Towards more accurate prediction of activation energies for polyalcohol dehydrogenation on transition metal catalysts in water†
Abstract
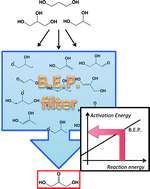
To sustain the development of novel metal-supported catalysts for transformation of polyalcohol molecules in silico, the development of efficient and accurate linear relationships to predict activation energies represents a major milestone. We establish here Brønsted–Evans–Polanyi relationships for CH and OH cleavages in monoalcohol molecules H-bonded with a chemisorbed water molecule over a series of transition metal surfaces. Then, we demonstrate that these relations can be used to predict glycerol and 1,2-propanediol dehydrogenation over Rh with remarkable accuracy and almost no systematic deviation. Lastly, we discuss the conditions to use these relations safely for screening dense catalytic reaction networks on different metallic surfaces.

 Please wait while we load your content...
Please wait while we load your content...