Palladium(ii) and platinum(ii) complexes of glyoxalbis(N-aryl)osazone: molecular and electronic structures, anti-microbial activities and DNA-binding study†
Abstract
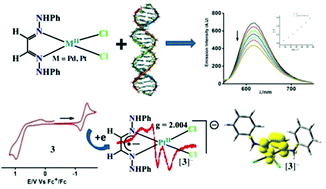
Palladium(II) and platinum(II) complexes of types [Pd(LNHPhH2)Cl2] (1), [Pd(LNH(ClPh)H2)Cl2] (2), [Pt(LNHPhH2)Cl2] (3) and [Pt(LNH(ClPh)H2)Cl2] (4) were successfully isolated, where LNHPhH2 and LNH(ClPh)H2 are osazone ligands. The molecular and electronic structures of 1–4 and their reduced analogues were confirmed by single crystal X-ray crystallography, EPR spectroscopy, and DFT calculations. Osazone is a redox non-innocent ligand and the redox activities of 1–4 were investigated by cyclic voltammetry. The redox activities of 1–4 are solvent dependent. In cyclic voltammetry, no redox wave of 1–4 is discernible in CH3CN, while in the less polar CH2Cl2 solvent, the cathodic waves of 3 and 4 gain some reversibility. Mulliken spin density analyses and EPR spectral data reveal that the unpaired electron of [3]− and [4]− ions is dominantly localized on the diimine fragment of the osazone ligands. Cell viability studied using MTT assay against leishmania promastigote shows that these compounds are strong leishmanicidal agents, while they have low anti-bacterial and anti-fungal activities. All the reported compounds are completely non-cytotoxic within the limit of 0–50 μM up to 72 h revealing their potentiality as therapeutic agents. The leishmanicidal activity of 1 and 3 is found to be higher than the ligands as well as 2 and 4. Furthermore, the interaction of 1 and 3 with DNA has been assessed as possibly intercalating in nature, which correlates with one of the requisite modes for anti-leishmanial activity.


 Please wait while we load your content...
Please wait while we load your content...