A Nafion/polybenzimidazole composite membrane with consecutive proton-conducting pathways for aqueous redox flow batteries†
Abstract
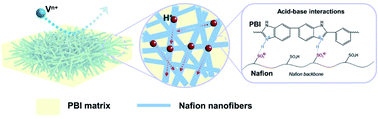
Nafion membranes are extensively used in aqueous redox flow batteries due to their high proton conductivity and excellent chemical stability, but their low ion selectivity and high cost restrict the further development of flow batteries. Here we demonstrate a composite membrane consisting of electrospun Nafion nanofibers embedded in a polybenzimidazole (PBI) matrix. The interconnected Nafion nanofibers act as consecutive proton-conducting pathways, allowing a high proton conductivity with low Nafion content, while the highly selective PBI matrix suppresses the crossover of redox species, ensuring a high ion selectivity. Moreover, the acid–base interactions between the Nafion and PBI along with the rigid nature of PBI offer excellent mechanical stability. The resultant membrane with only 40 wt% Nafion exhibits 58-fold improvement in blocking vanadium permeation and a nearly 60% retention in proton conductivity compared with the Nafion 212 membrane. More notably, the optimal membrane enables a vanadium redox flow battery to obtain a coulombic efficiency of 99.8% and an energy efficiency (EE) of 80.0% at a high current density of 210 mA cm−2, exceeding that with the Nafion 212 membrane (EE ∼ 77.2%). This work presents a new approach for developing efficient and affordable membranes for aqueous redox flow batteries.


 Please wait while we load your content...
Please wait while we load your content...