Themed collection Enzymes and Proteins

Electrostatic effects on (di)terpene synthase product outcome
Effect of specifically located hydroxyl group in counter-acting effect of pyrophosphate anion co-product in steering carbocation migration towards itself (e.g. by further cyclization, as shown).
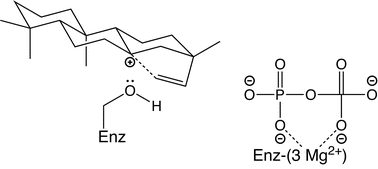
Chem. Commun., 2011,47, 4074-4080
https://doi.org/10.1039/C0CC02960B
Cytochromes P450 as useful biocatalysts: addressing the limitations
Factors limiting the exploitation of cytochrome P450s and significant steps which have been taken towards addressing these limitations are discussed.
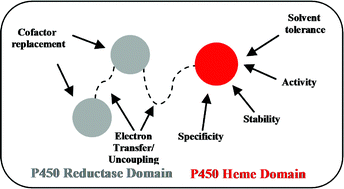
Chem. Commun., 2011,47, 2490-2501
https://doi.org/10.1039/C0CC03165H
Probing the stereoselectivity of P-glycoprotein—synthesis, biological activity and ligand docking studies of a set of enantiopure benzopyrano[3,4-b][1,4]oxazines
Enantiomerically pure benzopyrano[3,4-b][1,4]oxazines show significant differences in their inhibitory potency of P-glycoprotein. Docking studies provide first insights into potential binding sites for these compounds.
![Graphical abstract: Probing the stereoselectivity of P-glycoprotein—synthesis, biological activity and ligand docking studies of a set of enantiopure benzopyrano[3,4-b][1,4]oxazines](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C0CC03075A&imageInfo.ImageIdentifier.Year=2011)
Chem. Commun., 2011,47, 2586-2588
https://doi.org/10.1039/C0CC03075A
A new dehydrogenase from Clostridium acetobutylicum for asymmetric synthesis: dynamic reductive kinetic resolution entry into the Taxotère side chain
Asymmetric carbonyl reduction using a new Clostridium acetobutylicum ADH provides access to diverse value-added chiral building blocks using biorenewable redox equivalents.
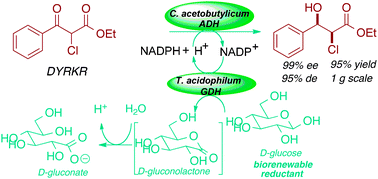
Chem. Commun., 2011,47, 2420-2422
https://doi.org/10.1039/C0CC04585C
Incorporation of chlorinated analogues of aliphatic amino acids during cell-free protein synthesis
Analogues of Val and Leu having a chloro substituent in place of a methyl group are efficiently incorporated into protein.
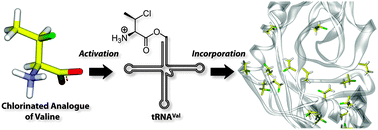
Chem. Commun., 2011,47, 1839-1841
https://doi.org/10.1039/C0CC02879G
Competing SN2 and E2 reaction pathways for hexachlorocyclohexane degradation in the gas phase, solution and enzymes
Quantum chemistry calculations reveal that the isomer specificity of enzymes that break down hexachlorocyclohexane is constrained by the intrinsic substrate reactivity.
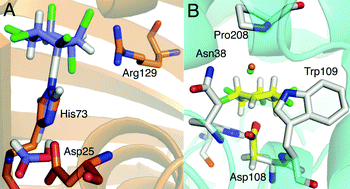
Chem. Commun., 2011,47, 976-978
https://doi.org/10.1039/C0CC02925D
Ribonuclease S redux
Modern methods ease the use and enhance the versatility of an historical protein-fragment complementation system.

Chem. Commun., 2011,47, 973-975
https://doi.org/10.1039/C0CC03864D
Screening of a minimal enriched P450 BM3 mutant library for hydroxylation of cyclic and acyclic alkanes
Several single and double mutants from a minimal cytochrome P450 BM3 library were found to effectively oxidize cyclic and acyclic alkanes. For example, the F87A/A328V mutant hydroxylates cyclododecane to cyclododecanol.
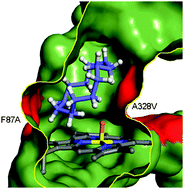
Chem. Commun., 2011,47, 944-946
https://doi.org/10.1039/C0CC02924F
Biocompatible functionalisation of starch
A combination of enzymatic synthesis and click chemistry is employed for the production of starch derivatives under mild, aqueous reaction conditions.
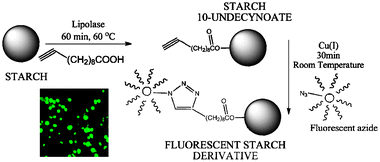
Chem. Commun., 2011,47, 683-685
https://doi.org/10.1039/C0CC02908D
A fast and sensitive assay for measuring the activity and enantioselectivity of transaminases
A fast and sensitive method for screening transaminase activity and enantioselectivity, using D- and L-amino acid oxidases, allows new amine substrates to be rapidly identified.
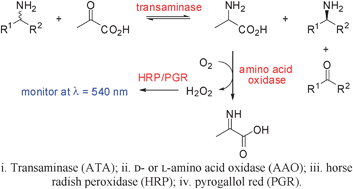
Chem. Commun., 2011,47, 773-775
https://doi.org/10.1039/C0CC02919J
Micelle to fibre biocatalytic supramolecular transformation of an aromatic peptide amphiphile
A mechanistic insight into a phosphatase-driven supramolecular transformation whereby a peptide amphiphile switches from micellar structure to a chiral nanofibre morphology.
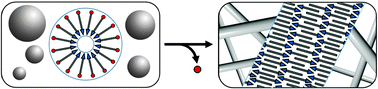
Chem. Commun., 2011,47, 728-730
https://doi.org/10.1039/C0CC03796F
Photochemical cleavage of leader peptides
A photolabile linker is described for light-mediated removal of leader peptides involved in biosynthesis of posttranslationally modified peptide natural products.
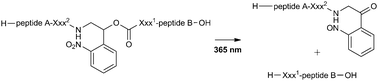
Chem. Commun., 2010,46, 8935-8937
https://doi.org/10.1039/C0CC02945A
Probing the proteasome cavity in three steps: bio-orthogonal photo-reactive suicide substrates
Tri-functional activity-based proteasome probes are used for a three-step chemical proteomics approach, in which proteasomal catalytic sites are covalently and irreversibly modified, followed by photocrosslinking of these to flanking subunits and Staudinger–Bertozzi ligation for visualization and identification of the resulting conjugates.
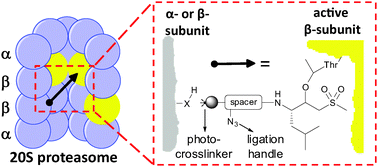
Chem. Commun., 2010,46, 9052-9054
https://doi.org/10.1039/C0CC03322G
Selective labeling of polypeptides using protein farnesyltransferase via rapid oxime ligation
An aldehyde-containing substrate for protein farnesyltransferase was shown to be enzymatically incorporated into a protein. The protein was subsequently immobilized onto aminooxy-functionalized agarose beads or labeled with a fluorophore. This method provides an alternative to the commonly employed Cu(I)-catalyzed click reaction.
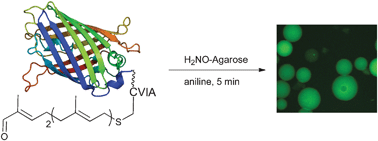
Chem. Commun., 2010,46, 8998-9000
https://doi.org/10.1039/C0CC03305G
MhyADH catalysed Michael addition of water and in situoxidation
MhyADH catalyses the Michael addition of water and also the subsequent oxidation to a diketone. In the absence of an oxidation reagent the reaction stops after the Michael addition.
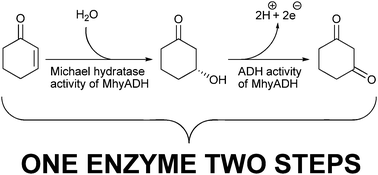
Chem. Commun., 2010,46, 8588-8590
https://doi.org/10.1039/C0CC03229H
Towards preparative-scale, biocatalytic alkene reductions
Reaction strategies for employing two complementary alkene reductase enzymes in laboratory-scale conversions have been developed.
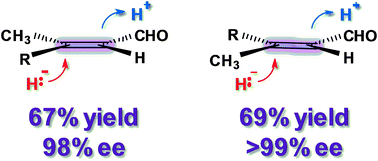
Chem. Commun., 2010,46, 8558-8560
https://doi.org/10.1039/C0CC03119D
Increasing the stability of an enzyme toward hostile organic solvents by directed evolution based on iterative saturation mutagenesis using the B-FIT method
Mutants of Bacillus subtilis lipase evolved for enhanced thermostability also show increased stability in organic solvents such as acetonitrile (ACN).
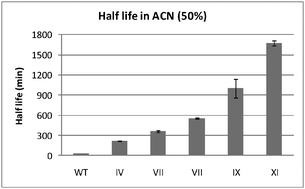
Chem. Commun., 2010,46, 8657-8658
https://doi.org/10.1039/C0CC02657C
Tandem driven dynamic self-inhibition of acetylcholinesterase
A concept of tandem driven dynamic enzyme self-inhibition is demonstrated for acetylcholinesterase through the action of dynamic stealth inhibitors using reversible transthiolesterification.
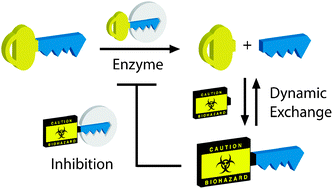
Chem. Commun., 2010,46, 8457-8459
https://doi.org/10.1039/C0CC02479A
Profiling small molecule inhibitors against helix–receptor interactions: the Bcl-2 family inhibitor BH3I-1 potently inhibits p53/hDM2
We validate a practical methodology for the rapid profiling of small molecule inhibitors of protein–protein interactions. We find that a well known BH3 family inhibitor can potently inhibit the p53/hDM2 interaction.
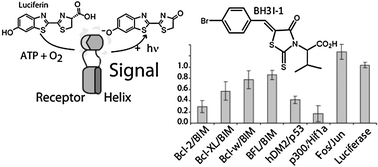
Chem. Commun., 2010,46, 8020-8022
https://doi.org/10.1039/C0CC02969F
Cyclen-hybrid compound captures copper to protect INS-1 cells from islet amyloid polypeptide cytotoxicity by inhibiting and lysing effects
I1–Cu(II) inhibits hIAPP cytotoxicity by interfering with aggregation and cleavage.
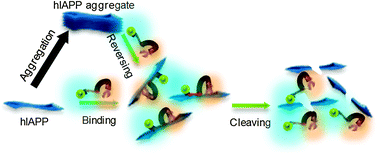
Chem. Commun., 2010,46, 8023-8025
https://doi.org/10.1039/C0CC02555K
Simultaneous iridium catalysed oxidation and enzymatic reduction employing orthogonal reagents
Employing orthogonal reaction conditions an iridium catalysed oxidative hydrogen transfer was successfully coupled concurrently to an asymmetric enzyme catalysed reduction.
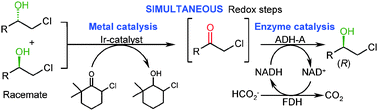
Chem. Commun., 2010,46, 8046-8048
https://doi.org/10.1039/C0CC02813D
Structural and biochemical basis for the firm chemo- and regioselectivity of the nitro -forming N-oxygenase AurF
Variants of the N-oxygenase AurF from Streptomyces thioluteus selectively transform guanidyl- and amidinyl-substituted anilines into the corresponding nitro compounds. Site-directed mutagenesis and structural data provide new insights into regioselective N-oxygenation.
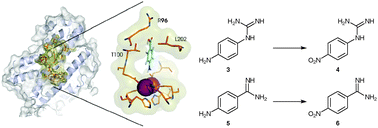
Chem. Commun., 2010,46, 7760-7762
https://doi.org/10.1039/C0CC02811H
Fluorinase mediated chemoenzymatic synthesis of [18F]-fluoroacetate
A novel preparation of sodium [18F]-fluoroacetate is described by a fluorinase catalysed reaction of S-adenosyl-L-methionine 1 with no carrier added [18F]-fluoride and then the [18F]-2 product is oxidized to [18F]-fluoroacetate by a Kuhn–Roth oxidative degradation.
![Graphical abstract: Fluorinase mediated chemoenzymatic synthesis of [18F]-fluoroacetate](/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=C0CC02264K&imageInfo.ImageIdentifier.Year=2010)
Chem. Commun., 2010,46, 7819-7821
https://doi.org/10.1039/C0CC02264K
Peptidomimetic inhibitors targeting the CCR5-binding site on the human immunodeficiency virus type-1 gp120 glycoprotein complexed to CD4
The display of two sulfotyrosine residues in the α-helical fold of CCR5 can be mimicked in a β-hairpin peptidomimetic.
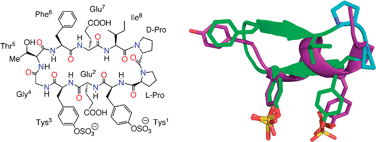
Chem. Commun., 2010,46, 7754-7756
https://doi.org/10.1039/C0CC02913K
Ligand dimerization programmed by hybridization to study multimeric ligand–receptor interactions
Peptidic ligands (16mer macrocycles) were dimerized with different geometries programmed by hybridization to recapitulate the interaction of TRAIL.
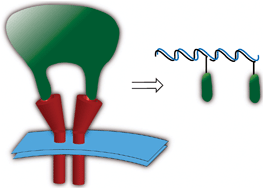
Chem. Commun., 2010,46, 7742-7744
https://doi.org/10.1039/C0CC02852E
Structure–activity relationships of the phosphonate antibiotic dehydrophos
A series of analogs of dehydrophos were prepared and tested for their antimicrobial activity.
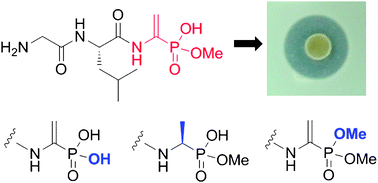
Chem. Commun., 2010,46, 7694-7696
https://doi.org/10.1039/C0CC02958K
A fluopol-ABPP HTS assay to identify PAD inhibitors
A fluopol-ABPP HTS assay identifies streptonigrin as a potent and selective PAD4 inactivator.
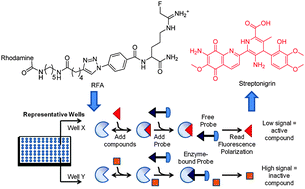
Chem. Commun., 2010,46, 7175-7177
https://doi.org/10.1039/C0CC02634D
Interaction of PqqE and PqqD in the pyrroloquinoline quinone (PQQ ) biosynthetic pathway links PqqD to the radical SAM superfamily
Data are presented that show PqqD interacting with PqqE.
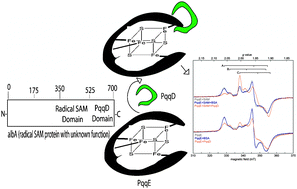
Chem. Commun., 2010,46, 7031-7033
https://doi.org/10.1039/C0CC00968G
Structural insights into substrate specificity and solvent tolerance in alcohol dehydrogenase ADH-‘A ’ from Rhodococcus ruber DSM 44541
The first structures of the widely-used alcohol dehydrogenase ADH-‘A’ provide new avenues for the structure-guided engineering of this useful enzyme.
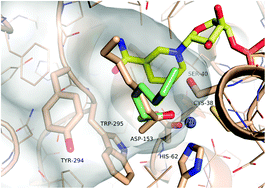
Chem. Commun., 2010,46, 6314-6316
https://doi.org/10.1039/C0CC00929F
Transaminations with isopropyl amine : equilibrium displacement with yeast alcohol dehydrogenase coupled to in situ cofactor regeneration
The unfavourable equilibrium in a transamination with isopropylamine is displaced in an enzymatic cascade reaction using alcohol dehydrogenase in situ.
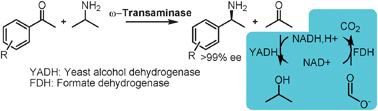
Chem. Commun., 2010,46, 5569-5571
https://doi.org/10.1039/C0CC00050G
Chemoenzymatic asymmetric total synthesis of (S)-Rivastigmine using ω-transaminases
Asymmetric total synthesis of (S)-Rivastigmine was achieved via stereoselective biocatalytic transamination of acetophenone derivatives.
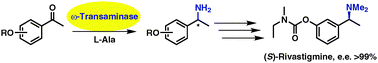
Chem. Commun., 2010,46, 5500-5502
https://doi.org/10.1039/C0CC00585A
Rational creation of mutant enzyme showing remarkable enhancement of catalytic activity and enantioselectivity toward poor substrates
Catalytic activity and enantioselectivity of lipase toward poor substrates bearing bulky substituents on both sides have been dramatically improved by rational design; the E value was increased from 5 (wild-type enzyme) to >200 (I287F/I290A double mutant) with an acceleration of the reaction rate.
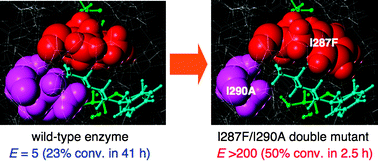
Chem. Commun., 2010,46, 5440-5442
https://doi.org/10.1039/C001561J
Inverting the enantioselectivity of P450pyr monooxygenase by directed evolution
The first example of directed evolution of a novel P450pyr monooxygenase with inverted enantioselectivity has been demonstrated.
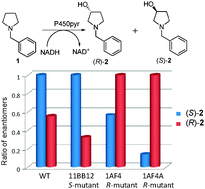
Chem. Commun., 2010,46, 5461-5463
https://doi.org/10.1039/C0CC00735H
Silver catalyzed cascade synthesis of alkaloid ring systems: concise total synthesis of fascaplysin, homofascaplysin C and analogues
A silver catalyzed and microwave assisted one-pot cascade synthesis provides efficient access to diverse alkaloid-inspired scaffold classes, and a concise and efficient total synthesis of homofascaplysin C and fascaplysin.
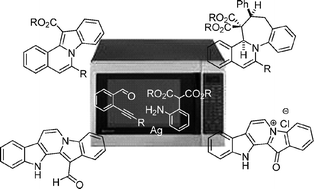
Chem. Commun., 2010,46, 4622-4624
https://doi.org/10.1039/C001350A
Enzymatically triggered amyloid formation: an approach for studying peptide aggregation
A strategy has been demonstrated that utilizes a phosphatase as a natural tool for the triggering and control of amyloid formation in a coiled coil peptide model under conditions that closely approximate a physiological environment.
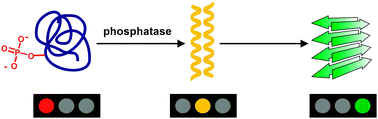
Chem. Commun., 2010,46, 3080-3082
https://doi.org/10.1039/C001460E
An unnatural amino acid that mimics phosphotyrosine
By replacing the phosphate group in pTyr with an isoxazole carboxylic acid, a novel unnatural amino acid has been successfully synthesized. Subsequently, its incorporation into a known PPI (protein–protein interaction) inhibitor of STAT3 protein (ISS 610) generated I2 which showed reasonable anti-STAT3 activity in both fluorescence polarization and cell-proliferation experiments.
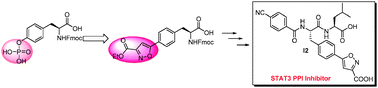
Chem. Commun., 2010,46, 2980-2982
https://doi.org/10.1039/C000283F
Solid phase total synthesis of the 3-amino-6-hydroxy-2-piperidone (Ahp) cyclodepsipeptide and protease inhibitor Symplocamide A
The Ahp cyclodepsipeptide Symplocamide A was synthesized on solid phase, using an unsaturated amino acid as a key building block for the formation of the central Ahp moiety.
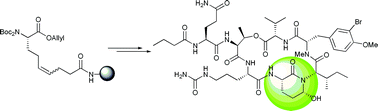
Chem. Commun., 2010,46, 8857-8859
https://doi.org/10.1039/C0CC02889D
O-Glycoligases, a new category of glycoside bond-forming mutant glycosidases, catalyse facile syntheses of isoprimeverosides
Near quantitative yields of alpha-xyloside products were obtained by coupling alpha-xylosyl fluoride with sugar acceptors using the acid/base mutant D482A of the GH31 alpha-xylosidase Yic-I
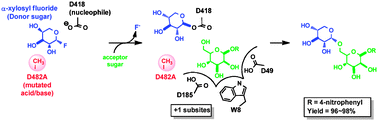
Chem. Commun., 2010,46, 8725-8727
https://doi.org/10.1039/C0CC03168B
Computational design of orthogonal nucleoside kinases
Rosetta design software was employed to remodel the substrate specificity of Drosophila melanogaster 2′-deoxyribonucleoside kinase for efficient phosphorylation of the nucleoside analog prodrug 3′-deoxythymidine.
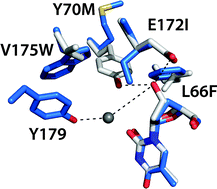
Chem. Commun., 2010,46, 8803-8805
https://doi.org/10.1039/C0CC02961K
Comparing dendritic with linear esterase peptides by screening SPOT arrays for catalysis
Direct fluorescence screening of a peptide library on a SPOT array identified oligohistidines as potent esterase peptides for the hydrolysis of acetoxypyrene trisulfonates.
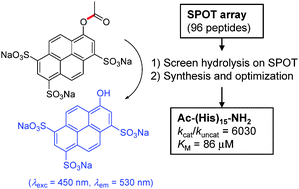
Chem. Commun., 2010,46, 8746-8748
https://doi.org/10.1039/C0CC02700F
Characterization of xenobiotic reductase A (XenA): study of active site residues , substrate spectrum and stability
Characterization on xenobiotic reductase A and its mutants for enzyme functionality and stability.
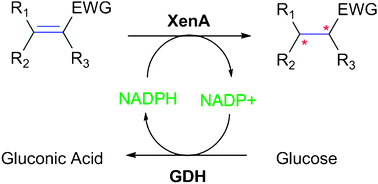
Chem. Commun., 2010,46, 8809-8811
https://doi.org/10.1039/C0CC02354J
Total chemical synthesis of human proinsulin
The protein enantiomers D- and L-proinsulin have been prepared by total synthesis using native chemical ligation.
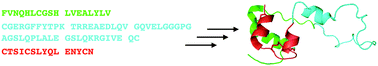
Chem. Commun., 2010,46, 8177-8179
https://doi.org/10.1039/C0CC03141K
Total chemical synthesis and X-ray structure of kaliotoxin by racemic protein crystallography
The first X-ray structure of kaliotoxin, enabled by ‘one pot’ chemical ligation of synthetic peptides and racemic protein crystallography.
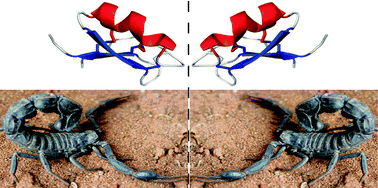
Chem. Commun., 2010,46, 8174-8176
https://doi.org/10.1039/C0CC03148H
Metabolic probes for imaging endosymbiotic bacteria within toxic dinoflagellates
We report a technique to visualize toxic dinoflagellates and suggest a preventative tool to warn of an impending danger and ensure the safe delivery of shellfish to the consumer market.
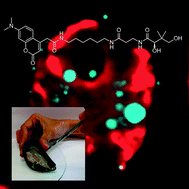
Chem. Commun., 2010,46, 8151-8153
https://doi.org/10.1039/C0CC02876B
Phage display selection of miniprotein binders of the Estrogen Receptor
Phage display selection of disulfide stabilized miniproteins identified Estrogen Receptor binders displaying an LXXLL motif on a short peptide helix.

Chem. Commun., 2010,46, 8207-8209
https://doi.org/10.1039/C0CC02727H
Engineering of an enantioselective tyrosine aminomutase by mutation of a single active site residue in phenylalanine aminomutase
An engineered tyrosine aminomutase with high enantioselectivity was constructed by homology-modelling based mutagenesis of the active site of phenylalanine aminomutase.
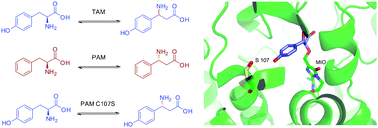
Chem. Commun., 2010,46, 8157-8159
https://doi.org/10.1039/C0CC02768E
About this collection
This web-based themed issue showcases high quality papers in the field of chemical biology, specifically research that deals with enzymes and proteins. The collated, invited, and peer-reviewed ChemComm articles highlight cutting edge contributions by international leaders in the field.
The guest editors of this web themed issue are Professors Nicholas Turner, Wilfred van der Donk and Herbert Waldmann. They are delighted by the overwhelming response that the letter of invitation produced and they express their sincere thanks to the authors. The articles received and published cover a diverse range of contemporary topics in chemical biology including biosynthesis, biocatalysis, structure/function studies, drug design and proteins as sensors.
Articles in this web themed issue will be added to the list below as soon as possible, after they are published. Please return to this page frequently to see this collection grow.