Strong electron affinity PDI supramolecules form anion radicals for the degradation of organic pollutants via direct electrophilic attack†
Abstract
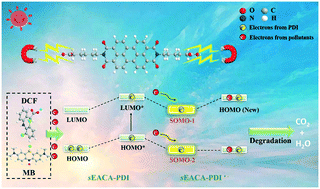
Pharmaceutical and personal care products (PPCPs) are the most common hazardous environmental pollutants. Accordingly, PDI anion radicals are promising photocatalysts to transform PPCPs into non-toxic CO2 and H2O for their clean and efficient production. In the present work, we successfully synthesized an electrophilic PDI supramolecular (sEACA-PDI) compound with high electron affinity through a simple gelation process. Its electron affinity is contributed by its side chain, which enhances the electrophilicity of its perylene core edge. Consequently, sEACA-PDI tends to form anion radicals to provide a SOMO orbital, which allows sEACA-PDI to obtain electrons successively from organic pollutants. Its side chain also generates a polarized surface electric field, building an efficient pathway for electron transfer. The high flexibility and freedom of the sEACA-PDI side chain is beneficial to form a cluster structure. Numerous strongly electrophilic groups are exposed on the surface of sEACA-PDI, which further enhance its electron affinity. sEACA-PDI exhibited high photocatalytic degradation efficiency for classical organic pollutants via successive electrophilic attack, i.e. the k for DCF was 0.923 h−1. Also, sEACA-PDI achieved a k value of 0.351 h−1 for the degradation of MB. This work proposes a promising strategy for developing PDI anion radicals with high electron affinity, and also provides new insights for the treatment of environmental pollutants.


 Please wait while we load your content...
Please wait while we load your content...