Cu-exchanged RTH-type zeolites for NH3-selective catalytic reduction of NOx: Cu distribution and hydrothermal stability†
Abstract
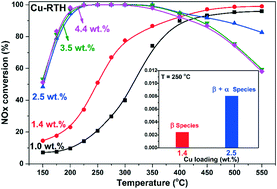
The Cu-exchanged RTH-type zeolites (Cu-RTH) were applied in ammonia-selective catalytic reduction (NH3-SCR) of NOx. The effects of Cu loading and distribution on the NOx reduction efficiency were investigated. The hydrothermal stabilities of a series of Cu-RTH catalysts were also presented. Excellent NOx conversion activity was observed for Cu-RTH catalysts with high Cu loadings (2.5–4.4 wt%) in the temperature range from 150 °C to 550 °C. Two possible sites (α and β species) for Cu2+ species were proposed based on the analysis of H2-TPR and DRIFTS results. The α species next to the 8-membered rings showed significantly higher TOF (turnover frequency) of NO conversion than the β species present next to the rth cage. The Cu-RTH catalysts showed lower activation energies (30–40 kJ mol−1) compared with that of the Cu-SSZ-13 catalyst (∼54 kJ mol−1) due to the different zeolite framework structures. After undergoing hydrothermal aging, although the zeolite framework structure stayed stable, the active Cu2+ species migrated to inactive sites, which resulted in the loss of NOx conversion for the NH3-SCR reaction. The saturated Cu2+-exchanged Cu4.4-RTH catalyst showed the best hydrothermal stability due to the limited Cu2+ mobility, which stabilized the active Cu2+ species.


 Please wait while we load your content...
Please wait while we load your content...