Enhanced photocatalytic activity of CO2 reduction to methanol through the use of a novel-structured CuCaAg2Se–graphene–TiO2 ternary nanocomposite†
Abstract
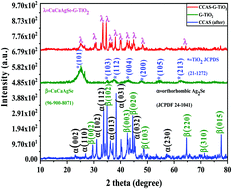
Effective charge transfer is a key factor in enhancing the catalytic activity of photocatalytic nanocomposite materials. In order to enhance the charge transfer of graphene and TiO2, we combined them with the quaternary chalcogenide (CuCaAg2Se) nanocomposite. The ternary nanocomposite was synthesized by a muffle-assisted hydrothermal method, which was used to obtain decent crystallinity and the polymer precursor solution was prepared by the Pechini method based on the metallic citrate polymerization using ethylene glycol C2H6O2 (Merck) and citric acid C6H8O7 (Merck) as the chelating cation. The morphology, structure, and charge-carrier separation analyses confirmed that the combination of CuCaAg2Se, graphene and TiO2 enhanced the photocatalytic activity of the ternary nanocomposite. All the analysis results pointed out that the ternary nanocomposite was successfully synthesized, which enhanced the performance of the nanocomposite. This ternary nanocomposite showed six times higher photoactivity performance towards CO2 reduction to generate methanol (CH3OH) under UV and visible light irradiation with different amounts of a scavenger, compared to TiO2, pure CuCaAg2Se, and the binary nanocomposite. More importantly, the as-synthesized nanocomposite displayed excellent stability during the recycling test with no obvious loss in the generation of methanol from CO2 photoreduction. The approach presented herein offers a promising route for the rational design of a new class of ternary-nanocomposites for various applications in environmental protection and solar energy conversion.


 Please wait while we load your content...
Please wait while we load your content...