Radical addition of ketones and cyanide to olefins via acid catalyzed formation of intermediate alkenyl peroxides†
Abstract
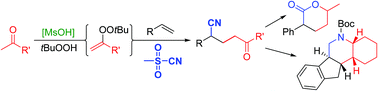
A Brønsted acid catalyzed method was developed for the synthesis of γ-cyanoketones from sulfonyl cyanides, olefins and ketones. The reaction is believed to proceed via intermediate formation of alkenyl peroxides by condensation of ketones with tert-butylhydroperoxide. These unstable compounds decompose by homolytic O–O bond cleavage, generating ketone-derived radicals which add to the olefins and generate the final products after reaction with the sulfonyl cyanide, thereby forming two new C–C bonds. A range of different ketones and olefins can be used, including steroidal ketones and simple alkyl olefins. The products can be further transformed to substituted lactones and piperidines, including a tetracyclic one. This reaction can thus be utilized to gain access to complex molecules from simple starting materials in only a few synthetic steps.
- This article is part of the themed collection: Recent Open Access Articles in Frontiers Journals


 Please wait while we load your content...
Please wait while we load your content...