Highly efficient Ru/CeO2 catalysts for formaldehyde oxidation at low temperature and the mechanistic study†
Abstract
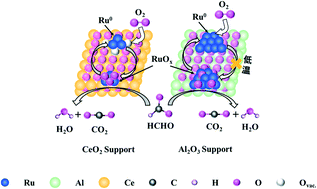
Formaldehyde (HCHO) elimination at low temperature is of great interest for indoor air purification. In this work, 1 wt% Ru supported on CeO2 and Al2O3 catalysts were prepared by an impregnation method and subsequently tested for the catalytic oxidation of HCHO at low temperature. The activity of the Ru/CeO2 catalyst was significantly superior to the Ru/Al2O3 catalyst, achieving complete conversion of 130 ppm HCHO at 90 °C with a GHSV of 100 000 mL gcat−1 h−1. Brunauer–Emmett–Teller (BET) measurements, X-ray powder diffraction (XRD), high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM), X-ray photoelectron spectroscopy (XPS) and H2-temperature-programmed reduction (H2-TPR) were carried out to characterize the physical and chemical properties of the catalysts. The results revealed that RuOx species, rather than Ru0, were directly responsible for the HCHO oxidation over Ru/CeO2, and the excellent performance of Ru/CeO2 was mainly attributed to the strong interaction between Ru and CeO2, which induced a high dispersion and high redox capacity of RuOx species. The reaction mechanism of HCHO oxidation was also investigated using in situ diffuse reflectance infrared Fourier transform spectroscopy (in situ DRIFTS). It was shown that the HCHO oxidation over the Ru/CeO2 and Ru/Al2O3 catalysts followed the same pathway of the direct formate oxidation (HCHO → HCOO + OH → H2O + CO2).


 Please wait while we load your content...
Please wait while we load your content...