One-step synthesis of CeFeO3 nanoparticles on porous nanocarbon frameworks derived from ZIF-8 for a boosted oxygen reduction reaction in pH universal electrolytes†
Abstract
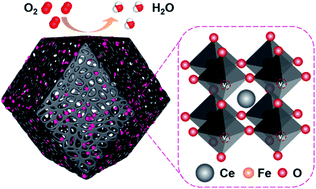
Perovskite oxide based electrocatalysts hold great promise as potential alternatives to platinum-based electrocatalysts for the oxygen reduction reaction (ORR), and thus have attracted tremendous research interest. Nevertheless, exploring high-performance ORR catalysts to improve pivotal reaction kinetics particularly in an acidic environment remains a great challenge. Herein, an N-doped porous nanocarbon framework supported perovskite CeFeO3 nanoparticle catalyst (Ce–Fe/NC) is fabricated from a predesigned Ce–Fe-doped ZIF-8 by a facile one-step route. The prepared catalyst displays a dodecahedral morphology with a large number of mesopores, which facilitate confining the aggregation of the CeFeO3 nanoparticles and promoting mass transport in the electrochemical process. Meanwhile, the introduced oxygen vacancies play a role in manipulating the local structure of the Fe active sites in CeFeO3, and boost their ORR performance. Impressively, the Ce–Fe/NC catalyst shows outstanding ORR half-wave potentials in pH universal electrolytes (0.913 V in alkaline and 0.791 V in acidic electrolytes), as well as good durability. A zinc–air battery utilizing the Ce–Fe/NC material as the air electrode offers an excellent specific capacity of 804 mA h g−1 and a long-term stability of 105 h in galvanostatic charge–discharge. This work provides a promise of non-precious-metal electrocatalysts with good prospects for realistic applications in zinc–air batteries and fuel cells.


 Please wait while we load your content...
Please wait while we load your content...