Cage-confinement of gas-phase ferrocene in zeolitic imidazolate frameworks to synthesize high-loading and atomically dispersed Fe–N codoped carbon for efficient oxygen reduction reaction†
Abstract
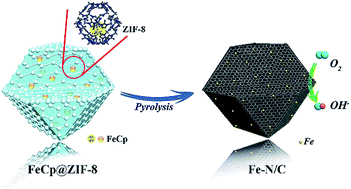
Fe–N codoped carbon (Fe–N/C) has emerged as one of the most promising non-precious electrocatalysts for the oxygen reduction reaction (ORR). However, the fabrication of Fe–N/C with high Fe and N loadings while maintaining atomic dispersion of the loaded Fe is still challenging. Herein, we present a cage-confinement synthesis strategy that utilizes zeolitic imidazolate framework-8 (ZIF-8) with an ordered microporous structure to uniformly adsorb gas-phase ferrocene (FeCp) molecules at mildly elevated temperatures followed by carbonization to fabricate Fe–N/C with a high content of atomically dispersed Fe and abundant N. The content of FeCp molecules adsorbed in the nanocavities of ZIF-8 could be precisely controlled by the adsorption temperature and time, and confining the adsorbed FeCp in the nanocavities of ZIF-8 could effectively prevent the Fe atoms from aggregating and/or forming Fe compounds during the carbonization process. The Fe–N/C fabricated under optimal conditions has a high iron loading of 5.86 wt% and abundant N content of 10.51 at%. The as-prepared Fe–N/C exhibits a remarkable ORR catalytic performance in 0.1 M KOH with a half-wave potential (E1/2) of 0.85 V (vs. RHE) and excellent long-term durability (less than 10 mV change of E1/2 after 10 000 cycles of the CV test and 11% current density decay after a 40 000 s long-term ORR test). This work provides a simple and controllable synthesis strategy for fabricating high-content atomically dispersed Fe and N codoped carbon catalysts, which might also shed light on the design and synthesis of other single-atom metal doped carbon materials for energy storage and conversion applications.


 Please wait while we load your content...
Please wait while we load your content...