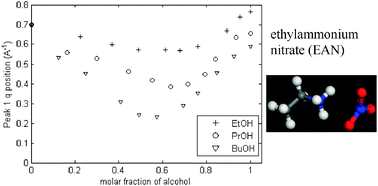
Phys. Chem. Chem. Phys., 2011,13, 13501-13509
https://doi.org/10.1039/C1CP20496C
Effect of alkyl chain length and hydroxyl group functionalization on the surface properties of imidazolium ionic liquids
Snapshot of the surface of the ionic liquid [C8OHC1im][BF4] obtained using molecular simulation (OH group in red).
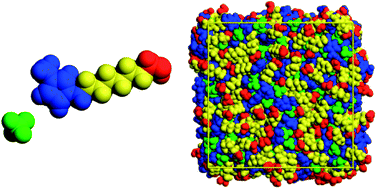
Phys. Chem. Chem. Phys., 2011,13, 13518-13526
https://doi.org/10.1039/C1CP20563C
On the interaction of ascorbic acid and the tetrachlorocuprate ion [CuCl4]2− in CuCl nanoplatelet formation from an ionic liquid precursor (ILP)
Addition of ascorbic acid to the ionic liquid butylpyridinium tetrachlorocuprate [C4Py]2[CuCl4] leads to the removal of one chloride from the [CuCl4]2− anion, leading to an intermediate IL/ascorbic acid adduct that is key for the formation of CuCl nanoplatelets from the IL.
![Graphical abstract: On the interaction of ascorbic acid and the tetrachlorocuprate ion [CuCl4]2− in CuCl nanoplatelet formation from an ionic liquid precursor (ILP)](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C1CP20648F&imageInfo.ImageIdentifier.Year=2011)
Phys. Chem. Chem. Phys., 2011,13, 13537-13543
https://doi.org/10.1039/C1CP20648F
Ruthenium
nanoparticles in ionic liquids : structural and stability effects of polar solutes
Structural and stability effects of 1-octylamine and water in the synthesis of RuNPs were investigated by NMR, mixing calorimetry, and molecular simulations.
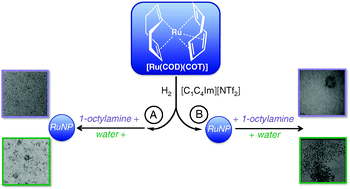
Phys. Chem. Chem. Phys., 2011,13, 13527-13536
https://doi.org/10.1039/C1CP20623K
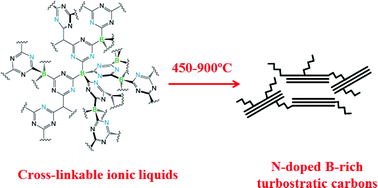
Boron and nitrogen -rich carbons from ionic liquid precursors with tailorable surface properties
Nitrogen-doped boron-rich carbons with slit-like pores, high surface areas and exhibiting high capacity for CO2 adsorption are prepared from ionic liquids containing cross-linkable anions at various carbonization temperatures.
Phys. Chem. Chem. Phys., 2011,13, 13486-13491
https://doi.org/10.1039/C1CP20631A
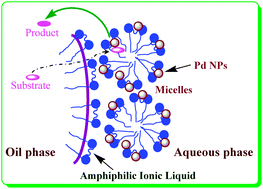
Amphiphilic ionic liquid stabilizing palladium nanoparticles for highly efficient catalytic hydrogenation
The amphiphilic ionic liquids (C12Im-PEG) stabilizing palladium nanoparticles (NPs) have been utilized for efficiently catalytic hydrogenation under very mild conditions.
Phys. Chem. Chem. Phys., 2011,13, 13492-13500
https://doi.org/10.1039/C1CP20255C
Interactions of adsorbed poly(ethylene oxide) mushrooms with a bare silica–ionic liquid interface
Colloid probe atomic force microscope measurements in an asymmetric geometry are used to investigate forces acting on poly(ethylene oxide) mushrooms in contact with an ionic liquid–silica interface.

Phys. Chem. Chem. Phys., 2011,13, 13479-13485
https://doi.org/10.1039/C1CP20174C
About this collection
PCCP is delighted to present a high-profile themed issue on Nanostructures in Ionic Liquids, Guest Edited by Jairton Dupont (UFRGS, Brazil) and Rob Atkin (University of Newcastle, Australia).